What is the Anthrax Vaccine Market Size in 2026?
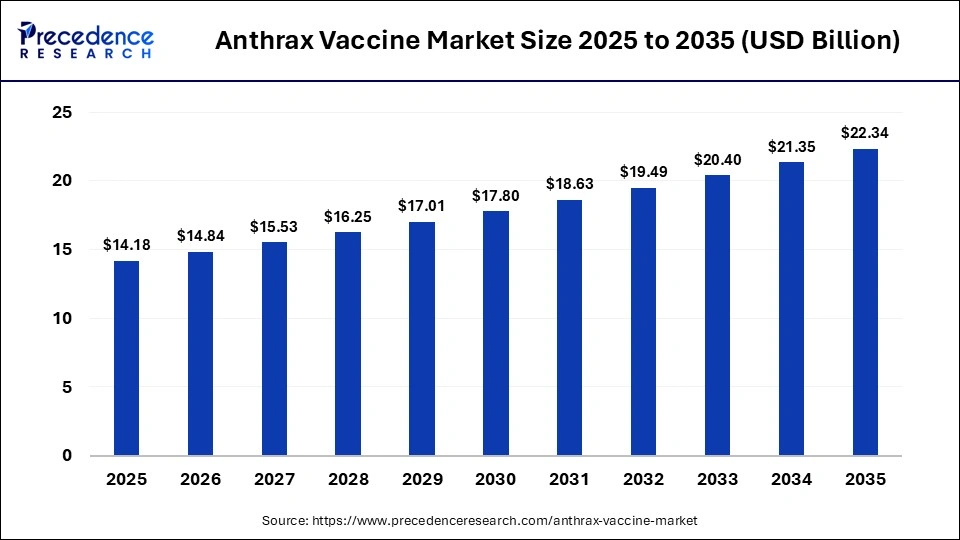
The global anthrax vaccine market size was calculated at USD 14.18 billion in 2025 and is predicted to increase from USD 14.84 billion in 2026 to approximately USD 22.34 billion by 2035, expanding at a CAGR of 4.65% from 2026 to 2035. The market is driven by increasing government focus on biodefense preparedness and rising investments in biodefense vaccine stockpiling programs.
Key Takeaways
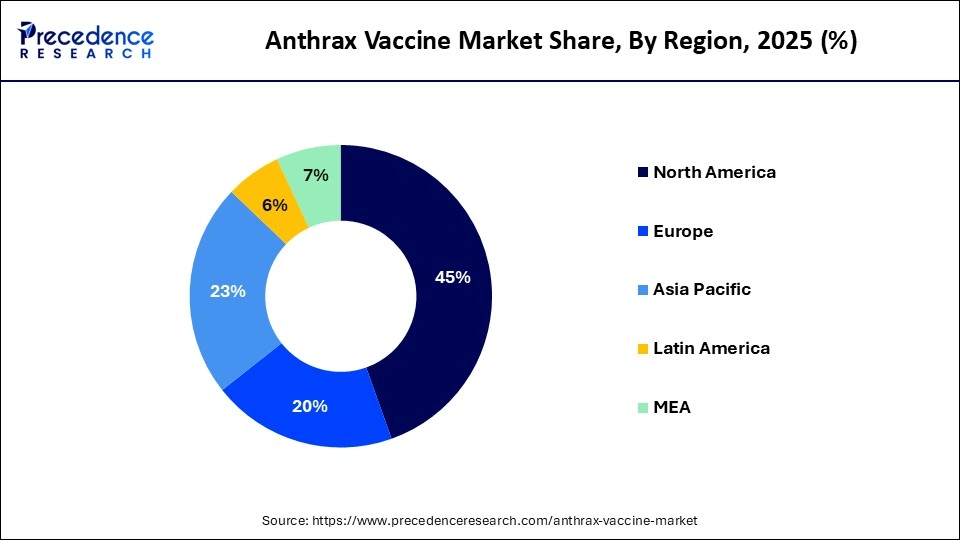
- By region, North America dominated the global market with a share of 44.70% in 2025.
- By region, Asia Pacific is expected to grow at the fastest CAGR during the forecast period.
- By vaccine platform, the cell-free protective antigen vaccine adsorbed segment held the largest market share of 63.40% in 2025.
- By vaccine platform, the recombinant protective antigen subunit vaccine segment is expected to grow at the fastest CAGR of 10.20% in the market between 2026 and 2035.
- By indication, the pre-exposure prophylaxis segment accounted for a major share of 58.70% in 2025.
- By indication, the post-exposure prophylaxis segment is expected to grow at the highest CAGR of 6.90% in the market during the studied years.
- By end user, the government stockpiles and public health agencies segment led the global market with a share of 44.60% in 2025.
- By end user, the hospitals and clinics segment is expected to expand rapidly in the market with a CAGR of 9.10% in the coming years.
- By procurement channel, the direct government procurement segment registered its dominance in the global market with a share of 52.80% in 2025.
- By procurement channel, the hospital and institutional procurement segment is expected to grow at the fastest CAGR of 8.20% over the forecast period.
Market Overview
The anthrax vaccine market consists of vaccines that are used to prevent anthrax disease caused by the bacteria Bacillus anthracis. They are vaccinated in pre-exposure and post-exposure prophylaxis, especially when the person is at high risk, including military personnel, laboratory workers, veterinarians, and emergency response teams. Anthrax vaccination works by inducing the immune system to produce antibodies against protective antigens of the bacterium, thus preventing severe infection in case of exposure. The growth of the market is mainly attributed to the rising global interest in biodefense preparedness and national security programs to safeguard the population against any possible biological attacks. Governments around the world are maintaining strategic vaccine stockpiles to ensure rapid response capabilities in the event of bioterrorism or severe disease outbreaks, contributing to the market.
How is AI Impacting the Anthrax Vaccine Market?
The anthrax vaccine market is being revolutionized by artificial intelligence (AI), driving the development of vaccines, enhancing disease surveillance, and increasing biodefense preparedness. AI-based computational models analyze the genetic structure and antigenic components of Bacillus anthracis, enabling researchers to identify effective vaccine targets and design formulations with high immunogenicity. Additionally, AI-driven simulation platforms allow preliminary testing of vaccine efficacy and safety in a shorter timeframe, reducing the need for prolonged experimental trials. Beyond R&D, AI technologies are also streamlining vaccine production, supply chain management, and inventory planning, ensuring timely distribution during emergencies or bioterrorism threats.
Anthrax Vaccine Market Trends
- Expansion of Biodefense Stockpiling Programs: Governments across the world are strengthening national biodefense strategies by stockpiling anthrax vaccines and related medical countermeasures. This trend is driven by heightened awareness of bioterrorism risks and the need for rapid emergency response capabilities, leading to increased procurement and long-term vaccine reserves.
- Focus on Next-Generation Vaccine Technologies: There is a growing shift toward the development of advanced anthrax vaccines using recombinant DNA, adjuvants, and novel delivery platforms. These next-generation candidates aim to enhance immunogenicity, extend the duration of protection, and improve safety profiles compared with traditional formulations.
- Government Funding & Biodefense: Growing government investments in biodefense preparedness are boosting anthrax vaccine demand. Many countries maintain national vaccine stockpiles and conduct preparedness activities to ensure a rapid response to potential biological threats or public health emergencies.
- R&D & Next-Generation Vaccines: Advancements in anthrax vaccine research, including recombinant and adjuvanted technologies, are influencing the market. Pharmaceutical companies and government agencies are focusing on these innovations to improve vaccine efficacy, safety, and long-term immunity.
- Expansion of Animal Vaccination Programs: Veterinary anthrax vaccines continue to see strong demand due to large-scale immunization efforts in livestock across endemic regions. Animal vaccination reduces disease transmission risks and economic losses, supporting growth in the market.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 14.18 Billion |
| Market Size in 2026 | USD 14.84 Billion |
| Market Size by 2035 | USD 22.34 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 4.65% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Vaccine Platform, Indication, End User, Procurement Channel, and region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Vaccine Platform Insights
Why Did the Cell-Free Protective Antigen Vaccine Adsorbed Segment Dominate the Anthrax Vaccine Market?
The cell-free protective antigen vaccine adsorbed segment dominated the market with the largest share of 63.40% in 2025. This is mainly due to the fact that this vaccine has long been used and has a well-established safety profile. It also has a high immunogenicity due to its protective antigen-based formulation. These vaccines are commonly used in national immunization programs and biodefense preparedness initiatives due to their ability to induce anthrax toxin immunity. Additionally, the manufacturing infrastructure and regulatory licensing for these conventional vaccines are well-established, making them easier to produce, distribute, and deploy during emergency response programs. Their long-term clinical efficacy has also fostered trust among public health authorities and government agencies globally.
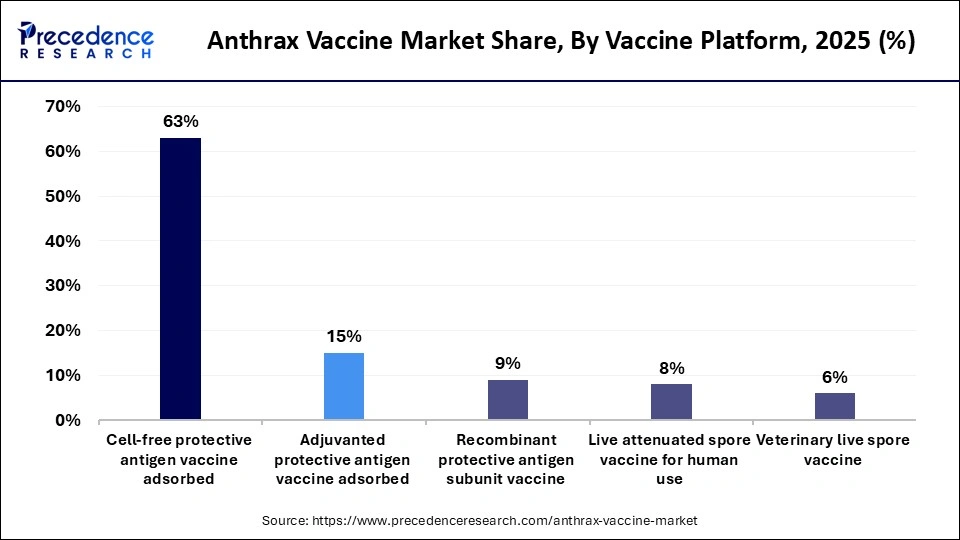
The recombinant protective antigen subunit vaccine segment is expected to grow at the fastest CAGR of 10.20% in the market between 2026 and 2035, driven by advances in recombinant biotechnology and the growing demand for next-generation vaccines with improved safety, efficacy, and scalability. These vaccines use genetically engineered protective antigen proteins to elicit strong immune responses while minimizing the risk of adverse reactions commonly associated with older vaccine formulations. Additionally, governments and healthcare organizations are increasingly investing in innovative vaccine technologies to strengthen biodefense preparedness and enhance rapid response capabilities against potential biological threats.
Indication Insights
Why Did the Pre-exposure Prophylaxis Segment Lead the Anthrax Vaccine Market?
The pre-exposure prophylaxis segment led the global market with a share of 58.70% in 2025 due to the preventive administration of anthrax vaccines in people at higher risk of being exposed to Bacillus anthracis, such as military personnel, laboratory researchers, veterinarians, and workers dealing with animal products. Government defense agencies and public health organizations prioritize pre-exposure vaccination programs to mitigate the potential impact of biological threats and bioterrorism incidents. Pre-exposure immunization provides long-term protection by priming the immune system before any exposure to Bacillus anthracis spores. The demand for these vaccines is further strengthened by large-scale procurement efforts by national stockpiles and defense departments worldwide.

The post-exposure prophylaxis segment is expected to grow at a CAGR of 6.90% between 2026 and 2035, driven by the increasing emphasis on rapid response measures for biological threats and accidental exposures. Post-exposure vaccination is administered following suspected or confirmed contact with anthrax spores to prevent disease onset. Health authorities are focusing on maintaining adequate vaccine stockpiles and establishing efficient distribution channels to ensure timely delivery during exposure incidents. Furthermore, ongoing research and development efforts are aimed at improving the efficacy and speed of immune responses generated by post-exposure vaccines, enhancing their effectiveness in emergency scenarios.
End User Insights
Why Did the Government Stockpiles and Public Health Agencies Segment Lead the Market?
The government stockpiles and public health agencies segment registered dominance in the global anthrax vaccine market with a share of 44.60% in 2025. As part of national biodefense and emergency preparedness strategies, governments actively acquire and maintain reserves of anthrax vaccines. Given that anthrax is a common biological threat agent, these stockpiles ensure a rapid response capability in the event of an outbreak or bioterrorism incident. Public health agencies manage the strategic allocation and emergency distribution of vaccines to protect high-risk populations and essential workforce members during critical situations.
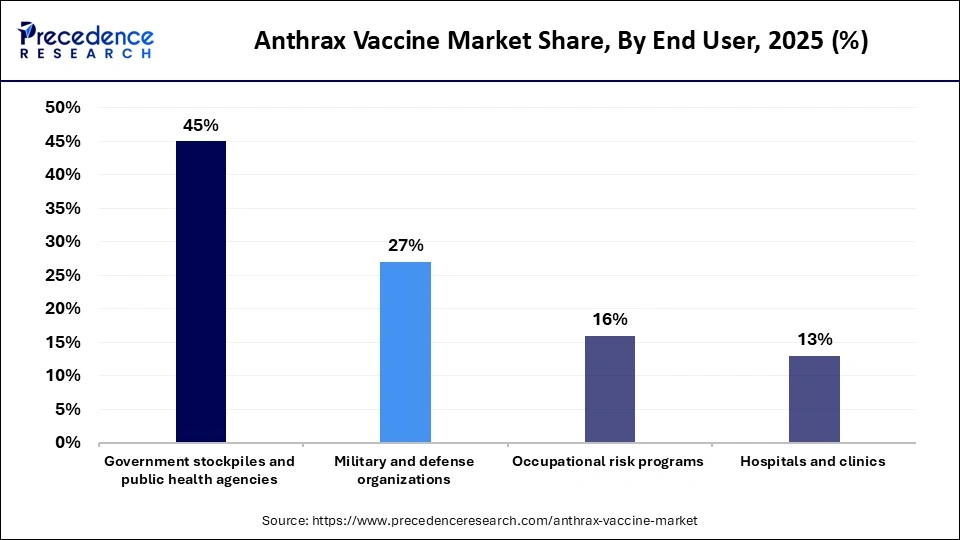
The hospitals and clinics segment is expected to expand at the fastest CAGR of 9.10% over the forecast period, owing to the increasing healthcare preparedness and the expansion of vaccination facilities within clinical settings. Hospitals and specialized healthcare centers play a critical role in administering vaccines as part of routine preventive programs as well as emergency response initiatives. They also serve as primary sites for post-exposure prophylaxis following suspected anthrax exposure. The growth of this segment is further supported by improvements in healthcare infrastructure, particularly in emerging economies, which are enhancing access to vaccination services and emergency response capabilities.
Procurement Channel Insights
Why Did the Direct Government Procurement Segment Hold a Major Share of the Market?
The direct government procurement segment led the global anthrax vaccine market while holding a major share of 52.80% in 2025, since these vaccines are critical for national security and public health preparedness. Governments purchase vaccines in large volumes through long-term contracts and supply agreements with pharmaceutical companies, ensuring a stable and reliable supply. These vaccines are primarily used for military immunization programs, emergency preparedness initiatives, and national medical stockpiles. Direct procurement also enables governments to maintain stringent quality standards, regulate distribution channels, and guarantee vaccine availability during emergencies or bioterrorism events.
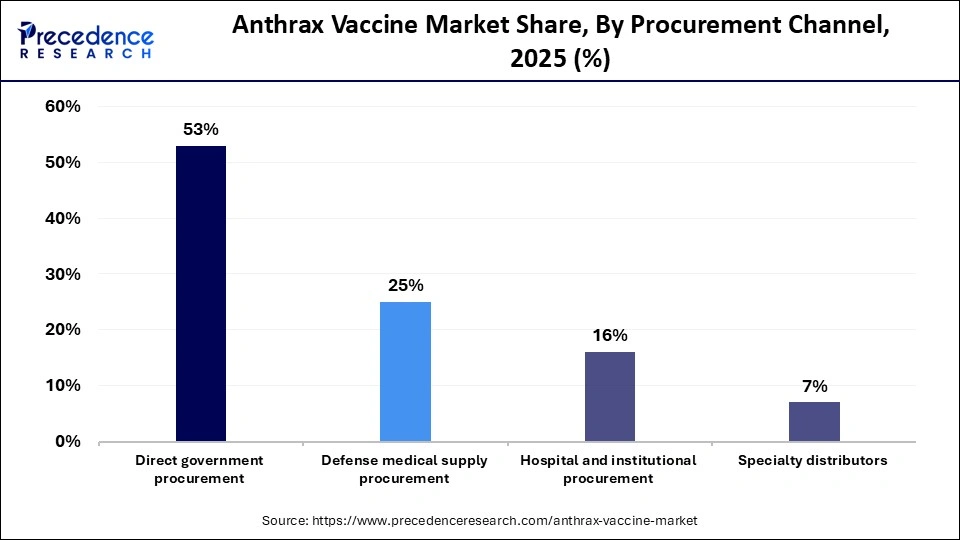
The hospital and institutional procurement segment is expected to expand at a robust CAGR of 8.20% in the coming years, driven by the increasing involvement of healthcare institutions in vaccination programs and emergency preparedness initiatives. Hospitals, research centers, and specialized healthcare facilities are progressively expanding their procurement of anthrax vaccines to support post-exposure treatment and preparedness measures. Rising awareness of bioterrorism threats has prompted healthcare institutions to strengthen their medical countermeasure inventories for rapid response to potential exposure events. Additionally, improvements in medical infrastructure and increased funding for emergency response capabilities are further encouraging organizations to invest in anthrax vaccine procurement.
Regional Insights
North America Anthrax Vaccine Market Size and Growth 2026 to 2035
The North America anthrax vaccine market size is estimated at USD 6.38 billion in 2025 and is projected to reach approximately USD 10.16 billion by 2035, with a 4.76% CAGR from 2026 to 2035.
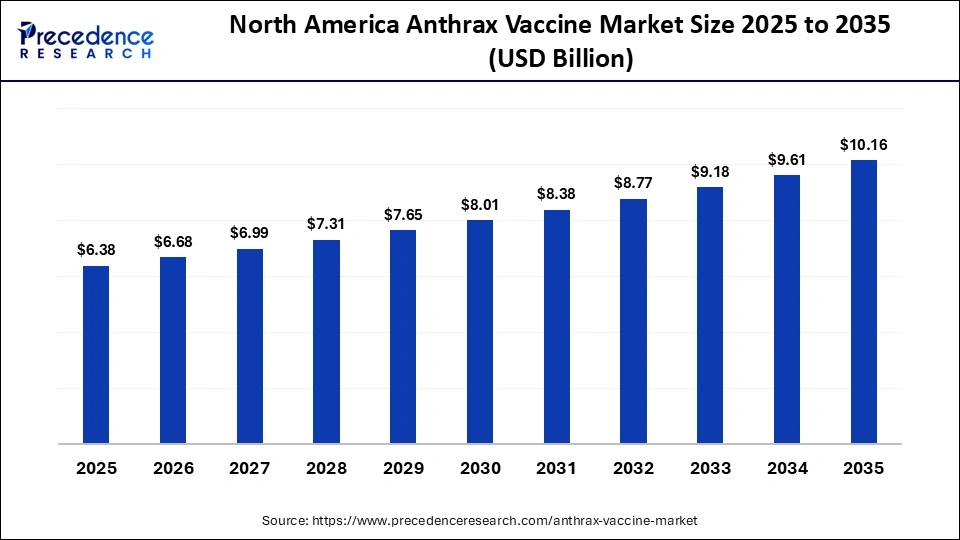
Why Did North America Lead the Global Anthrax Vaccine Market?
North America led the anthrax vaccine market by holding a 44.70% share in 2025 and is expected to sustain its position in the upcoming period. This is because of substantial government investments in biodefense preparedness and national security initiatives. The U.S., in particular, maintains extensive strategic national stockpiles of vaccines to address potential biological threats, including anthrax exposure from bioterrorism incidents. The region also benefits from access to advanced pharmaceutical research infrastructure and leading biotechnology companies, supporting the continued development and availability of anthrax vaccines. Furthermore, strong government backing for biomedical research, combined with coordinated efforts between public health agencies and defense departments, has enhanced vaccine production capabilities and optimized supply chains for rapid deployment during emergencies.
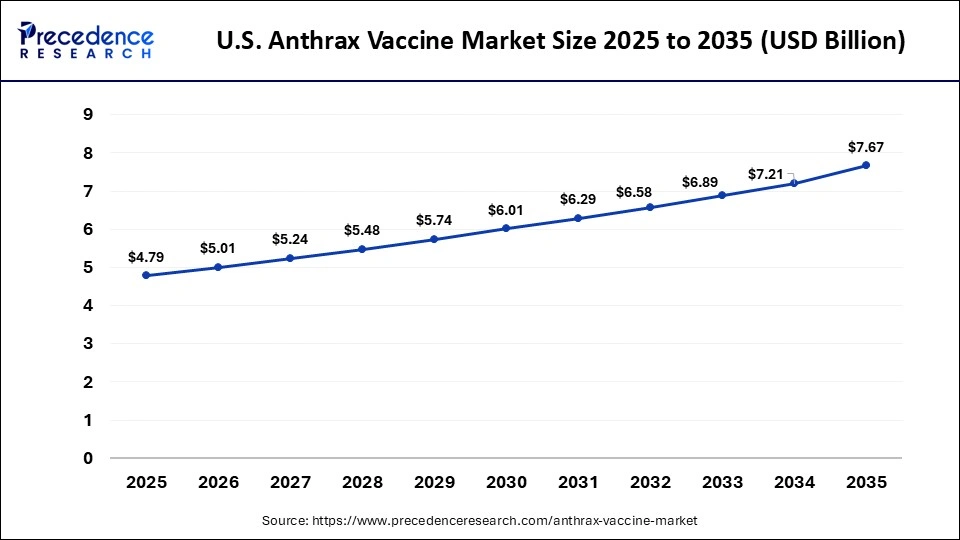
U.S. Anthrax Vaccine Market Size and Growth 2026 to 2035
The U.S. anthrax vaccine market size is calculated at USD 4.79 billion in 2025 and is expected to reach nearly USD 7.67 billion in 2035, accelerating at a strong CAGR of 4.82% between 2026 and 2035.
U.S. Market Trends
The U.S. leads the market in North America. It is driven by increased reliance on AI-driven ECUs, intelligent sensors for real-time monitoring, along with software-defined and over-the-air (OTA) update capabilities for continuous engine refinement. While gasoline engines presently dominate, there is a rapid shift toward EMS solutions monitored for hybrid vehicles to handle complex powertrain transitions.
Why is Asia Pacific Experiencing the Fastest Growth in the Anthrax Vaccine Market?
Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period due to the rising government attention toward the prevention of infectious diseases and the management of veterinary health. The high livestock populations in several countries in the region have driven the need for extensive animal vaccination programs to prevent anthrax outbreaks and reduce economic losses in the agricultural sector. Growing concerns about the transmission of zoonotic diseases are prompting governments to strengthen immunization initiatives targeting both animals and high-risk human populations. Additionally, the rapid growth of the market is supported by increased investments in public health infrastructure and ongoing advancements in biotechnology research, which enhance vaccine development and deployment capabilities.
Anthrax Vaccine Market Companies
- Emergent BioSolutions
- GC Biopharma
- Indian Immunologicals Limited
- Porton Biopharma
- Valneva
- DynPort Vaccine Company
- Altimmune
- Colorado Serum Company
- Merck
- Bayer
- Zoetis
- Botswana Vaccine Institute
- VECOL S.A.
- Biogénesis Bagó
- Agrovet Market Animal Health
Recent Developments
- In April 2025, Barythrax (GC-1109), the first recombinant anthrax vaccine in the world, co-developed with the Korea Disease Control and Prevention Agency, was approved by the Ministry of Food and Drug Safety for GC Biopharma. The acceptance enhances the biodefense capacities of South Korea and advances the next-generation development of vaccines against anthrax.
(Source: https://www.prnewswire.com) - In July 2024, Emergent BioSolutions won USD 250 million in contract extensions with the Biomedical Advanced Research and Development Authority to assist in supplying CYFENDUS to the Strategic National Stockpile. The agreement guarantees the ongoing procurement of anthrax vaccines with a view to enhancing the preparedness of the U.S. people against a preparedness of emergencies associated with health.(Source: https://investors.emergentbiosolutions.com)
- In January 2024, Emergent BioSolutions announced an indefinite delivery, indefinite quantity (IDIQ) bid to supply BioThrax as a pre-exposure prophylaxis (PrEP) vaccine against anthrax to all the branches of the United States Armed Forces. The contract will help increase the supply of vaccines among military staff members and contribute to the rise of the company's turnover.(Source: https://investors.emergentbiosolutions.com)
Segments Covered in the Report
By Vaccine Platform
- Cell-free protective antigen vaccine adsorbed
- Adjuvanted protective antigen vaccine adsorbed
- Recombinant protective antigen subunit vaccine
- Live attenuated spore vaccine for human use
- Veterinary live spore vaccine
By Indication
- Pre-exposure prophylaxis
- Post-exposure prophylaxis
By End User
- Government stockpiles and public health agencies
- Military and defense organizations
- Occupational risk programs (laboratories, first responders, animal handlers)
- Hospitals and clinics
By Procurement Channel
- Direct government procurement
- Defense medical supply procurement
- Hospital and institutional procurement
- Specialty distributors
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
