What is the Bone Densitometer Devices Market Size in 2026?
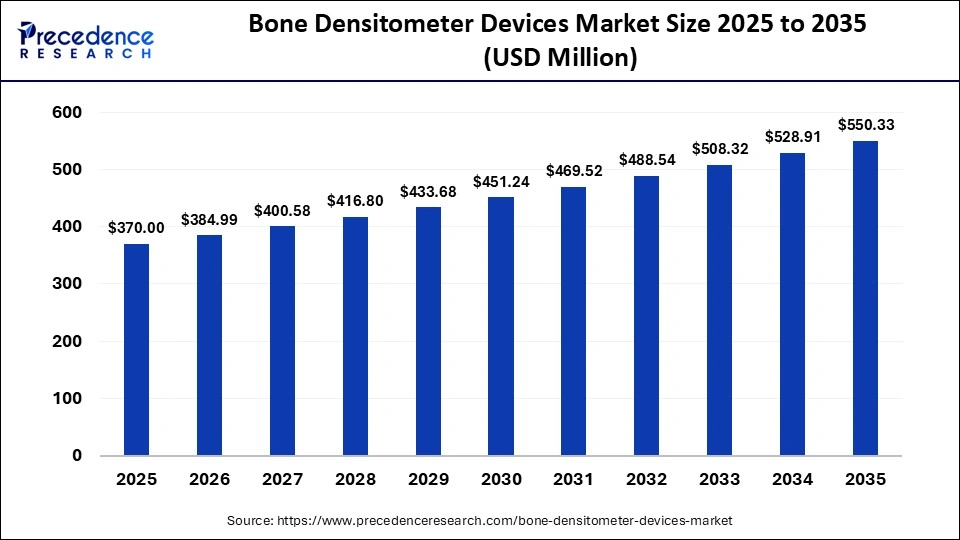
The global bone densitometer devices market size was calculated at USD 370.00 million in 2025 and is predicted to increase from USD 384.99 million in 2026 to approximately USD 550.33 million by 2035, expanding at a CAGR of 4.05% from 2026 to 2035. The market is driven by the rising prevalence of osteoporosis and bone-related disorders, the increasing geriatric population, and the growing adoption of advanced imaging technologies for early diagnosis and monitoring.
Key Takeaways
- North America led the bone densitometer devices market in 2025.
- Asia Pacific is observed to be the fastest-growing region in the forecasted period.
- By product type, the peripheral bone densitometer segment led the market in 2025.
- By product type, the axial bone densitometer segment is observed to grow at the fastest rate in the coming years.
- By technology, the dual energy X-ray absorptiometry segment led the global market in 2025.
- By technology, the quantitative ultrasound (QUS) segment is expected to grow at a significant CAGR in the foreseen period.
- By end-user, the hospitals segment led the global market in 2025.
- By end-user, the diagnostic center segment is expected to grow at the fastest CAGR in the upcoming period.
What Drives the Bone Densitometer Devices Market?
The increasing prevalence of bone density disorders, particularly osteoporosis, is contributing to the growing demand for bone densitometer devices. A bone densitometer measures X-ray absorption by bone and soft tissue to determine bone mineral density, and can also assess soft tissue density, lean-to-fat ratio, and total body mass. Osteoporosis often remains undiagnosed even after trauma, which highlights the importance of timely and accurate diagnosis. As a result, early detection of bone health conditions has become a key focus within healthcare systems. Furthermore, the rising geriatric population, along with increasing awareness of bone health, is encouraging early diagnosis and proactive management of bone density disorders, thereby supporting the growth of the bone densitometer market.
How is Artificial Intelligence Impacting the Bone Densitometer Devices Market?
Artificial intelligence is increasingly being integrated into bone health diagnostics, improving the detection and assessment of bone density disorders. AI-assisted tools have demonstrated strong diagnostic performance and are considered useful not only in hospitals and clinics, but also for screening individuals in the general population. For instance, researchers in Japan developed an AI-assisted diagnostic system capable of estimating lumbar spine and femoral bone mineral density using anteroposterior X-ray images.
Additionally, the integration of healthcare IT technologies such as machine learning, deep learning, and digital detectors is supporting the advancement of bone health diagnostics. Several imaging-based AI tools are now being explored for opportunistic osteoporosis diagnosis, and recent systematic reviews and meta-analyses indicate that these AI algorithms can achieve diagnostic performance comparable to clinical experts, with combined AI and specialist expertise further improving diagnostic outcomes.
Overall, AI is influencing the bone densitometer devices market by improving the accuracy and speed of bone density analysis, enabling earlier detection of conditions such as osteoporosis. AI-powered algorithms automatically analyze imaging data, identify fracture risks, and assist clinicians in making more precise diagnostic and treatment decisions.
Bone Densitometer Devices Market Trends
- The increasing integration of AI-assisted diagnostic systems and advanced imaging technologies is improving bone mineral density estimation from X-ray images, supporting early osteoporosis screening and clinical decision-making.
- Growing adoption of axial and peripheral bone densitometers, along with technologies such as quantitative ultrasound and digital X-ray absorptiometry, is expanding accessible and efficient bone density assessment.
- Manufacturers are developing compact and handheld densitometers that can be used in outpatient clinics, community health programs, and rural healthcare settings. Portable systems improve accessibility to bone health screening and enable broader population-level osteoporosis detection.
- Healthcare systems are increasingly emphasizing early detection of osteoporosis, particularly among aging and postmenopausal populations. This shift toward preventive healthcare is increasing the adoption of densitometry in routine screenings across hospitals, diagnostic centers, and community healthcare programs.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 370.00 Million |
| Market Size in 2026 | USD 384.99 Million |
| Market Size by 2035 | USD 550.33 Million |
| Market Growth Rate from 2026 to 2035 | CAGR of 4.05% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, Technology, End-User, and region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Product Insights
How Did the Peripheral Bone Densitometer Segment Dominate the Market in 2025?
The peripheral bone densitometer segment dominated the bone densitometer devices market with the largest share in 2025. This is mainly due to its lower cost, faster scanning speed, and portability. Peripheral densitometers are small, portable devices that can be used in various settings beyond hospitals. These systems are designed to assess bone mineral density in appendicular sites such as the heel, wrist, and forearm. Owing to their affordability and ease of use, they are often utilized in primary care settings, community screening programs, and mobile health units. Their compact design allows their use even in small healthcare setups, making bone density assessment more accessible. These advantages support wider adoption and contribute to the growth of the segment in the market.
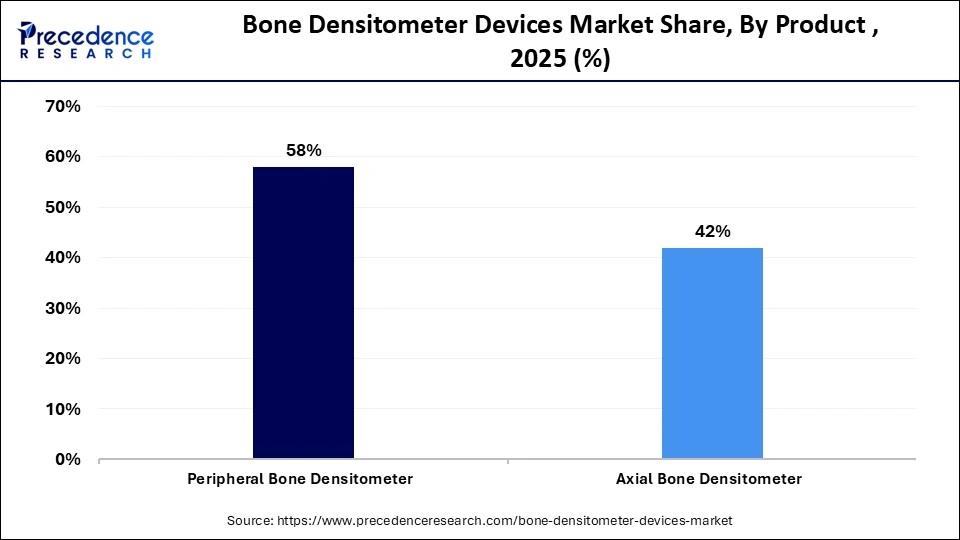
The axial bone densitometer segment is expected to grow at the fastest CAGR during the forecast period due to its increased adoption rate, higher diagnostic accuracy, and integration of advanced imaging technologies. These systems are widely used in clinical settings, as they enable precise evaluation of bone mineral density, particularly in central skeletal sites such as the spine and hip. The segment predominantly includes central dual-energy X-ray absorptiometry (DXA) tools, which are considered essential for obtaining reliable and accurate results in diagnostic procedures. In addition, quantitative computed tomography (QCT) provides a 3D assessment of bone density and offers more detailed insights compared to traditional DXA. Through these capabilities, axial bone densitometry fulfills different requirements of clinicians and supports effective diagnostic evaluation of bone health.
Technology Insights
Why Did the Dual Energy X-Ray Absorptiometry (DEXA) Segment Lead the Market?
The dual-energy X-ray absorptiometry (DEXA) segment led the bone densitometer devices market in 2025. The segment's dominance is attributed to its high precision, strong reproducibility, and ability to deliver detailed diagnostic information, which has supported its wide adoption in both clinical and research settings for bone density assessment. DEXA uses a very small dose of ionizing radiation to generate images of the internal structure of the body and to measure bone loss. It may also be indicated for the diagnosis, staging, and follow-up of individuals with conditions that result in pathologically increased bone mineral density (BMD), such as osteopetrosis or prolonged exposure to fluoride. In addition, it may be used to assess regional and whole-body fat and lean mass in patients with malabsorption, cancer, or eating disorders. These benefits are promoting the growth of the segment.
The quantitative ultrasound (QUS) segment is projected to grow at a significant rate during the forecast period. This is because of its offerings, like relatively low cost per test, and the equipment is compact, handheld, portable, and cost-effective. These devices are designed for convenient use and do not require a dedicated technician to conduct the test. In addition, QUS techniques are radiation-free, eliminating radiation-related hazards while ensuring safe and user-friendly operation.
The portable design further enables their use in diverse clinical and screening settings for the assessment of bone mineral density. However, a key limitation in the clinical application of QUS is that evaluation outcomes can be influenced by the actual acquisition conditions of the RF echo signal. Despite this, ongoing technological improvements are helping to mitigate these challenges.
End-User Insights
How Does the Hospitals Segment Dominate the Market?
The hospitals segment dominated the bone densitometer devices market while holding a major share in 2025 due to advanced diagnostic facilities and comprehensive patient care infrastructure. Hospitals provide access to multidisciplinary teams, including orthopedic physicians, surgeons, radiologists, endocrinologists, and pediatricians, enabling detailed evaluation of bone mineral density and related conditions. The presence of specialized professionals supports early diagnosis and more accurate clinical assessment. In addition, hospitals offer integrated care, combining bone density evaluation with the assessment of associated complications and treatment planning. The availability of skilled staff and advanced medical equipment further enhances device utilization, driving strong adoption of bone densitometry systems in hospital settings.
The diagnostic centers segment is expected to grow at a significant CAGR over the projection period due to increasing investments in advanced diagnostic equipment such as Dual Energy X-ray Absorptiometry (DXA), Peripheral Dual Energy X-ray Absorptiometry (pDXA), Quantitative Ultrasound (QUS), and the development of well-equipped infrastructure. These centers often employ trained technicians who can perform specialized diagnostic procedures efficiently. In addition, many diagnostic centers maintain collaborations or referral networks with physicians and specialists, enabling patients to receive appropriate recommendations for bone density testing. Supportive regulatory initiatives and the growing focus on early disease detection are further encouraging the expansion of diagnostic facilities, contributing to the rapid growth of this segment in the market.
Regional Insights
What Made North America the Dominant Region in the Bone Densitometer Devices Market?
North America dominated the bone densitometer devices market by capturing the largest share in 2025. This is mainly due to high healthcare spending, well-developed medical infrastructure, and continuous advancements in diagnostic technologies. The increased prevalence of osteoporosis, particularly among the aging population, is further driving demand for bone density testing across the region. Growing awareness of bone health and the importance of early diagnosis is also encouraging more individuals to undergo routine screening. Countries such as the U.S., Canada, and Mexico are contributing to regional market growth, driven by favorable government policies and the presence of leading medical device manufacturing companies.
U.S. Market Analysis
Among the countries in the region, the U.S. holds a leading position in the North American market, supported by a large patient pool and strong healthcare facilities. The widespread adoption of bone health screening programs, especially for postmenopausal women and elderly individuals, is significantly contributing to market growth. In addition, continuous investment in advanced imaging technologies and supportive reimbursement frameworks is accelerating the adoption of bone densitometer devices across hospitals and diagnostic centers in the region.
How is the Opportunistic Rise of Asia Pacific in the Bone Densitometer Devices Market?
Asia Pacific is expected to grow at the fastest rate in the market, driven by rapid urbanization, rising per capita income, and continuous improvements in healthcare infrastructure. Growing awareness of bone health and the importance of early diagnosis is further supporting the adoption of bone density testing. In addition, government initiatives, including awareness programs on bone disorders and preventive healthcare, are expanding access to diagnostic services.
China Market Analysis
China is considered a major contributor to regional market growth. Increasing use of bone densitometer devices among individuals over 50 years of age, particularly women, is driving demand. The rising burden of bone-related disorders, combined with a rapidly expanding geriatric population, is further accelerating market expansion. According to estimates from the World Health Organization, China could have more than 400 million people affected by some form of bone disorder by 2040, underscoring the need for enhanced diagnostic capabilities.
India Market Analysis
India is also projected to record strong growth in the bone densitometer devices market during the forecast period. Rising disposable incomes, greater adoption of advanced medical technologies, and ongoing improvements in healthcare infrastructure are supporting market development. Additionally, the establishment of approximately 150,000 Health and Wellness Centers under government healthcare initiatives is improving access to medical services and early diagnostic support, which is likely to encourage wider use of bone densitometer devices across the country.
Bone Densitometer Devices Market Value Chain Analysis
- R&D
This stage focuses on improving diagnostic accuracy, minimizing radiation exposure, and broadening access via portable, AI-enabled technologies.
Key Players: Bone Health Technologies, OsteoSys Corp
- Clinical Trials and Regulatory Approvals
Clinical and regulatory frameworks are shifting to AI-driven software and surrogate endpoints, expediting diagnostics and therapeutics.
Key Players: GE HealthCare, Echolight, Bone Health Tech
- Packaging and Serialization
Regulated by ISO 11607 and 21 CFR 801, ensuring protected, compliant, and fully traceable diagnostic devices.
Key Players: GE HealthCare, DMS Imaging, OsteoSys Corp
- Distribution
Uses direct sales teams for advanced central DXA systems and third-party distributors for portable devices.
Key Players: GE HealthCare, Hologic Inc., DMS Imaging
- Support and Services
After-sales services extend device life and accuracy via preventive maintenance, AI updates, and remote calibration.
Key Players: GE HealthCare, Hologic Inc.
Bone Densitometer Devices Market Companies
- GE HealthCare (U.S.)
- Hologic Inc. (U.S.)
- DMS Imaging (France)
- OsteoSys Corp (South Korea)
- Swissray International (Switzerland)
- Scanflex Healthcare AB (Sweden)
- BeamMed Ltd (Israel)
- Trivitron Healthcare (India)
- Diagnostic Medical Systems (DMS) Group (France)
- Shenzhen XRAY Electric (China)
- Echolight S.p.A. (Italy)
- Medilink (France)
- Fujifilm Holdings Corp (Japan)
- Hitachi Ltd (Japan)
- Eurotec Medical Systems Srl (Italy)
Recent Developments
- In 2025, Osteoboost Health launched an FDA-cleared prescription wearable device for low bone density, addressing a critical treatment gap for more than 60 million Americans and bridging next-generation consumer health technology with rigorously tested medical innovation.(Source: https://www.businesswire.com)
- In November 2024, Echolight signed a reseller agreement with Siemens Healthineers to distribute Echolight's REMS-based bone densitometers, an ultrasound technology that measures bone density and microarchitecture without radiation, enabling safer and repeatable bone health monitoring.(Source: https://www.prnewswire.com)
- In August 2024, OsteoSys Co., Ltd. received approval under the European Union Medical Device Regulation 2017/745 (EU MDR), enabling its bone mineral densitometer (BMD) devices and related software to meet the EU's stringent safety and quality standards for medical devices.(Source: https://osteosys.com)
Segment Covered in the Report
By Product
- Peripheral Bone Densitometer
- Axial Bone Densitometer
By Technology
- Dual Energy X-Ray Absorptiometry (DXA)
- Peripheral Dual Energy X-Ray
- Absorptiometry (pDXA)
- Quantitative Ultrasound (QUS)
- Others
By End-User
- Hospitals
- Orthopedic Clinics
- Diagnostic Centers
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
