What is the CRISPR-Based Diagnostics Market Size?
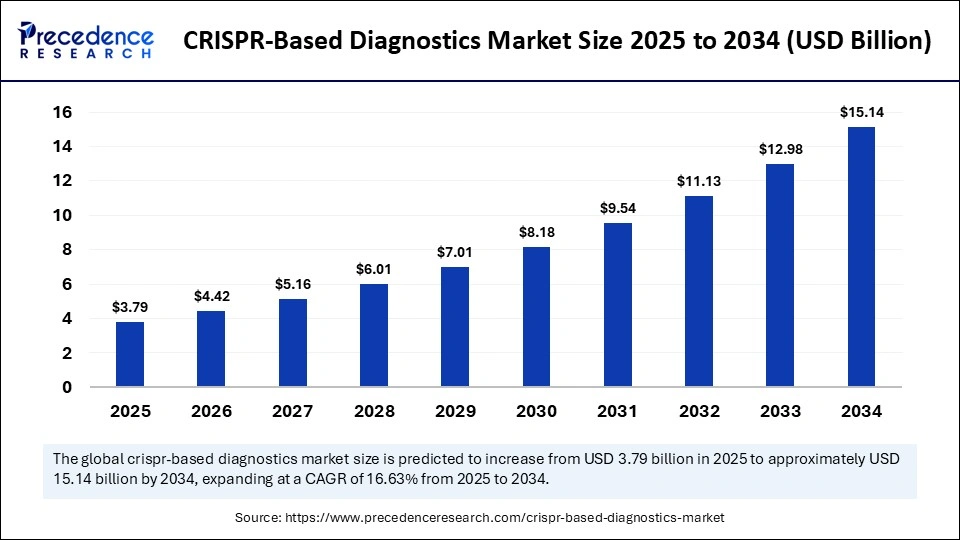
The global CRISPR-based diagnostics market size is anticipated at USD 3.79 billion in 2025 and is predicted to increase from USD 4.42 billion in 2026 to approximately USD 15.14 billion by 2034, expanding at a CAGR of 16.63% from 2025 to 2034. The market growth is attributed to growing disease burden, increasing demand for accurate diagnostics and point-of-care testing, as well as growing innovations and the application of CRISPR-based diagnostics.
Market Highlights
- North America dominated the global CRISPR-based diagnostics market holding more than 37% of market share in 2024.
- Asia Pacific is expected to be the fastest-growing at a notable CAGR from 2025 to 2034.
- By product type, the kits & assays segment held the major market share in 2024, accounting for 44%.
- By product type, the services segment is expected to be the fastest growing at a CAGR from 2025 to 2034.
- By technology/CRISPR platform, the Cas12-based diagnostics segment contributed the biggest market share in 2024.
- By technology/CRISPR platform, the Cas13-based diagnostics segment is expected to be the fastest growing from 2025 to 2034.
- By detection method/readout, the fluorescence-based detection segment dominated the CRISPR-based diagnostics market in 2024.
- By detection method/readout, the optical & imaging-based detection is expected to be the fastest growing from 2025 to 2034.
- By application, the infectious disease detection segment dominated the global market with the largest share in 2024.
- By application, the oncology biomarker testing segment is expected to be the fastest growing from 2025 to 2034.
- By distribution channel, the direct sales to hospitals, labs & research centers segment held the major market share in 2024.
- By distribution channel, the online platforms & e-commerce segment is expected to be the fastest growing from 2025 to 2034.
Market Overview
The CRISPR-Based Diagnostics Market encompasses products, platforms, and services leveraging Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) technology for the rapid, accurate, and cost-effective detection of nucleic acids associated with infectious diseases, genetic disorders, oncology biomarkers, and other conditions. CRISPR diagnostics use guide RNA (gRNA)-directed Cas enzymes (e.g., Cas12, Cas13, Cas9) to recognize specific DNA/RNA sequences and generate detectable signals through fluorescence, colorimetry, or electrochemical readouts. The market is driven by increasing demand for point-of-care (POC) testing, advancements in CRISPR platforms like SHERLOCK, DETECTR, and FELUDA, growing investments in genomics, and rising infectious disease outbreaks.
Impact of Artificial Intelligence on the CRISPR-Based Diagnostics Market
The precision of the CRISPR-based diagnostics can be enhanced by integrating it with AI. With the use of AI, the accuracy of CRISPR can be improved for disease detection. At the same time, the effectiveness of the CRISPR-based diagnostic can also be improved using AI. A vast amount of genomic data can be analyzed by AI, offering precision in the disease diagnosis by CRISPR. The disease risk prediction and complex genetic information are also provided by AI, promoting early diagnosis. Thus, by integrating AI, the diagnostic accuracy can be enhanced.
CRISPR-Based Diagnostics Market Growth Factors
- Growing diseases: Due to growing incidences of diseases, there is a rise in the demand for early and accurate diagnostics, which is increasing the use of CRISPR-based diagnostics.
- Growth in research and development: Due to growing R&D for developing therapeutic approaches, the use of CRISPR-based diagnostics is increasing, where they are also focusing on enhancing its applications.
- Rising use for point-of-care testing: The CRISPR-based diagnostics are increasingly being used for emergency situations, remote areas, and home testing for rapid and accurate diagnosis of diseases.
- Increasing demand for personalized medicine: Due to growing demand for personalized medicine, the use of CRISPR-based diagnostics is increasing, as it can identify the genetic information of the patient, promoting their development.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 3.79 Billion |
| Market Size in 2026 | USD 4.42 Billion |
| Market Size by 2034 | USD 15.14 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 16.63% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Product Type, Technology/CRISPR Platform, Detection Method/Readout, Application, Distribution Channel, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Why is the Growing Demand for Accurate Diagnostics a Driver in the CRISPR-Based Diagnostics Market?
Due to growing diseases, there is a rise in the demand for accurate diagnoses. This, in turn, increases the use of CRISPR-based diagnostics due to their high sensitivity and specificity. This helps in detecting the disease rapidly with improved accuracy. It also helps in the detection of mutating targets, which in turn increases their use for early diagnosis of various diseases. At the same time, it also offers reproducible diagnostics, which is increasing its use. Thus, this drives the CRISPR-based diagnostics market growth.
Restraint
High cost
For the development of CRISPR-based diagnostics, specialized equipment are required along with recurrent use of reagents. This increases the cost of their development, making it expensive. This limits the adoption of the diagnostics by hospitals with limited infrastructure or in remote areas. Thus, high cost restrains the use of CRISPR-based diagnostics.
Opportunity
How Does Growing CRISPR-Based Diagnostics Innovation Expand Opportunities in the CRISPR-Based Diagnostics Market?
There is a growth in the innovations of CRISPR-based diagnostics to enhance their accuracy and applications. Different types of platforms are being developed to improve their speed and sensitivity. At the same time, innovations are also being made to promote its use as a point-of-care diagnostic solution or for home testing. Additionally, the industries are also focusing on using it for the detection of multiple targets simultaneously, for developing multiplexing diagnostics. Similarly, portable CRISPR-based diagnostics are also being developed. Thus, these innovations are promoting the CRISPR-based diagnostics market growth.
CRISPR-Based Diagnostics Market Segment Insights
[[segment_insights]]
CRISPR-Based Diagnostics Market Regional Insights
[[regional_insights]]
Quick Picks by Regional Insights for CRISPR-Based Diagnostics Market
| Company | Country | Product/Platform | Uses |
| Sherlock Biosciences | U.S. | SHERLOCK/INSPECT CRISPR-based assay | Rapid, point-of-need detection of infectious diseases (e.g. COVID-19) using CRISPR-Cas13/12 technology. |
| Mammoth Biosciences | U.S. | Ultra-small Cas systems (Cas12, Cas14) for diagnostics | Development of next-generation CRISPR diagnostics platforms with enhanced sensitivity and versatility. |
| CrisprBits | India | PathCrisp CRISPR diagnostics platform | Affordable CRISPR-based diagnostics for infectious diseases and rare disease screening in resource-constrained settings. |
| CASPR Biotech | Argentina/U.S. | Portable CRISPR-Cas12 device | Low-cost CRISPR diagnostics tool intended for infectious disease detection via new Cas enzymes from extremophiles. |
| Thermo Fisher Scientific | U.S. | CRISPR-based molecular diagnostics reagents & platforms | Supplies materials and platforms supporting CRISPR diagnostics development (drives ecosystem). |
CRISPR-Based Diagnostics Market Vlaue Chain
[[value_chain]]
CRISPR-Based Diagnostics Market Companies
[[market_company]]
Other Companies in the CRISPR-Based Diagnostics Market
- Caspr Biotech: Caspr Biotech develops portable CRISPR-Cas diagnostic devices for rapid and accurate detection of infectious pathogens. Its field-deployable diagnostic kits are designed to operate in resource-limited settings, providing high specificity and affordability. Casprs CRISPR systems are being adapted for use in emerging disease surveillance and public health response programs.
- CRISPR Diagnostics Ltd.: CRISPR Diagnostics Ltd. specializes in developing next-generation CRISPR-based point-of-care platforms for infectious disease detection. The companys microfluidics-integrated systems enable multiplexed, high-throughput CRISPR assays with quick turnaround times, addressing growing demand for fast and scalable molecular testing.
- Inscripta, Inc.: Inscripta provides CRISPR-enabled genome engineering platforms, including its Ony Digital Genome Engineering System, which supports assay optimization for diagnostic applications. Inscripta automated platform helps accelerate the design and validation of CRISPR-based diagnostic assays, enhancing accuracy and development speed for research and clinical use.
- Synthego Corporation: Synthego is a major provider of CRISPR reagents, synthetic guide RNAs, and genome editing tools, supporting diagnostic developers worldwide. The companys automation-driven workflows and synthetic RNA capabilities facilitate rapid prototyping and optimization of CRISPR-based detection systems.
- Danaher Corporation (Cepheid & IDT): Through subsidiaries Cepheid and Integrated DNA Technologies (IDT), Danaher plays a key role in CRISPR diagnostics. Cepheid provides molecular testing instruments and consumables, while IDT supplies CRISPR enzymes, gRNAs, and oligonucleotides essential for CRISPR assay development. This synergy positions Danaher as a strategic enabler of CRISPR diagnostic commercialization.
- Thermo Fisher Scientific Inc.: Thermo Fisher provides a comprehensive portfolio of molecular biology tools, Cas enzymes, and assay development kits used in CRISPR-based diagnostics. Its infrastructure supports scalable manufacturing of CRISPR components and integration with qPCR and sequencing platforms for advanced molecular detection.
- Merck KGaA (Sigma-Aldrich): Mercks Sigma-Aldrich brand supplies high-quality CRISPR-Cas nucleases, guide RNAs, and assay reagents for diagnostics and life sciences research. The companys global production capacity and regulatory compliance make it a preferred supplier for CRISPR assay developers.
- Agilent Technologies, Inc.: Agilent provides analytical instruments, molecular biology reagents, and PCR systems that complement CRISPR-based diagnostic workflows. Its precision detection and assay validation tools are instrumental in ensuring reproducibility and clinical-grade performance of CRISPR tests.
- Bio-Rad Laboratories, Inc.: Bio-Rads digital PCR and molecular diagnostic technologies play a crucial role in CRISPR-based assay quantification and validation. Its Droplet Digital PCR (ddPCR) systems enable ultra-sensitive nucleic acid quantification, supporting the verification of CRISPR diagnostic performance.
- Ginkgo Bioworks: Ginkgo Bioworks applies synthetic biology to enhance CRISPR enzyme discovery and optimization for diagnostics. Its biofoundry platform supports high-throughput screening and design of CRISPR-based detection systems, enabling customized solutions for diagnostic developers.
- GenScript Biotech Corporation: GenScript supplies custom CRISPR reagents, enzymes, and molecular biology components essential for CRISPR-based diagnostic research. Its global logistics network and manufacturing expertise support large-scale assay development and commercialization.
- F. Hoffmann-La Roche Ltd.: Roche integrates CRISPR-based detection into its advanced molecular diagnostics portfolio. Leveraging its strengths in PCR, sequencing, and digital health technologies, Roche is exploring CRISPR diagnostics for infectious disease, oncology, and genetic testing, with an emphasis on clinical-grade validation and scalability
Recent Developments
- In January 2025, in Bengaluru, a CRISPR gene editing and diagnostics laboratory was inaugurated by CrisprBits Private Limited, which is a Bengaluru-based biotechnology startup. The new facility consists of advanced infrastructure, which includes spaces for cell and tissue culture, molecular biology research, cleanroom operations, lyophilization, and bacterial culture. Moreover, to support the stem cell research specialized tissue culture area and a gene editing section will also be provided in this laboratory. (Source: https://www.prnewswire.com)
- In June 2025, a follow-on investment from Oost NL and SHIFT was received by Scope Biosciences B.V. A total of €2.5 million EIC Transition Grant was also provided to the company. Thus, a total of €6 million in seed-stage funding was received by ScopeBio by combining venture and strategic capital investments with non-dilutive support from leading Dutch and European innovation programs. These investments will be used by the company to accelerate the development of its next-gen CRISPR-based molecular diagnostics platform, which is scopeDx, to offer field-ready, ultra-precise, portable, single-nucleotide detection, eliminating the need for centralized labs and proprietary hardware, which is the ultimate mission of ScopeBio. (Source: https://www.wur.nl)
CRISPR-Based Diagnostics Market Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
