What is Diagnostic Contract Manufacturing Market Size in 2026?
The global diagnostic contract manufacturing market size was calculated at USD 33.45 billion in 2025 and is predicted to increase from USD 37.76 billion in 2026 to approximately USD 112.25 billion by 2035, expanding at a CAGR of 12.87% from 2026 to 2035. The market is witnessing high growth, mainly because of the increasing demand for diagnostic assays, kits, and reagents globally. Increasing rates of chronic diseases, the growth of personalised medicine, and the emphasis on early disease detection are also contributing factors.
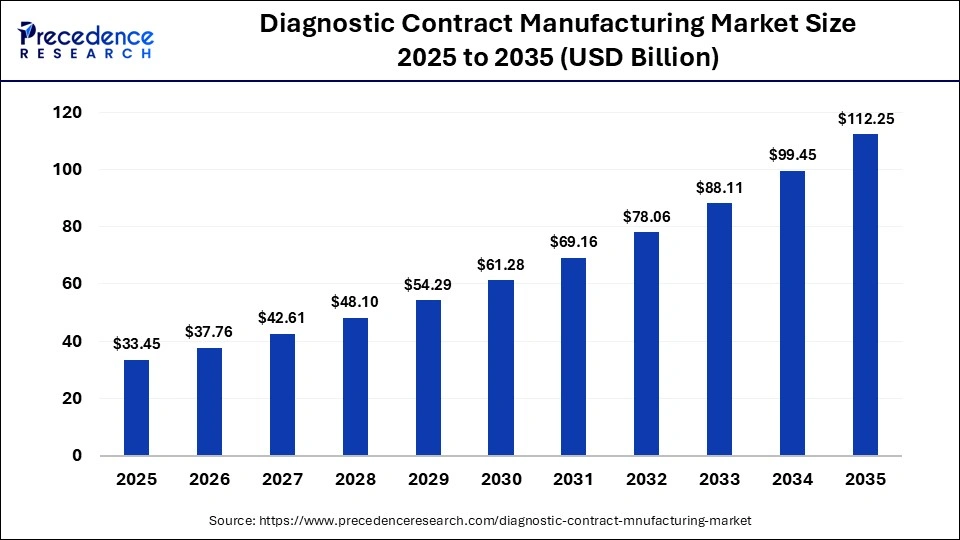
Key Takeaways
- North America held the largest revenue share of the diagnostic contract manufacturing market in 2025.
- Asia-Pacific is expected to experience the fastest growth during the predicted timeframe.
- By device type, the diagnostic imaging devices segment held a dominant revenue share of the market in 2025.
- By device type, the in vitro diagnostic (IVD) devices segment is expected to grow at the fastest CAGR in the market between 2026 and 2035.
- By application, the cardiology segment registered its dominance over the market in 2025.
- By application, the oncology segment is expected to be the fastest-growing in the market during the forecast period.
- By end user, the medical device companies segment dominated the market in 2025.
- By end user, the academic and research institutes segment is expected to grow with the highest CAGR in the market during the studied years.
What is Diagnostic Contract Manufacturing?
The diagnostic contract manufacturing market refers to the outsourcing of the production of diagnostic products like assays, reagents, and test kits. Contract manufacturing allows companies to concentrate on research and marketing while others handle production and quality. Factors affecting the growth of the market include high disease incidence, technological innovations, and increasing investments. Personalised medicines and rapid diagnostic technologies are also affecting the growth of the market. Companies are using modern technologies and collaborations to gain a foothold in the global market.
How is AI Transforming the Diagnostic Contract Manufacturing Market?
The role of artificial intelligence (AI) in reshaping the future of the market is improving production efficiency through optimized manufacturing processes and reducing human error and quality issues. AI is also helping manufacturers improve forecast accuracy and reduce waste and inventory issues. Machine learning (ML) is being applied to analyze large volumes of data from diagnostic tests for improving product design and performance.
AI is also helping improve manufacturing processes through robotics in terms of speed and reducing manufacturing costs. In terms of innovation and R&D, AI is helping accelerate the validation of new diagnostic products. It also ensures regulatory compliance through monitoring processes in real time. AI is able to improve productivity and competitive advantage in the market.
Diagnostic Contract Manufacturing Market Trends
- Automation and Digital Transformation
The increase in demand for automation, robotics, and advanced manufacturing technologies is changing the landscape of diagnostic contract manufacturing. Automated assembly lines, real-time monitoring systems, and AI-based quality management tools are improving product precision and lowering operational costs. Digital technologies are improving supply chain management and inventory management. These technologies are helping manufacturers scale production while maintaining high-quality standards, catering to the increasing global need for diagnostic equipment and supplies. - Increasing Outsourcing and Strategic Partnerships
Biotech and diagnostic companies are increasingly outsourcing manufacturing operations to contract manufacturers to minimize capital and operational costs. Strategic partnerships and public-private partnerships have also started to emerge to enhance manufacturing capabilities and innovations. Outsourcing operations have enabled companies to concentrate more on R&D and marketing while ensuring the production process with high quality output. The trend has been witnessed among small and mid-sized diagnostic companies. - Development of Advanced and Personalized Diagnostics
The increase in molecular diagnostics, genetic testing, and screening technologies is creating a significant impact on the demand for specialized manufacturing services. For instance, there is a need for highly accurate and customized production services for precision diagnostics. Contract manufacturers are investing more in advanced technologies for biomarker-based testing and new-generation diagnostic kits. In addition, there is a growing awareness of the need for early disease detection and targeted therapy. - Shift Towards Point-of-Care and Home-based Testing
There has been a growing emphasis in healthcare systems to adopt point-of-care and home-based testing solutions. Such solutions require compact and user-friendly solutions. Thus prompting contract manufacturers to adopt flexible production systems. The need for quick testing solutions for chronic and infectious diseases has also spurred the adoption of point-of-care and home-based solutions. - Sustainability and Regulatory Compliance Focus
Sustainability and international quality standards are emerging as major issues in diagnostic contract manufacturing services. Companies are adopting eco-friendly manufacturing processes and minimizing waste and energy consumption. Adherence to international quality standards is necessary to ensure the safety and quality of the products manufactured. International authorities are enforcing strict standards, and this is prompting contract manufacturers to adopt sophisticated quality management systems to sustain their business in the long term.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 33.45 Billion |
| Market Size in 2026 | USD 37.76 Billion |
| Market Size by 2035 | USD 112.25 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 12.87% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Device Type, Application, End-User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segmental Insights
Device Type Insights
Which Device Type Segment Dominated the Diagnostic Contract Manufacturing Market?
The diagnostic imaging devices segment contributed the biggest market share in 2025, because imaging technologies are essential for the diagnosis, management, and treatment of diseases. Contract manufacturers assist in the production of components, assembly, and integration of these technologies. Technological advancements lead to the development of novel imaging techniques, enabling radiologists to detect minor changes in patients. Diagnostic laboratories and hospitals are at the forefront of installing advanced imaging devices, potentiating the need for contract manufacturing.
The in vitro diagnostic (IVD) devices segment is expected to show the fastest growth over the forecast period. This is due to the increasing demand for disease detection at an early stage, personalized medicine, and molecular testing, leading to an increase in outsourcing for these devices. Additionally, the increasing prevalence of infectious and chronic diseases is also contributing to the segment's growth. The segmental growth is also attributed to the increasing lab automation and the demand for high testing platforms.
Application Insights
Why Did the Cardiology Segment Dominate the Diagnostic Contract Manufacturing Market?
The cardiology segment dominated the market in 2025, due to the high global burden of cardiovascular diseases, which remains one of the leading causes of mortality worldwide. The growing incidents such as heart-related conditions due to various factors like obesity, diabetes, high blood pressure, and sedentary lifestyles has led to an increased need for diagnostic solutions.
Hospitals and diagnostic centres have been using various imaging solutions, cardiac biomarker-based diagnostic solutions, and monitoring solutions for the detection of various heart-related conditions. Continuous innovations in cardiac testing solutions and the growing awareness about preventive healthcare boost the segment's growth.
The oncology segment is expected to witness the fastest growth in the market over the forecast period. The increasing prevalence of cancer, growing awareness of early diagnosis, and rising healthcare expenditure drive the segment's growth. The growing research activities in oncology lead to the development of innovative oncology testing devices. Early cancer detection enables healthcare professionals to provide timely intervention, providing personalized care.
End User Insights
Which End User Segment Led the Diagnostic Contract Manufacturing Market?
The medical device companies segment led the market in 2025, due to technological innovations and increasing partnerships among medical device companies and contract manufacturing organizations (CMOs). The rising number of large companies and startups increases competition among companies, necessitating them to expand their product pipelines and strengthen their market positions. By outsourcing manufacturing, companies can focus on their core competencies, such as product sales and marketing.
The academic and research institutes segment is expected to expand rapidly in the market in the coming years. The growing research activities and the lack of suitable manufacturing infrastructure necessitate academic institutes to outsource diagnostic manufacturing. Academic institutes frequently outsource the manufacturing of diagnostic devices, especially while transitioning from research to clinical trials and commercialization. They receive funding from government and private bodies to conduct advanced research and clinical trials.
Regional Insights
Why North America Dominated the Diagnostic Contract Manufacturing Market?
North America held a major revenue share of the market in 2025. The region has a high standard of healthcare infrastructure, increasing healthcare expenditure, and the presence of major diagnostic and biotechnology companies. The rising prevalence of chronic diseases and the high adoption of molecular diagnostic technologies at an early stage propel the market. The region has high standards of regulatory requirements. Companies prefer to collaborate with experienced contract manufacturers to meet regulatory requirements. North America has high research funding, an availability of a skilled workforce, and high product innovation.
U.S. Market Trends
The U.S. leads the market in North America due to the increase in the demand for molecular and point-of-care diagnostics, outsourcing, adoption of AI and automation, and high regulatory focus. The presence of key players and the growing demand for personalized medicines support diagnostic contract manufacturing. Key players, such as Drucker Diagnostics, EKF Diagnostics, and Thermo Fisher Scientific, are major contributors to market growth.
Why is Asia-Pacific Expanding Rapidly in the Diagnostic Contract Manufacturing Market?
Asia-Pacific is expected to grow at the fastest CAGR in the market during the forecast period. This can be attributed to the high rate of development in healthcare infrastructure, healthcare expenditure, and disease incidence. For instance, there has been an expansion in diagnostic infrastructure in countries like China, India, and South Korea. The region provides cost advantages for manufacturers, labor resources, and regulatory environments that make it conducive for diagnostic companies to outsource their production to this region.
China Market Trends
China's market is expanding rapidly, with multinational corporations having a presence, and domestic players are growing rapidly. Key players Jabil Inc, Flec Ltd, Plexus Corp, Sanmina Corporation, Sanner Group, and Local Specialised CMOs, these are specializing in medical device, diagnostics, and imaging components. The Chinese government also provides funding for manufacturing and developing diagnostic devices. It also encourages the indigenous development of medical devices through its “Made in China” policy.
Will Europe Grow in the Diagnostic Contract Manufacturing Market?
Europe is expected to grow at a notable CAGR in the foreseeable future. The rising prevalence of chronic disorders, growing research activities, and the rising adoption of advanced technologies foster market growth. The European Medicines Agency (EMA) regulates the approval of novel diagnostic devices in the region. People are becoming aware of personalized medicines and point-of-care diagnostics, facilitating the development of novel diagnostics. The increasing collaboration, public-private partnerships, and investments also contribute to market growth.
Top Companies in the Diagnostic Contract Manufacturing Market
- Merck KGaA
- Thermo Fisher Scientific Inc.
- TCS Biosciences
- Jabil Inc.
- FLEX LTD.
- Plexus Corp.
- Savyon Diagnostics
- KMC Systems
- Nova Biomedical
- Sanmina Corporation
Recent Developments in the Diagnostic Contract Manufacturing Market
- In November 2025, Abingdon Health plc announced that it signed a Master Service Agreement (MSA) with a U.S.-based company for the development and scale-up to manufacture a semi-quantitative, multiplex lateral flow test system. The $2 million contract covered the feasibility, optimization, scale-up, and technology transfer for manufacturing. (Source: https://www.abingdonhealth.com)
- In October 2025, Bluejay Diagnostics, Inc. and SanyoSeiko Co., Ltd. collaborated to manufacture and distribute Bluejay's Symphony platform, a near-patient testing system designed to provide rapid and reliable results for key biomarkers, such as IL-6 in sepsis management.
- In January 2025, Akoya Biosciences, Inc. collaborated with Argonaut Manufacturing Services to manufacture its IVD assays, underscoring both companies' commitment to advancing precision medicine through high-quality diagnostics. The collaboration was made to leverage Argonaut's deep expertise and reliable systems. (Source: https://www.contractpharma.com)
Segments Covered in the Report
By Device Type
- In Vitro Diagnostic (IVD) Devices
- Equipment
- Consumables
- Diagnostic Imaging Devices
- CT Scanners
- MRI Systems
- X-Ray Systems
- Ultrasound Devices
By Application
- Cardiology
- Oncology
- Infectious Diseases
- Orthopedics
By End User
- Medical Device Companies
- Diagnostic Laboratories
- Hospitals
- Ambulatory Surgical Centres
- Academic & Research Institutes
By Region
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
