What is the Pharmaceutical Stability And Storage Services Market Size?
The global pharmaceutical stability and storage services market size is calculated at USD 3.56 billion in 2025 and is predicted to increase from USD 3.75 billion in 2026 to approximately USD 6.1 billion by 2035, expanding at a CAGR of 5.53% from 2026 to 2035.
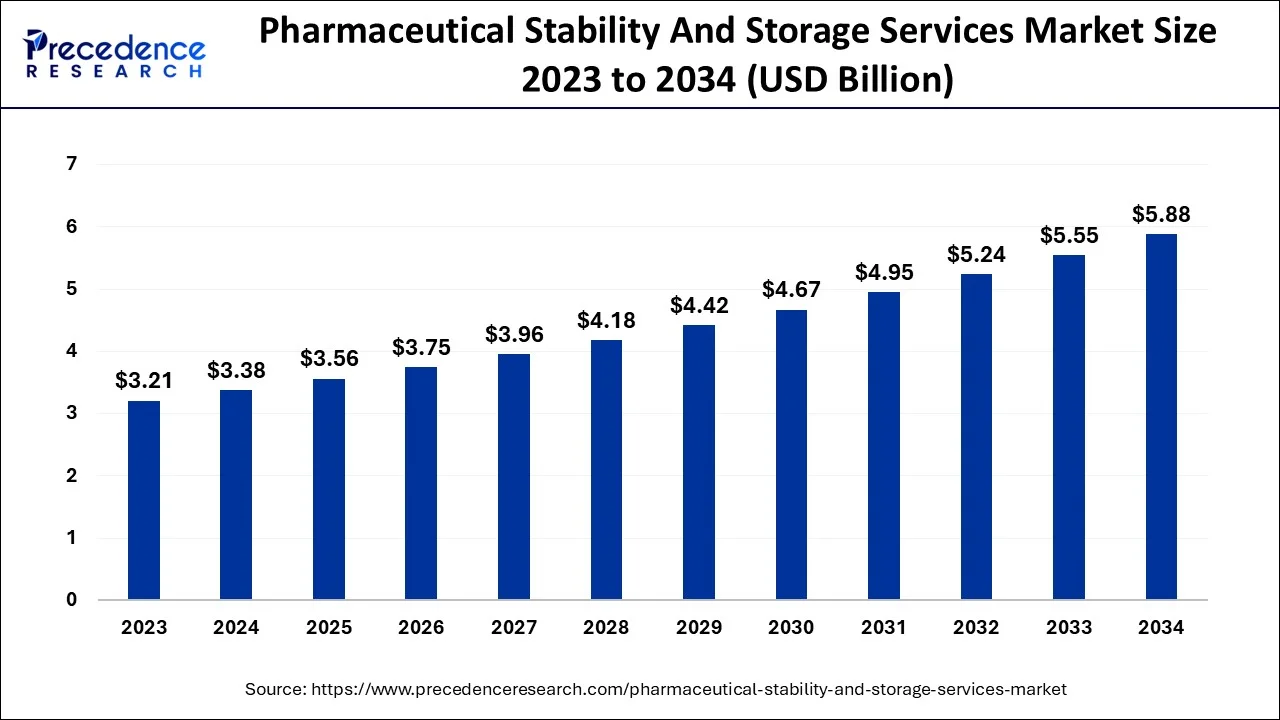
Pharmaceutical Stability And Storage Services Market Key Takeways
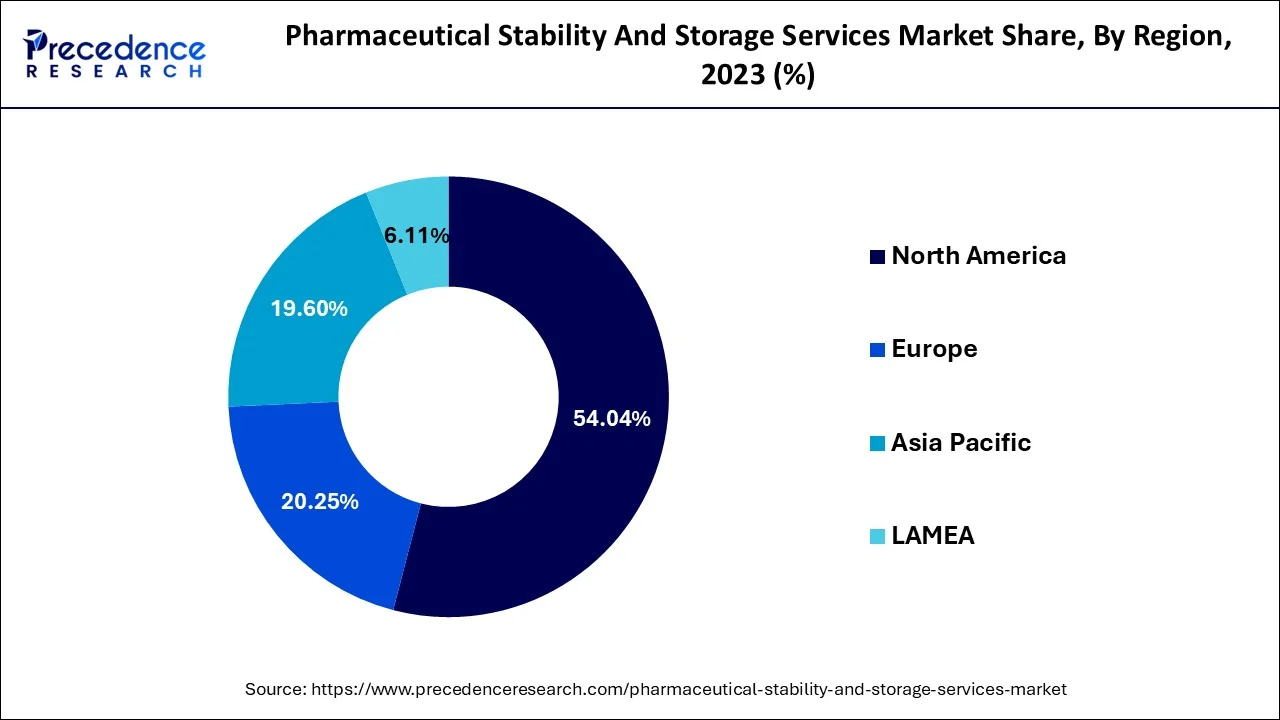
- North America generated more than 54.18% of the revenue share in 2025.
- Europe is expected to expand at the biggest CAGR of around 6.10% between 2026 and 2035.
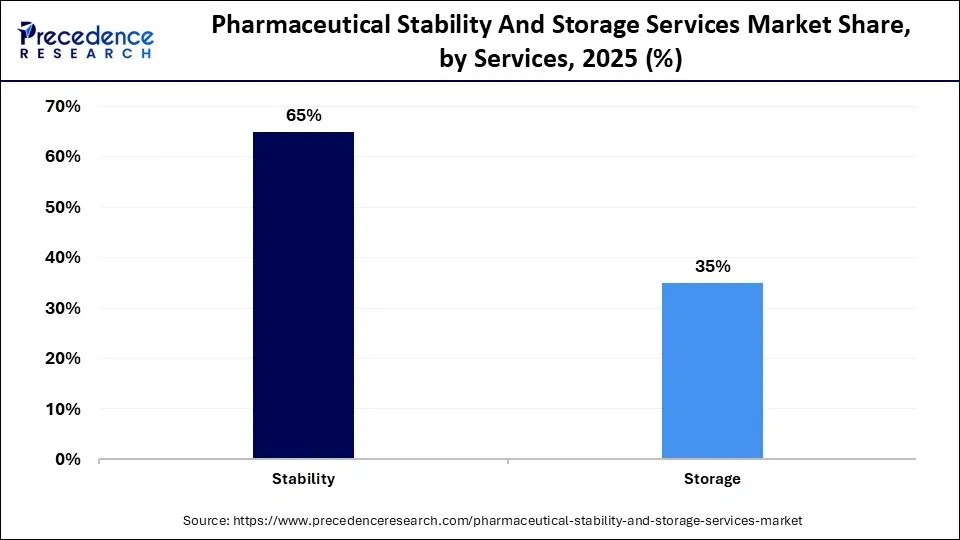
- By Services, the stability segment captured more than 65% of the revenue share in 2025.
- By Services, The storage services segment is projected to grow at the fastest CAGR of 5.4% between 2026 and 2035.
- By Molecule, The small molecule segment generated the highest revenue share of around 55% in 2025.
- By Molecule, The large molecule segment is predicted to expand at the fastest CAGR between 2026 and 2035.
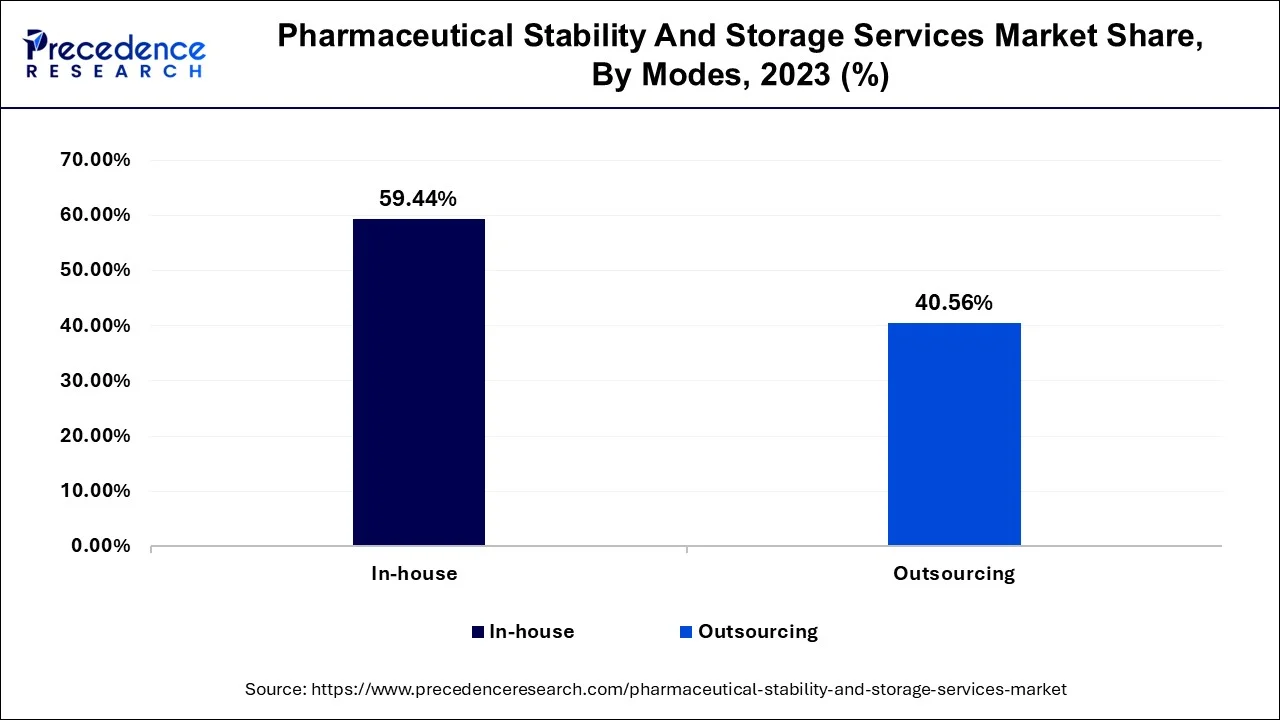
- By Mode, The in-house mode segment contributed the highest share of around 60% in 2025.
- By Mode, The outsourcing mode segment is expected to grow at the fastest rate between 2026 and 2035.
Market Overview
Due to rising R&D spending by pharma firms, the pharmaceutical stability and storage services industry is growing significantly. Pharmaceutical firms are increasingly focusing their R&D efforts in order to remain competitive as well as versatile. An increase in novel approval applications, centralization, and globalization of both large and small molecule industries are driving the market.
Furthermore, with the sustained expansion of biological drugs, the worldwide pharmaceutical stability and storage industry is expected to expand rapidly. Medicinal companies are moving towards decentralized stability testing and centralized management and reporting, whereas pharmaceutical companies are combining stability storage and restricting testing to one service.
AI-Driven Reinvention of Pharma Stability & Storage Services
The pharmaceutical stability and storage services market is transforming due to the use of Artificial Intelligence (AI) to increase efficiencies in operations and improve accuracy, compliance, and costs. The use of AI technology has enabled the automation of stability data analysis, more accurately predicting the manufacturer's estimated shelf life, and eliminates much of the manual processes associated with stability reporting thus improving regulatory readiness by speeding up the delivery of the stability report. The combination of machine learning and predictive analytics will lead to improvements in cold chain management and environmental risk mitigation for the storage of temperature sensitive biologics and other products.
he advantages of these software applications enhance the strengths of the quality assurance of the products, as well as enable the companies to make better decisions on supply chain management. The momentum for the pharmaceutical industry to adopt AI solutions is strong as investment continues to accelerate and the transformation of pharmaceutical companies continues and their productivity increases. Recent advancements relating to AI-based platforms specifically focused on the prediction of stability and regulatory analysis are an indication that the pharmaceutical industry is increasingly using better data analysis tools in an effort to increase the value of their service and differentiate themselves from their competitors.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 3.56 Billion |
| Market Size in 2026 | USD 3.75 Billion |
| Market Size by 2035 | USD 6.1 Billion |
| Growth Rate from 2026 to 2035 | CAGR of 5.53% |
| Largest Market | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Services, Molecule, Mode and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America and Middle East & Africa |
Market Dynamics
Drivers
Vaccines are in high demand
Demand for COVID-19 vaccines that are stable and able to be stored for an extended period of time is expected to rise as government agencies rapidly increase their vaccination efforts. In recent years, the total expenditure on R&D in the pharmaceutical sector has grown significantly. The increased spending on R&D is expected to increase the number of drugs.
Another factor influencing market growth is the requirement for stability testing before receiving approval for clinical trial phases. Furthermore, biosimilar drugs are identical copies of biological drugs, so their costs are much lower when compared to biological drugs.
Rising biosimilar demand
Biosimilars have grown in popularity in recent years owing to their close similarity to biological drugs as well as their typically lower cost. Biosimilars have gained popularity in the treatment of cancer, autoimmune diseases, and other chronic conditions. Due to the global prevalence of all these diseases, the requirement for biosimilars is anticipated to rise, as well as the need for their stability and storage.
Marketing products across markets is complicated because different regulatory authorities have other data requirements and testing guidelines for determining the stability of a product. Hence, increasing demand for a biosimilar is expected to increase the demand for outsourcing services in the market.
Restraints
Rare highly skilled employees
Pharmaceutical stability and storage service providers face challenges in recruiting and maintaining highly skilled personnel. Pharmaceutical, contract research, medical device, biotechnology, and academic and research institutions compete for qualified and experienced scientists.
The pharmaceutical industry's shift towards innovative molecular structures as well as mechanisms of action makes a significant contribution to a scarcity of skilled professionals in this field. Pharmaceutical companies, CROs, and healthcare analytical testing industries must collaborate to develop training programs tailored to the healthcare sector.
Opportunities
The recent trend in multifunctional pharmaceutical businesses is to define conditions for global marketing stability testing. Companies are developing protocols to cover a single set of extreme environmental conditions for this purpose. The specific changes for global testing include extending the accelerated testing period from 6 to 12 months and conducting additional tests at 500 degrees Celsius/75% relative humidity for three months.
It avoids the repetition of stability testing for the region and makes the most efficient use of resources because all tests are performed in one laboratory. Testing under a combination of three environmental factors, namely temperature, humidity, and light, has been shown to have a more severe impact on drug compounds and products than humidity alone.
Impact of COVID-19:
The COVID-19 pandemic has raised the global demand for COVID-19 vaccines. The government's increasing vaccine push is likely to increase the need for commercial COVID-19 vaccine stability and storage. Pharmaceutical R&D expenditure has increased significantly in recent years. Rising R&D expenditure is predicted to boost the number of drugs undergoing clinical trials. Each stage of a clinical study requires stability testing before it can be approved, and this is accelerating market growth.
Furthermore, biosimilar drugs are remarkably similar copies of biologics and are significantly less expensive than biologics. Several diseases have increased since COVID-19. According to a report published by Children's National Hospital- pediatric research and clinical innovations center, a study was conducted on 737 diabetes-diagnosed youths, and it discovered an increased incidence of pediatric Type 1 Diabetes by 15.2% and Type 2 Diabetes by 182% between March 2018, and March 2021. The increase in incidence rate is anticipated to boost drug production, which will drive market demand.
Service Insights
The stability segment generated more than 63.02% of the revenue share in 2023. Stability testing is an essential parameter that pharmaceutical industries must analyze and report on in order to obtain regulatory approval for marketing. The necessity of stability testing further increases its market demand during each stage of the clinical trials.
During the projected period, the storage sector is anticipated to grow at the fastest rate. Drugs are stored in cold & non-cold conditions in controlled environments compartments to see if the performance of the drug alters over time under humidity, temperature, and light conditions. A large number of small-molecule drugs must be placed in non-cold conditions, and the abundance of commercially accessible small molecules is propelling market demand for non-cold storage.
Global Pharmaceutical Stability and Storage Services Market Revenue, By Services, 2023-2025 (USD Million)
| Services | 2023 | 2024 | 2025 |
| Stability | 2,080.1 | 2,191.0 | 2,310.0 |
| Storage | 1,131.3 | 1,187.3 | 1,247.4 |
Molecules Insights
The small molecule sector had the highest revenue share of over 55.78% in 2023. Drug development for small molecules has advanced significantly over the past three decades. Small molecules account for roughly 90% of all pharmaceutical medications. It is used to treat fevers, migraines, cancer, diabetes, as well as other common illnesses. The increasing application of small-molecule drugs to treat common disorders and diseases is raising the need for stable testing & storage.
Over the forecast period, the large molecule sector is expected to expand at the fastest pace. Large molecules are commonly used to treat cancer, infectious diseases, and autoimmune diseases. The high prevalence of these illnesses is expected to fuel market growth for large molecules and, as a result, demand for large molecule stability and storage.
Global Pharmaceutical Stability and Storage Services Market Revenue, By Molecules, 2023-2025 (USD Million)
| Molecules | 2023 | 2024 | 2025 |
| Small Molecule | 1,763.9 | 1,857.7 | 1,958.3 |
| Large Molecule | 1,447.4 | 1,520.6 | 1,599.1 |
Mode Insights
The in-house segment had the highest share of around 59.44% in 2023. A large percentage of pharmaceutical manufacturers prefer in-house stability testing because these tests have been necessary for all drugs and at all stages of clinical trials. One of the factors that pharmaceutical industries take into cogitation when considering in-house services for testing is the cost-effectiveness of owning a stability chamber for carrying out stability studies. An in-house unit conducting stability testing is more efficient for pharmaceutical firms that are able to afford the initial development cost; these attributes influence market demand for in-house services.
During the projected period, the outsourcing sector is anticipated to grow the fastest. The market's demand for outsourcing is driven by potential complications with stability testing. Various regulatory agencies have different data demands and testing guidelines, which makes marketing products difficult, particularly in different supply chains. Even though the EMA, as well as FDA, follow ICH regulations for stability testing, their microbiological criteria for stability tests differ. Such complicated stability testing increases the need for outsourcing services.
Global Pharmaceutical Stability and Storage Services Market Revenue, By Modes, 2023-2025 (USD Million)
| Modes | 2023 | 2024 | 2025 |
| In-house | 1,911.9 | 2,013.8 | 2,123.2 |
| Outsourcing | 1,299.4 | 1,364.5 | 1,434.1 |
U.S. Pharmaceutical Stability And Storage Services Market Size and Forecast 2026 to 2035
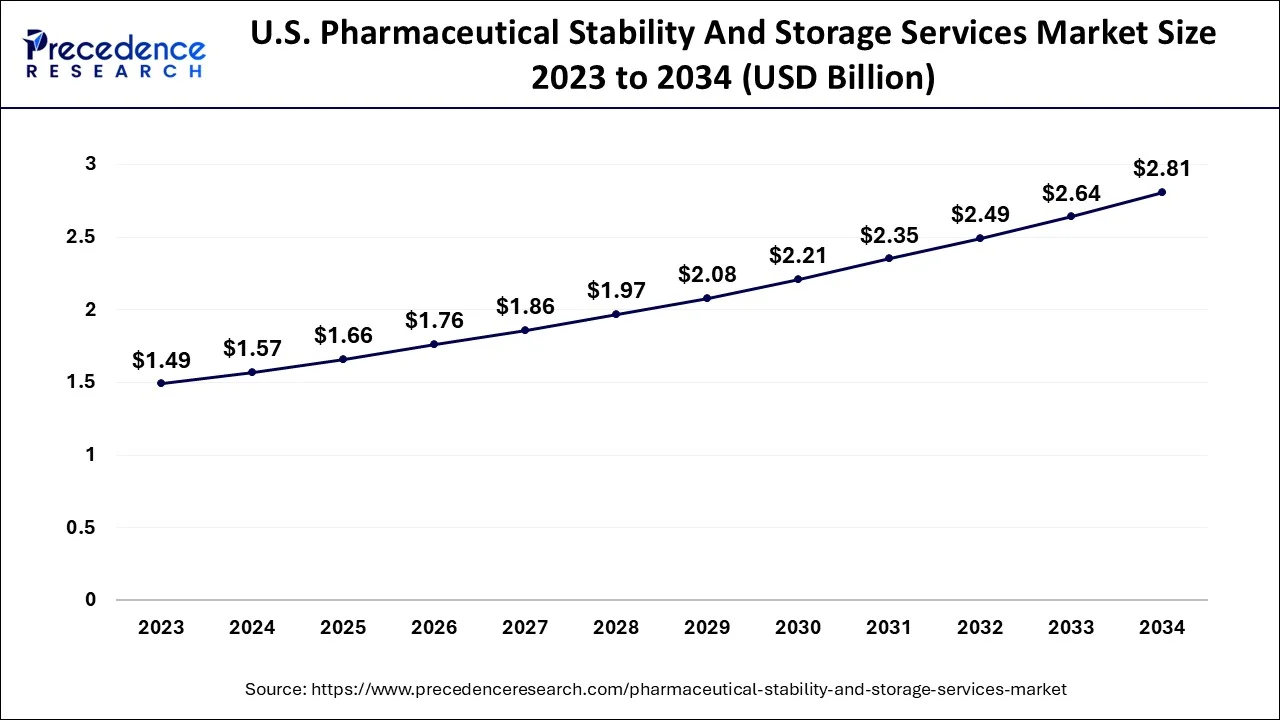
The U.S. pharmaceutical stability and storage services market size is estimated at USD 1.66 billion in 2025 and is expected to be worth around USD 2.92 billion by 2035, expanding at a CAGR of 5.81% between 2026 and 2035.
North America had the highest revenue share of over 54.18% of the pharmaceutical stability & storage market. This is due to the large number of pharmaceutical businesses located in the United States and Canada. The presence of key market players in the region offering stability & storage services is anticipated to considerably contribute to market expansion.
United States (North America):
The U.S. is the global leader in pharmaceutical stability and storage services because of the regulatory certainty and its infrastructure. The FDA still mandates that pharmaceutical companies follow stability testing protocols relating to ICH guidelines, which means those that do not cannot sell a legitimate pharmaceutical product. In 2024, U.S.-registered firms accounted for over 60% of global pharmaceutical R&D investments. There are more than 800 GMP-certified pharmaceutical storage facilities in the U.S. alone. Companies are increasingly focused on temperature-controlled logistics and storage facilities, which are particularly relevant for biological and mRNA-based pharmaceuticals.
Asia Pacific is predicted to expand at the fastest CAGR of around 7% between 2025 and 2035. Due to the existence of countries such as China, India, and Japan, the region has the fastest-growing pharmaceutical market. Initiatives by public organizations to reduce prices for drugs in order to improve access to high-quality products have aided market growth. Clinical research is also a priority in the region. According to Global data, China will account for 26% of worldwide clinical trials operation in 2021, which will benefit the Asia Pacific market.
India (APAC):
India's strength in the APAC region for pharmaceutical stability and storage services is punctuated by its development of a robust generic drug industry and the accomplishment of regulatory compliance with WHO and ICH standards. In 2024, India had more than 500 WHO-GMP-certified pharmaceutical manufacturing units. The Indian government's “Pharma Vision 2020” initiative bolstered the cold chain logistics and storage opportunities for products and aided its ability to export medicines and drugs to more than 200 countries. In recent news, India is also paying significant attention to the stability testing of biosimilars to help respond to global demand.
How Is Europe notably growing in pharmaceutical stability and storage services market?
Europe is a uniquely growing area of the pharmaceutical stability and storage services market due to strong regulatory requirements and a mature pharmaceutical industry. Quality assurance, environmental control, and validated stability protocols support the continued need for specialized geographical locations to meet consumer needs. As more biologics, advanced therapies, and biosimilars continue to develop, the need for controlled temperature storage facilities will increase. There is also a growing number of sustainability initiatives that are impacting facility improvements such as creating energy-efficient stability chambers. Additionally, as the numbers of cross-border drug distributions continue to expand, it will be necessary for regulatory agencies to harmonize requirements for storing these products between countries and thus will lead to increased reliance on third-party service providers to meet compliance, data integrity, and longer-term product stability.
Germany
Germany will remain a key player in the Pharmaceutical Industry in Europe due to its long-term and well established pharmaceutical manufacturing infrastructure and R&D base. The focus on producing high-quality pharmaceuticals under strict regulatory compliance has created a significant demand for certified stability testing services. The growths of biotechnology start-ups and new developments in advanced therapy research have increased the need for accurate environmental monitoring and the availability of long-term storage facilities. The location of Germany in the center of Europe also provides an advantage by allowing for regional distribution networks which will only enhance the leadership role of Germany in the demand for stability and storage services of pharmaceuticals.
How Latin America Emerging in Pharmaceutical Stability And Storage Services Market?
Latin America has emerged as an area of growth for the pharmaceutical stability and storage services market with the gradual increase in pharmaceutical manufacturing capacity. Governments throughout the region have improved their regulatory frameworks and become better aligned with international standards, which will lead to increased demand for compliant stability-testing services. Participation in global clinical trials and a growth in generic drug production have fostered a need for structured storage infrastructure. Although there are continuing infrastructure gaps in various areas, investments in cold-chain logistics and temperature-controlled warehouses are improving service reliability. Multinational pharmaceutical companies are expanding their operations throughout the region, and as this happens, demand for outsourced stability studies and validated storage solutions will steadily increase.
Brazil
Brazil is the regional leader because of its more developed pharmaceutical manufacturing sector and relatively robust regulatory development. Domestic drug production is increasing in Brazil in addition to Brazilian participation in clinical research activities, which create a continued demand for stability-testing services. Brazil's improvement of cold-chain infrastructure to be compliant with international distribution standards has increased storage capabilities, and the size of Brazil's healthcare market and focus on biosimilars are both major contributors to the regional growth of the service sector.
How Is Middle East & Africa Growing?
The Middle East & Africa region is notably growing in the Pharmaceutical Stability and Storage Services Market, supported by healthcare infrastructure modernization and rising pharmaceutical imports. Governments are investing in regulatory harmonization and quality control standards, increasing the need for validated stability testing and temperature-controlled storage. Expansion of vaccine programs and specialty drug distribution has accelerated demand for cold-chain solutions. Although the market remains developing, strategic partnerships with global pharmaceutical firms are enhancing service capabilities. Growing awareness of product integrity and supply chain reliability is gradually strengthening the role of outsourced pharmaceutical storage services across the region.
Saudi Arabia
Saudi Arabia leads regional growth due to significant healthcare investments and efforts to localize pharmaceutical manufacturing. Regulatory reforms and quality assurance initiatives have increased demand for compliant stability testing facilities. The country's expanding hospital infrastructure and vaccine distribution programs require advanced temperature-controlled storage systems. Strategic initiatives to attract pharmaceutical production and research collaborations are further stimulating the development of specialized storage and stability service capabilities.
Pharmaceutical Stability and Storage Services Market Companies
- Catalent, Inc.
- Charles River Laboratories International, Inc
- Almac Group
- Eurofins Scientific
- Lucideon Limited
- Intertek Group Plc
- Alcami Corporation
- Element Materials Technology
- Q1 Scientific
- BioLife Solutions
- Masy BioServices
- Roylance Stability Storage Limited
- Reading Scientific Services Ltd.
- Als Ltd.
- Auriga Research Private Limited
- Q Laboratories
- Precision Stability Storage
- PD Partners
Recent Developments
- In January 2024, Alcami Corporation ("Alcami"), a leading contract development and manufacturing organization (CDMO), announced the opening of its new 65,000 ft² state-of-the-art Pharma Storage and Services operations facility in Garner, North Carolina, close to the Research Triangle Park biotech and pharma hub.
- In July 2024, Catalent completed a USD 25 million expansion of its clinical supply facility in Germany, adding 32,000 sq. ft. to increase temperature-controlled storage capacity and introduce a new automatic bottle filling line.
- In July 2024, Cambrex announced the expansion of its stability storage business, Q1 Scientific, which offers environmentally-controlled stability storage services to the pharmaceutical, medical device, and life sciences industries.
- In December 2022, Almac Group completed the first stage of its GMP API facility expansion.
- In 2022, Alcami established a CGMP Bio storage Laboratory in North Carolina.
- In June 2022, Cambrex acquired Q1 Scientific, an Irish storage company.
- Charles River Laboratories International, Inc. announced the release of its manufacturing capacity in Bruntwood SciTech's Alderley Park in February 2022.
- BioLife Solutions acquired a majority stake in Sexton Biotechnologies in September 2021.
Market Segmentation
By Services
- Stability
- Drug Substance
- Stability indicating method validation.
- Accelerated stability testing.
- Photostability Testing
- Other stability testing methods
- Storage
- Cold
- Non-cold
By Molecule
- Small Molecule
- Research Products
- Commercial Products
- Large Molecule
- Research Products
- Commercial Products
By Mode
- In-house
- Outsourcing
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- The Middle East and Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
