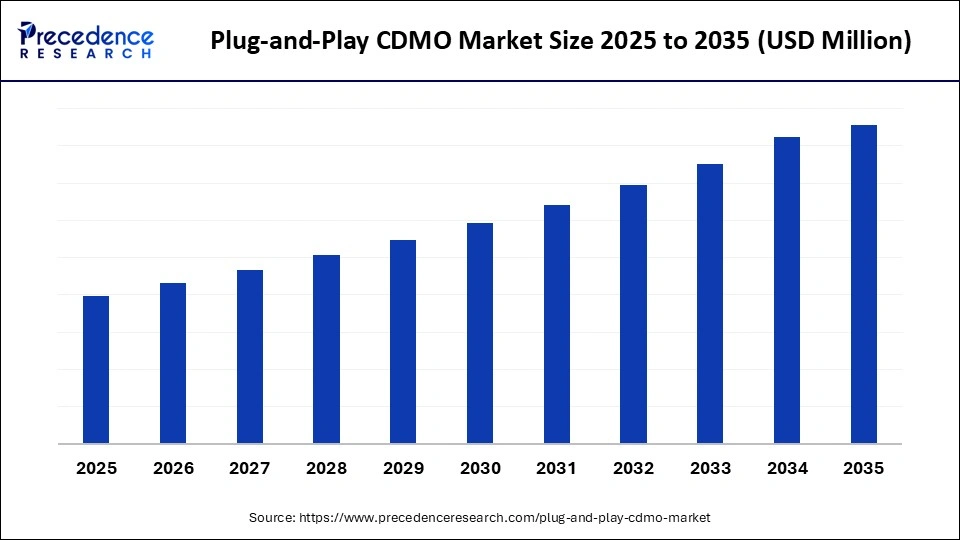
What is the Plug-and-Play CDMO Market Size?
The global plug-and-play CDMO market serves pharmaceutical and biotechnology companies seeking cost-efficient and compliant manufacturing models. The market is driven by several qualified scientists, economic production, and the well-established generic and biosimilar sectors.
Market Highlights
- North America is dominating the market, holding the largest share in 2025.
- Asia Pacific is projected to grow at a significant CAGR from 2026 to 2035.
- By service type, the end-to-end plug-and-play services segment led the market while holding the largest share in 2025.
- By service type, the advanced therapies-focused DS + DP services segment is expected to grow at a CAGR between 2026 and 2035.
- By molecule type, the biologics segment contributed the largest market share in 2025.
- By molecule type, the advanced therapies segment is projected to grow at a significant CAGR from 2026 to 2035.
- By development stage, the clinical manufacturing segment led the market while holding the largest share in 2025.
- By development stage, the commercial-scale manufacturing segment is expected to grow at a CAGR between 2026 and 2035.
- By manufacturing platform, the single-use segment led the market while holding the largest share in 2025.
- By manufacturing platform, the continuous manufacturing platforms segment is expected to grow at a CAGR between 2026 and 2035.
What is the Plug-and-Play CDMO Market?
The plug-and-play CDMO market continues to gain traction in the biopharma industry. The players look increasingly to flexible, ready-to-go manufacturing offerings. By providing standardized, modular, and quickly scalable production platforms, these models avoid time-consuming setup cycles. As the pressure mounts to move drugs faster, companies are looking at PNP partners that can shorten timelines and reduce operational burden. The marketplace is built on creativity that marries pace with regulatory adherence. Those innovation pioneers can focus on the science and outsource pounding nuts and bolts. Rising biologics and personalized therapies are further pushing the need for adaptable, turnkey manufacturing systems. Overall, the market is evolving into a powerful enabler of agility, efficiency, and accelerated commercialization in the life sciences industry.
Key Technological Shifts in the Plug-and-Play CDMO Market
The Plug-and-Play CDMO Market is experiencing a technological shift with a focus on increased agility and scalability for manufacturers as they take advantage of these developments to create more technologically advanced factories. The most notable trend within the Plug and Play CDMO market is that manufacturers are beginning to move away from traditional methods of building and designing clean-room environments that are modular or flexible to allow for fast deployment, expansion, and reconfiguration without long periods of downtime due to construction phases. In addition, advances in manufacturing technologies, including single-use bioprocessing systems, have fundamentally changed the way that biopharmaceutical products are produced by removing the need for manual cleaning and disinfection of traditional equipment, allowing for a much simpler process.
Additionally, manufacturers of Plug and plug-and-play CDMO are utilizing Digital Twins and Automated Process Control to create simulated environments where they can monitor and optimize their manufacturing lines and processes in real time. As an important advancement in technology, closed and continuous manufacturing systems are becoming more widely used, allowing for increased safety, reduced time to market, and reduced facility footprint. As a result of these advancements in technology, the definition of a true Plug and Play CDMO ecosystem is evolving and companies are beginning to build smarter, modular, and interconnected manufacturing ecosystems.
Key Market Trends in Plug-and-Play CDMO Market
- Rapid Onboarding: CDMOs promise near-instant facility readiness for clients racing against clinical deadlines.
- Multi-modal capability: Where plug-and-play suites can handle biologics, mRNA, cell therapies, or viral vectors with minimal retooling.
- Sustainability: The CDMOs are adopting energy-efficient HVAC systems, recyclable single use technologies, and carbon light modular units.
- Virtual biotech companies: as CDMO market is taking setup boost in the virtual avtars and AI industry.
Market Scope
| Report Coverage | Details |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Service Type, Molecule Type, Development Stage, Manufacturing Platform, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Service Type Insights
Why Is End-to-End Plug-and-Play Services Foremost in the Plug-and-Play CDMO Market?
End-to-end plug-and-play services are dominating the Plug-and-Play CDMO market due to biopharma companies wanting an easy way to get from early work to making stuff under GMP rules. They do not want to mess with lots of different companies. These services handle everything, designing the process, getting the place ready, setting up the gear, and doing what the rules say, all in one package. They really cut down on how long things take, so companies trying to get to clinical trials love them. It is like getting everything in one spot, which makes things way easier, especially for small biotech teams. Since getting things done fast is super important now, end-to-end services are the go-to choice. They are simple, can handle more work, and are reliable, so they are a key part of the plug-and-play CDMO idea.
Advanced Therapies-focused DS + DP services is set to be the fastest-growing segment in the plug-and-play CDMO market, due to gene therapies needing operations that are super specialized and flexible. These services mix high-tech stuff for both upstream and downstream tasks with fill-finish lines that are set up for delicate treatments. Companies making treatments that use a person's own cells or viruses rely on these setups to keep things from slowing down or messing up the manufacturing. They need things to be precise, sterile, and able to switch over fast, so they are using these DS+DP systems a lot. Because new therapies are popping up all over, the need for these plug-and-play services is going up way fast. This area is becoming the spot for new things in the CDMO world.
Molecule Type Insights
Why is Biologics Dominating the Plug-and-Play CDMO Market?
Biologics are dominating the Plug-and-Play CDMO market, because modular cleanrooms and bioreactors that you only use once are great for complex protein treatments. Companies are working on antibodies, enzymes, and proteins made with fancy tech, like being able to increase production fast without having to totally redo their facilities. Plug-and-play systems lower the risk of contamination, which is super important when making biologics. They also make it easier to switch between different batches, which is something that biologics projects often need. Since biologics are still a big part of what is happening, this area is the most popular. Modular manufacturing and the complex nature of biologics go hand in hand, so this category is staying in the lead.
Advanced Therapies is the fastest growing in the Plug-and-Play CDMO market, because the people creating them want quick ways to make stuff that can change easily to fit their cell and gene therapy plans. These therapies need small, controlled spaces where they can switch between making different things without wasting much time. Plug-and-play modules do just that, so they're perfect for making vectors, processing cells, or doing mRNA work. The market is moving toward spaces that are super flexible and closed off to keep things from getting contaminated. As more and more of these therapies are being tested, the people creating them are using pre-made setups that are good for ATMPs to avoid running out of space. This category is moving faster than any other type of molecule.
Development Stage Insights
Why is Clinical Manufacturing Dominating the Plug-and-Play CDMO Market?
Clinical Manufacturing is dominating the Plug-and-Play CDMO market because companies in early stages of development really need to move fast, follow the rules, and not spend too much money. Plug-and-play setups let them start making stuff under GMP without waiting for facilities to be built or equipment to arrive. These ready-to-go cleanrooms lower risk and make trials faster. Clinical teams want to be able to run a bunch of small batches while they improve their processes. This stage is all about being quick and flexible, so plug-and-play solutions are a great fit. So, clinical manufacturing is still what's driving demand right now.
Commercial-Scale Manufacturing is the fastest growing in the Plug-and-Play CDMO market, which is work that moves past the trial stage and must be produced at scale in a repeatable manner. Companies are beginning to go with modular units to grow the amount that can be made without needing tons of big, permanent stuff. Plug-and-play setups let commercial teams add more units as they are needed, which keeps them from spending on something bigger. The push to get to market faster and keep things the same all over the world is part of what is making this happen. As more biologics and ATMPs get approved, plug-and-play for commercial manufacturing is getting more traction. The ability to increase production volume quickly to make good stuff under GMP rules is transforming how production is done after approval.
Manufacturing Platform Insights
Why Is Single-Use Dominating the Plug-and-Play CDMO Market?
Single-use are dominating the Plug-and-Play CDMO market, because the systems take away the complexity of cleaning, reduce downtime, and allow for safer transitions between products. For plug-and-play CDMOs, single-use equipment is what supports quick setup and flexibility. It cuts contamination risk and validation issues, two big worries when working with multiple clients. Companies can get projects started faster, run different batches, and switch between therapies without any headaches. The savings on costs that involve operations and the environment make single-use systems the standard. To be brief, single-use equipment is a key part of plug-and-play manufacturing.
Continuous Manufacturing Platforms is the fastest growing in the plug-and-play CDMO market, seeking the perfect situation that consists of automation, flexibility, and savings. The systems combine partial automation and modular cleanrooms, allowing for great operations without having companies give up the property of adaptation. The systems support biologics and advanced therapies that involve little reconfiguration needs. The attraction resides in the ability of the systems to scale up operations and keep it reasonable. CDMOs are transitioning towards more systems that are connected and smart: semi-integrated solutions are rising rapidly.
Regional Insights
Why is North America Leading the Plug-and-Play CDMO Market?
North America is dominating the plug-and-play CDMO market, driven by its early investment in the modular, rapid-deployment biomanufacturing platforms. This accompanying infrastructure is supporting the growing number of biopharmaceutical industries located in the continent. North America is making significant investments in research and development services, driven by government policies in the healthcare sector. This environment fosters a continuous drug development process that relies on Contract Development and Manufacturing Organizations (CDMOs) to support market growth.
U.S. Plug-and-Play CDMO Market Analysis
The U.S. is leading the North America region in the plug-and-play CDMO market by setting the pace with access that enables the formation of a rapidly deployable production environment with pre-validated cleanrooms and exceptional digital automation capability. This extreme efficiency allows for accelerated timelines for onboarding new products. The biologics cluster continues to expand throughout the globe.
How is Asia Pacific the Fastest Growing in the Plug-and-Play CDMO Market?
Asia Pacific is set to be the fastest-growing in the plug-and-play CDMO market, increasingly looking for cost-efficient, flexible manufacturing to support their booming clinical activity. This is underlined by the rapid expansion of biotech startups in the region and increased investment in biologics that heighten the need for modular CDMO infrastructure, which can be deployed quickly. Driven by aggressive facility expansions, China leads this growth, with strong adoption of single-use, ready-to-operate cleanroom systems.
India Plug-and-Play CDMO Market Analysis
India further solidifies this momentum through its thriving generics and biologics ecosystem, wherein plug-and-play facilities help shorten production cycles and reduce compliance burden. Other countries in Southeast Asia are embracing modular facilities to attract global outsourcing partnerships. Combined, the region of Asia Pacific is setting new frontiers of speed, scalability, and affordability in the plug-and-play CDMO landscape.
Who are the Major Players in the Global Plug-and-Play CDMO Market?
The major players in the plug-and-play CDMO market include Lonza Group, Catalent, Inc., Thermo Fisher Scientific (Patheon), WuXi AppTec, Recipharm AB, Boehringer Ingelheim (BioXcellence), Samsung Biologics, Siegfried Holding AG, Aenova Group, and Almac Group.
Recent Developments
- In November 2025, India's energy future is increasingly focused on achieving net-zero goals through the convergence of refining, retail, and mobility sectors. This integration aims to enhance efficiency and reduce carbon emissions in the energy supply chain. By embracing sustainable practices and innovative technologies, India seeks to transform its energy landscape to meet growing demands while addressing environmental concerns. Collaborative efforts between various stakeholders will be crucial in implementing these changes and achieving a sustainable energy ecosystem.(Source: https://energy.economictimes.indiatimes.com)
Segments Covered in the report
By Service Type
- End-to-End Plug-and-Play Services
- Integrated R&D-to-Commercial Manufacturing
- Platform-based development & tech transfer
- Plug-in modular service bundles
- Drug Substance (DS) Services
- Cell line development/Strain development
- Upstream process development
- Downstream purification
- Scale-up & optimization
- DS Manufacturing
- Drug Product (DP) Services
- Formulation development
- Aseptic fill-finish
- Lyophilization services
- Packaging & labelling
- Ready-to-market DP modules
- Analytical & Quality Services
- Method development & validation
- Release testing
- Stability testing
- In-process QC
- Characterization services
- Regulatory & Support Services
- CMC documentation
- Regulatory filing support (IND, NDA, BLA)
- Tech transfer & project management
- Supply chain & logistics integration
- Advanced Therapies-focused DS + DP services
By Molecule Type
- Small Molecules
- Biologics
- Monoclonal antibodies
- Recombinant proteins
- Vaccines
- Biosimilars
- Advanced Therapies
- Cell therapies (CAR-T, NK, stem cell)
- Gene therapies (AAV, LV, plasmid DNA)
- mRNA therapeutics
- Hybrid/Complex Molecules
- Antibody–Drug Conjugates (ADC)
- Peptides & Oligonucleotides
By Development Stage
- Pre-clinical Development
- Clinical Manufacturing
- Phase I
- Phase II
- Phase III
- Commercial-Scale Manufacturing
By Manufacturing Platform
- Single-Use/Disposable Modular Systems
- Hybrid Modular Manufacturing Systems
- Continuous Manufacturing Platforms
- Pre-validated, Pre-configured Suites
- Standalone Plug-and-Play Modules
- Upstream modules
- Downstream modules
- Fill-finish modules
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content
.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
