Medical Affairs Outsourcing Market Revenue to Attain USD 4.12 Bn by 2035
Medical Affairs Outsourcing Market Revenue and Trends 2026 to 2035
The global medical affairs outsourcing market revenue stood at USD 2.08 billion in 2025 and is projected to increase from USD 2.23 billion in 2026 to approximately USD 4.12 billion by 2035, expanding at a CAGR of 7.07% from 2026 to 2035. The medical affairs outsourcing market is gaining prominence as pharmaceutical and biotechnology companies increasingly seek specialized external support to manage scientific communication and regulatory engagement. This market is evolving rapidly with the growing complexity of drug development and the need for real-world evidence generation.

Market briefly
The medical affairs outsourcing market is expected to witness strong growth driven by rising R&D investments and the expanding pipeline of complex therapies. Companies are increasingly outsourcing functions such as medical writing, pharmacovigilance, and field medical support to improve efficiency and reduce operational costs. The growing importance of real-world evidence and post-marketing surveillance is further boosting demand for specialized service providers. Technological advancements, including data analytics and digital platforms, are enhancing the quality and scalability of outsourced services. Additionally, increasing regulatory scrutiny and the need for faster market access are expected to sustain long-term market expansion.
Market Forecast
The IIHMR Delhi blog underscores the major government initiatives to improve the healthcare system in India in terms of policy reforms and digitalization as well as accessibility. Such large-scale schemes as Ayushman Bharat are aimed at universal health coverage and financial insurance, and the National Digital Health Mission is aimed at the establishment of digital health infrastructure and data aggregation. Programs such as the Rashtriya Kishor Swasthya Karyakram and the National AIDS Control Organisation deal with targeted health issues within the populace and enhance service provision. The government is also promoting foreign investment, innovation, and startup involvement to improve on the healthcare infrastructure and affordability. Overall, these initiatives aim to build a more accessible, efficient, and technology-driven healthcare ecosystem in India.
Secondary Data
Factors involved in decision-making regarding pharmaceutical outsourcing
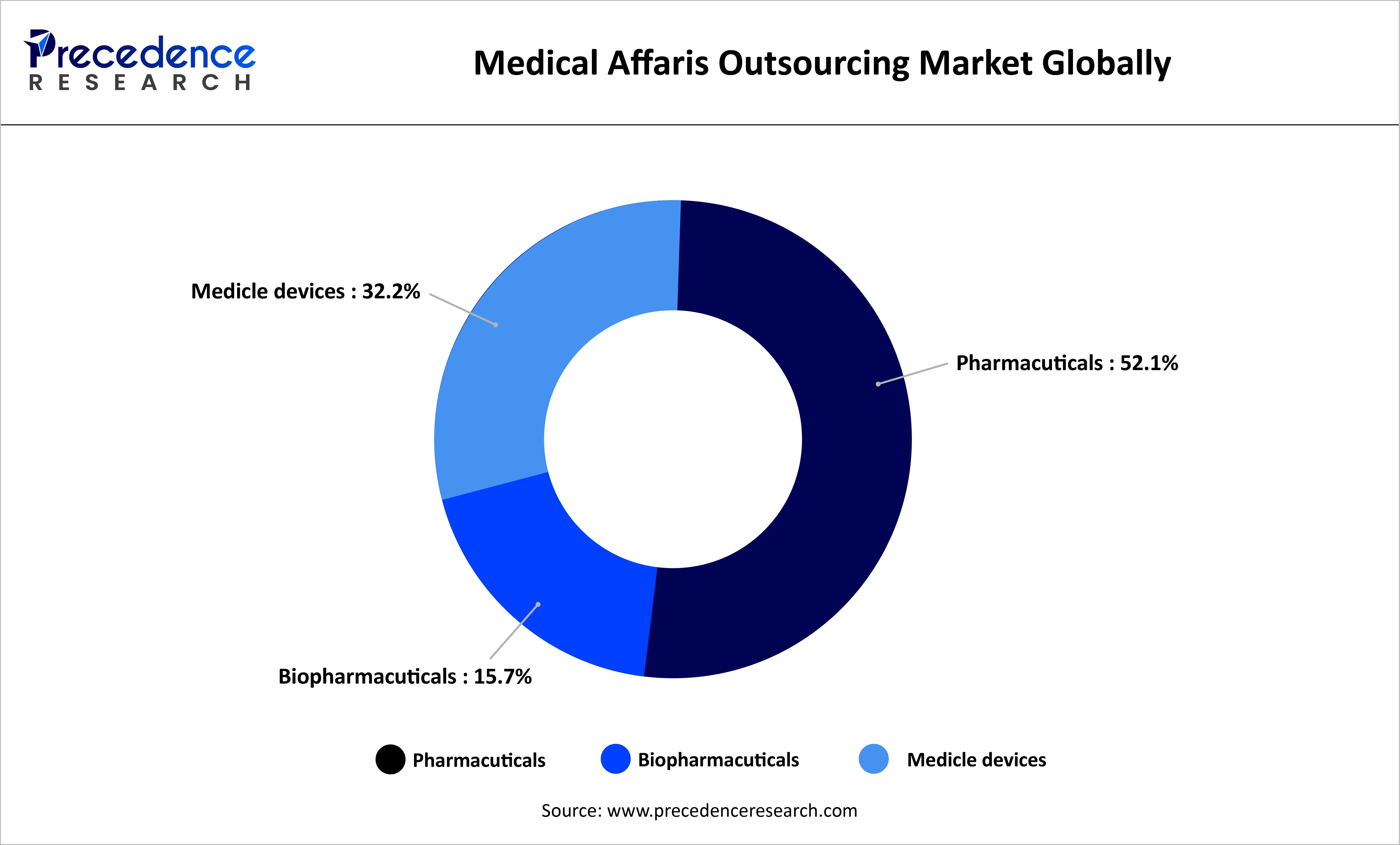
The chart depicts the market of the global medical affairs outsourcing by the end-user in 2022 at a value of 1.7 billion. The pharmaceutical segment has the highest of 52.1, showing that it has very high dependency on outsourcing services in regulatory and scientific support. The medical devices segment comes next with 32.2% and is motivated by the necessity of innovation and compliance. The biopharmaceutical division is smaller, comprising of 15.7% shares but is still important because of increasing biologics development. All in all, pharmaceuticals control the market, with other segments slowly growing to help it develop.
Segmental Insights
Component Insights
The medical affairs outsourcing market is controlled by the service segment because of the great need to have specialized knowledge in medical writing, pharmacovigilance, and field medical support. The pharmaceutical and biotechnology firms are increasingly engaging external service providers to do complex regulatory and scientific communications. Outsourcing services allows for minimising the cost of operations and enhancing efficiency and scalability. The increasing requirement of real-world evidence and post-marketing surveillance further makes this segment stronger.
The solutions segment is the most rapidly expanding, with the increased use of digital channels and integrated solutions. Such solutions are data analytics systems, medical information systems, and compliance management systems. Firms are investing in sophisticated solutions that are aimed at streamlining work processes and improving decision-making. The digital transformation and automation in healthcare is increasing the demand of these platforms. Moreover, the growing regulatory complexity is promoting the adoption of standardized and scalable technological solutions.
Delivery Mode Insights
Web and cloud-based EDI is the most successful in the delivery mode segment because it is flexible and scalable and cost-effective. It allows the flow of information and immediate access to information between various stakeholders. Cloud-based systems are more desirable to organizations because they can be integrated with existing healthcare IT infrastructure. This segment is also increasing with the increased use of digital health technologies and remote operations. Moreover, cloud-based solutions are very reliable with improved data security and compliance capabilities.
The most rapidly expanding part is mobile EDI which is supported by the rising trend of smartphone and mobile app usage in healthcare. It allows healthcare professionals to access and exchange data on the go, improving efficiency and responsiveness. Real-time communication and decision-making are increasing its pace of adoption. Remote monitoring and decentralized clinical operations are also supported by mobile solutions. Mobile EDI is likely to experience a wide growth as digital health ecosystems continue to grow.
End User Insights
The market is dominated by healthcare payers since they need detailed data management, claims processing, and regulatory compliance solutions. They are progressively outsourcing medical affairs functions to increase efficiency in operation and decrease the administrative load. Payer organizations are in dire need of correct and prompt information flow. The increasing costs and complicated reimbursement frameworks in healthcare are also contributing to the need to outsource. Moreover, the implementation of online systems promotes better data management and analytics.
The most rapidly expanding segment is healthcare providers with growing numbers of patients and the necessity of effective clinical operations. Hospitals and clinics are outsourcing services to administer medical information, compliance, and interaction with patients. The change to value-based care and digital health solutions is increasing demand. Another trend that is being taken advantage of by providers is outsourcing to save on costs and concentrate on patient care activities. This market will grow at an alarming rate as the healthcare infrastructure continues to develop.
Regional Insights
North America
North America controls the medical affairs outsourcing market because of the existence of a well-established pharmaceutical and biotechnology industry. The United States is a leading player that invests heavily in R&D and has a well-developed healthcare system. The area is enjoying good regulatory systems and an early embrace of outsourcing models. Market growth is facilitated by the growing demand for real-world evidence and post-marketing surveillance. Also, the existence of major service providers promotes competitiveness and innovation. North America is also dominant due to the continuous technological advancements.
Asia Pacific
Asia Pacific is the most rapidly developing region with the growing pharmaceutical markets and the growing number of clinical research activities. India and China are the other countries that are becoming major outsourcing destinations because of the cost benefits and availability of skilled labor. Demand is being boosted by growing healthcare infrastructure and increasing investments in R&D. Digital health technologies and outsourcing services are also on the rise in the region. Market growth is being boosted by favorable government efforts and regulatory improvements. All in all, the Asia Pacific will continue to grow at a very fast pace in the next few years.
Top Companies in the Medical Affairs Outsourcing Market
- OSP: Creates artificial intelligence-based healthcare software, such as telehealth EHR systems and HIPAA licensed RCM systems.
- MCKESSON CORPORATION: Offers pharmaceutical distribution and health IT solutions such as the supply chain management system and pharmacy automation system.
- NXGN Management, LLC: Offers combined EHR practice management cloud services, RCM solutions, and patient engagement tools.
- Optum, Inc. : Provides smarter EDI solutions that allow claims processing and eligibility of claims based on claims and guarantee a safe provider-payer connection.
Segments Covered in the Report
By Component
- Services
- Solutions
By Delivery Mode
- Web and Cloud-based EDI
- EDI Value Added Network (VAN)
- Direct (Point-to-Point) EDI
- Mobile EDI
By End-Use
- Healthcare Payers
- Healthcare Providers
- Pharmaceutical & Medical Device Industries
- Others
By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
Get this report to explore global market size, share, CAGR, and trends, featuring detailed segmental analysis and an insightful competitive landscape overview @https://www.precedenceresearch.com/sample/1113
You can place an order or ask any questions, please feel free to contact us at sales@precedenceresearch.com |+1 804 441 9344