What is the Rheumatology Therapeutics Market Size?
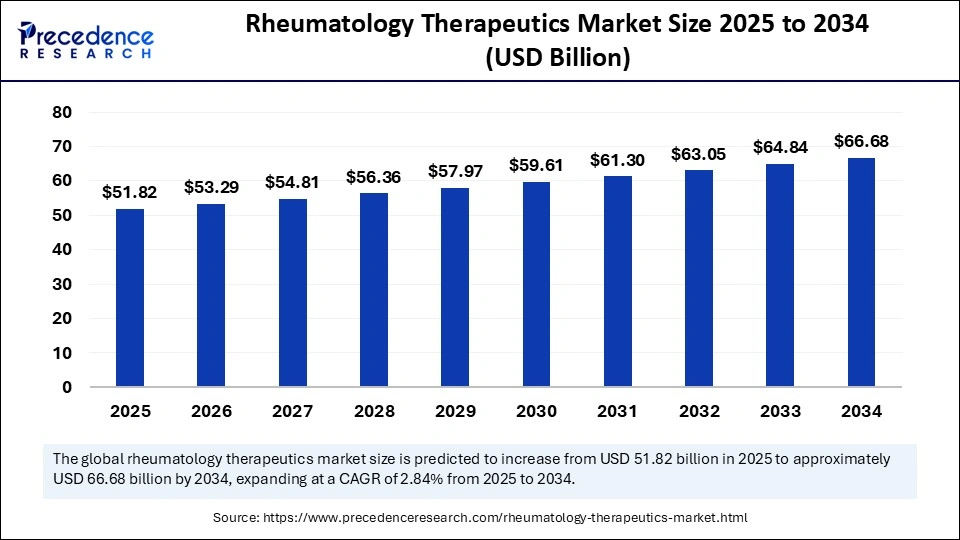
The global rheumatology therapeutics market size is calculated at USD 51.82 billion in 2025 and is predicted to increase from USD 53.29 billion in 2026 to approximately USD 66.68 billion by 2034, expanding at a CAGR of 2.84% from 2025 to 2034. Market growth is driven by the rising prevalence of autoimmune diseases, the growing adoption of biologics, and the integration of advanced technologies, including AI, for personalized treatment.
Market Highlights
- North America segment held a dominant presence in the market in 2024, accounting for an estimated 41% market share.
- The Asia Pacific segment is expected to grow at the fastest rate in the market during the forecast period of 2025 to 2034.
- By drug type, the DMARDs segment accounted for the biggest market share of 55% in 2024.
- By drug type, the biologic DMARDs segment is projected to experience the highest growth rate between 2025 and 2034.
- By disease indication, the rheumatoid arthritis segment led the market, accounting for an estimated 42% market share in 2024.
- By disease indication, the gout segment is set to experience the fastest rate from 2025 to 2034.
- By route of administration, the oral segment held the major market share of 60% in 2024.
- By route of administration, the injectable/infusion segment is anticipated to grow with the highest CAGR between 2025 and 2034.
Advancing Therapeutics in Autoimmune and Inflammatory Diseases
The rising popularity of autoimmune disorders, including rheumatoid arthritis (RA), is stimulating the use of artificial intelligence (AI) in rheumatology therapy. The use of AI technologies is based on sophisticated data analysis, which enables early diagnosis, tailored treatment regimens, and better patient outcomes. AI algorithms have shown that they assist clinical professionals in predicting treatment response in RA patients, enabling clinicians to select more appropriate treatment options.
The U.S. Centers for Medicare & Medicaid Services (CMS) has acknowledged the importance of precision medicine in rheumatology, with tests such as PrismRA endorsed to predict treatment response. Thus, treatment decisions are made to mitigate unnecessary disease progression and resultant healthcare expenses. Besides, the report of the Digital Rheumatology Network conference highlights growing interest and cooperation in AI applications in rheumatology, as 15 countries were represented by their participants, discussing progress and issues in the field.
AI and Analytics in Life Sciences: From Hype to Real-World Value
Artificial intelligence (AI) is transforming the therapeutic experience in rheumatology by improving drug discovery, clinical decision-making, and patient care. Firms are developing AI-based models to identify new targets for autoimmune and inflammatory diseases, maximize opportunities in biologics and small molecules, and better predict patient responses. Moreover, artificial intelligence enhances supply chain and production operations, ensuring on-time deliveries and preserving biological stability for temperature-sensitive products.
Unlocking Growth Through Innovation in Life Sciences
- Rising Adoption of Biologic Therapies: Growing clinical evidence and patient preference for targeted biologics are boosting market expansion and treatment effectiveness.
- Expansion of Telemedicine and Digital Health Tools: The integration of remote monitoring and AI-enabled platforms is driving more personalized care and improved disease management.
- Increasing Clinical Trial Initiatives: Rising global clinical trial volumes for novel autoimmune therapies are fuelling innovation and faster drug approvals.
- Growing Awareness of Early Diagnosis: Enhanced educational campaigns and patient screening programs are propelling early intervention and improved treatment outcomes.
- Rising Investment in Biopharmaceutical R&D: Growing funding by biotech and pharmaceutical companies is boosting pipeline development and next-generation therapeutics.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 51.82 Billion |
| Market Size in 2026 | USD 53.29 Billion |
| Market Size by 2034 | USD 66.68 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 2.84% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Drug Type, Disease Indication, Route of Administration, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Regulatory Landscape Table for the Rheumatology Therapeutics Market
| Country/Region | Regulatory Body | Key Regulations | Focus Areas | Notable Notes |
| United States | U.S. FDA (Food and Drug Administration) | Biologics License Application (BLA) IND (Investigational New Drug) FDA Guidance on Biosimilars | Clinical trial approval Biologic safety & efficacy Manufacturing standards | The FDA provides fast-track and breakthrough therapy designations for RA biologics. Real-world evidence increasingly informs regulatory decisions. |
| European Union | EMA (European Medicines Agency) | Centralized Marketing Authorization Advanced Therapy Medicinal Products (ATMP) Regulation Biosimilar Guidelines | Safety and efficacy of biologics Biosimilar approval pathways Post-marketing surveillance | EMA leads in biosimilar approvals for RA; it encourages harmonized clinical trial requirements across member states. |
| China | NMPA (National Medical Products Administration) | Drug Administration Law Technical Guidelines for Biologics and Biosimilars | Biologic registration Clinical trial authorization Good Manufacturing Practice (GMP) compliance | Recent reforms aim to speed up approval for innovative biologics; local clinical trials are often required for market entry. |
| Japan | PMDA (Pharmaceuticals and Medical Devices Agency) | Pharmaceutical and Medical Device Act Guidelines for Biologics and Biosimilars | Pre-market approval Post marketing surveillance Manufacturing quality | Japan has a dedicated pathway for biosimilars, with emphasis on demonstrating similarity to reference products. |
| India | CDSCO (Central Drugs Standard Control Organization) | Drugs and Cosmetics Act Guidelines on Biosimilars Clinical Trial Rules, 2019 | Approval of biologics and biosimilars Clinical trial oversight Pharmacovigilance | India is expanding biosimilar adoption; local clinical trial data are often required for regulatory approval. |
| Brazil | ANVISA (Agencia Nacional de Vigilancia Sanitaria) | Brazilian Health Regulatory Agency Regulations for Biologics Clinical Trials Regulations | Biologic registration Clinical trial approval Pharmacovigilance | ANVISA has a simplified pathway for biosimilars; post-marketing surveillance is mandatory. |
| South Korea | MFDS (Ministry of Food and Drug Safety) | Pharmaceutical Affairs Act Biologics and Biosimilars Guidelines | Clinical trial approval Biologics safety and efficacy GMP compliance | South Korea emphasizes robust clinical trial data for biosimilars; rapid approval is encouraged for unmet medical needs. |
| Australia | TGA (Therapeutic Goods Administration) | Therapeutic Goods Act Guidelines for Biosimilars | Pre-market approval Safety monitoring Post-market reporting | TGA aligns biosimilar guidelines closely with EMA; it encourages international clinical trial data submission. |
Charting the Future of Rheumatology Therapies: Global Data, Clinical Advances, and Access Insights
- As of 2021,17.9 million people globally live with rheumatoid arthritis (RA), with an age-standardized prevalence of 208.8 per 100,000 population. High-income countries report the highest prevalence rates, reflecting better diagnosis and reporting systems. This rising prevalence drives demand for advanced biologic therapies.
- Women make up 70% of the global RA population, with 55% over 55 years of age. The higher disease burden among older adults increases the need for effective long-term biologic treatments. Age and gender factors influence prescribing trends and market penetration of targeted therapies.
- In 2023, 13 million RA patients experience moderate-to-severe disease, indicating high unmet medical need. These patients are the primary candidates for biologic therapies, driving prescription volumes and market growth. Advanced therapies improve disease control, reduce complications, and influence market adoption trends.
- An estimated 2.4 million patients require rehabilitation for joint function and mobility in 2022. Integration of biologic therapies with physical therapy improves outcomes. Addressing rehabilitation needs influences market growth and therapy adherence in both developed and emerging markets.
- As of 2024, over 1,100 clinical trials for rheumatoid arthritis (RA) have been initiated since 2018. Mainland China leads with 58% of these trials, followed by South Korea and the United States. This concentration reflects Chinas significant role in RA research and development.
- The global RA pipeline comprises over 80 treatment therapies developed by more than 75 key companies. These therapies include both small molecules and biologics, addressing various aspects of RA management. The distribution of these therapies varies by region, with North America and Europe hosting a significant portion of the pipeline.
- Between 2021 and 2023, pharmaceutical companies invested over $21 billion globally into autoimmune disease research, development, and production capacity expansion. In North America, R&D investments exceeded $9.8 billion, with multiple phase II and III programs targeting rheumatoid arthritis, systemic lupus, psoriasis, and inflammatory bowel disease.
- In H1 2025, significant R&D partnerships were formed, including a $3.4 billion deal between Syneron Bio and AstraZeneca, and a $1.7 billion collaboration between Earendil Labs and Sanofi. These partnerships focus on advancing therapies for autoimmune diseases, highlighting the industrys commitment to innovation.
- China has emerged as a global leader in life sciences R&D, surpassing the U.S. in the number of clinical trials conducted. In 2024, China conducted over 7,100 clinical trials, compared to about 6,000 in the U.S., reflecting a strategic national focus on biopharmaceutical innovation.
Rheumatology Therapeutics Market Segment Insights
[[segment_insights]]
Rheumatology Therapeutics Market Regional Insights
[[regional_insights]]
Rheumatology Therapeutics Market Value Chain
[[value_chain]]
Rheumatology Therapeutics Market Companies
[[market_company]]
Other Companies in the Rheumatology Therapeutics Market
- Johnson & Johnson (Janssen Pharmaceuticals): Janssen leads in biologic immunotherapies with Remicade (infliximab) and Simponi (golimumab). The companys pipeline includes IL-23 and IL-12 inhibitors targeting immune-mediated inflammatory diseases, with ongoing research into combination therapies and next-gen biologics for arthritis and lupus management.
- Amgen Inc.: Amgens rheumatology franchise centers on Enbrel (etanercept) and its biosimilars, used extensively in rheumatoid and psoriatic arthritis. The companys R&D pipeline focuses on osteoporosis, inflammatory cytokine modulation, and T-cell regulation, reinforcing its legacy in immune and bone health therapeutics.
- Bristol-Myers Squibb Company (BMS): BMS markets Orencia (abatacept), a CTLA-4 immunoglobulin fusion protein for RA and juvenile idiopathic arthritis. The company is advancing novel checkpoint and cytokine inhibitors to address unmet needs in autoimmune rheumatic disorders, supported by strong biologic development expertise.
- Novartis AG: Novartis offers Cosentyx (secukinumab), an IL-17A inhibitor indicated for psoriatic arthritis and ankylosing spondylitis, and Ilaris (canakinumab) for systemic autoinflammatory diseases. Its focus on IL-targeted and biologic therapies positions it as a major innovator in chronic inflammation management.
- UCB S.A.: UCB is a key player in autoimmune and inflammatory disorders, marketing Cimzia (certolizumab pegol) for RA, psoriatic arthritis, and ankylosing spondylitis. The company pipeline includes bimekizumab, a dual IL-17A/F inhibitor, demonstrating its strength in next-generation biologics for immunology.
- Eli Lilly and Company: Lillys rheumatology portfolio features Taltz (ixekizumab), an IL-17A inhibitor for psoriatic and axial spondyloarthritis, and Olumiant (baricitinib), a JAK inhibitor for RA. The company continues to expand its immunology and inflammation division, exploring oral and biologic therapies with improved patient outcomes.
- Gilead Sciences, Inc.: Gilead has established a foothold in rheumatology through JAK inhibitor-based therapies, notably filgotinib (Jyseleca), developed with Galapagos NV. The company focuses on oral targeted immunomodulators and strategic partnerships to enhance its inflammation and autoimmune therapeutic pipeline.
- Takeda Pharmaceutical Company Ltd.: Takedas rheumatology portfolio emphasizes immunoglobulin therapies and biologics addressing systemic autoimmune diseases. The company invests in novel anti-inflammatory pathways and personalized immunotherapy platforms targeting both rheumatologic and gastrointestinal immune disorders.
- Roche Holding AG: Roche, through its Genentech division, markets Actemra (tocilizumab), an IL-6 receptor inhibitor used for rheumatoid arthritis and giant cell arteritis. The companys continued innovation in B-cell depletion and cytokine blockade reinforces its leadership in immune-targeted biologics.
- AstraZeneca plc: AstraZeneca is expanding in rheumatology via its Alexion Pharmaceuticals acquisition, focusing on rare autoimmune and complement-mediated disorders. Its biologics and precision-medicine pipeline target lupus, vasculitis, and other systemic autoimmune diseases.
- Sandoz (Novartis): Sandoz leads the biosimilars market with affordable alternatives to major biologics such as etanercept and adalimumab. Its focus on expanding global access to rheumatology biologics underpins its strength in the cost-competitive segment of immune therapies.
- Biogen Inc.: Biogen is developing immunology therapeutics targeting B-cell modulation and inflammatory signaling. The companys collaboration and pipeline investments in anti-TNF biosimilars and autoimmune therapies broaden its footprint in rheumatologic indications.
- Sanofi S.A.: Sanofi, in collaboration with Regeneron, offers Kevzara (sarilumab), an IL-6 receptor antagonist for rheumatoid arthritis. The company robust immunology R&D also includes Dupixent (dupilumab), extending into systemic inflammatory diseases relevant to rheumatology.
Recent Developments
- In October 2025, Fate Therapeutics, Inc. unveiled fresh and updated findings from the first ten patients treated with FT819, an investigational iPSC-derived off-the-shelf cell therapy, in a Phase 1 trial for moderate-to-severe systemic lupus erythematosus (SLE). The announcement came during the American College of Rheumatology (ACR) Convergence 2025 in Chicago, underscoring Fates ongoing efforts to make advanced cellular immunotherapies broadly accessible to patients. (Source: https://ir.fatetherapeutics.com)
- In October 2025, Novartis confirmed it will present 27 abstracts spanning its Immunology pipeline and portfolio at the ACR Convergence 2025. The presentations will include pivotal Phase III data from the NEPTUNUS-1 and NEPTUNUS-2 trials assessing ianalumab in Sjögren disease, as well as new biomarker insights from a Phase 1/2 study of rapcabtagene autoleucel in severe refractory SLE. The company will also share fresh results on Cosentyx across multiple rheumatology indications. (Source: https://www.novartis.com)
- In October 2025, Artiva Biotherapeutics, Inc. announced that the U.S. FDA had granted Fast Track Designation to its investigational therapy AlloNK (AB-101) for refractory rheumatoid arthritis (RA) in combination with rituximab. The company named refractory RA its lead program, marking a significant milestone for AlloNK, which is considered the first deep B-cell depleting therapy to receive this designation in RA. (Source: https://investors.artivabio.com)
- In July 2025, SetPoint Medical reported FDA approval for its innovative SetPoint System, a neuroimmune modulation device designed to treat adults suffering from moderate-to-severe rheumatoid arthritis (RA) who have shown inadequate response or intolerance to biologic or targeted synthetic DMARDs. The approval positions the SetPoint System as a novel, device-based alternative in RA therapy. (Source: https://setpointmedical.com)
- IN October 2025, UCB shared new three-year follow-up data from Phase 3 clinical studies and open-label extensions evaluating BIMZELX (bimekizumab-bkzx) in patients with psoriatic arthritis (PsA), non-radiographic axial spondyloarthritis (nr-axSpA), and ankylosing spondylitis (AS). As the first therapy to selectively block interleukin-17A and interleukin-17F, BIMZELX continued to show durable inflammation control and strong efficacy across these rheumatologic conditions.(Source: https://www.prnewswire.com)
Rheumatology Therapeutics Market Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content







 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
