What is S1P Receptor Modulator Drugs Market Size in 2026?
The global S1P receptor modulator drugs market size was calculated at USD 3.16 billion in 2025 and is predicted to increase from USD 3.42 billion in 2026 to approximately USD 6.96 billion by 2035, expanding at a CAGR of 8.22% from 2026 to 2035. The rising incidence of multiple sclerosis and ulcerative colitis, unmet oral therapy needs, and safety are driving the growth of these transformative pharmaceutical drugs.
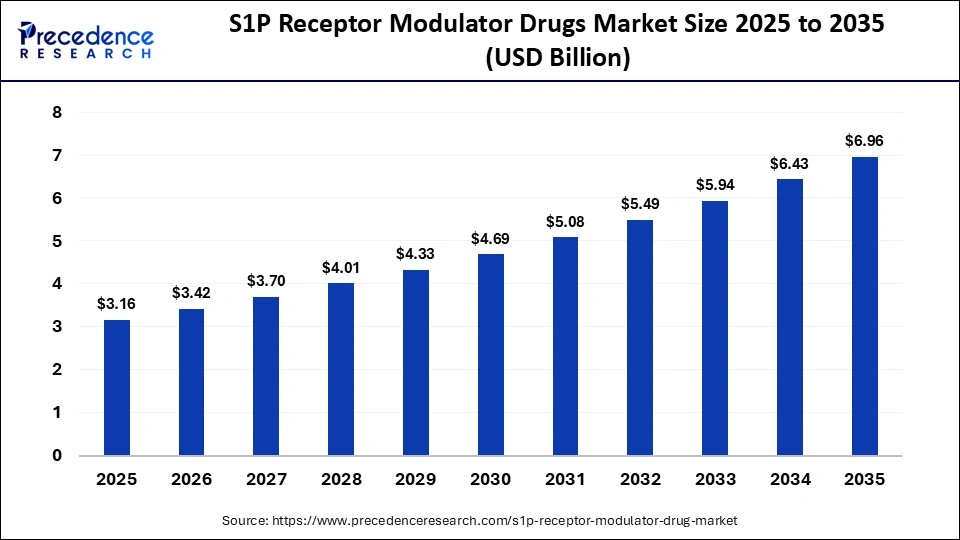
Key Takeaways
- North America dominated the S1P receptor modulator drugs market in 2025.
- Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period.
- By application, the multiple sclerosis segment dominated the market in 2025.
- By application, the cardiovascular diseases segment in the market is expected to grow at the fastest CAGR in the market during the forecast period.
- By route of administration, the oral segment dominated the market in 2025.
- By route of administration, the injectable segment is expected to grow at the fastest rate in the market in 2025.
- By drug class, the sphingosine 1-phosphate receptor modulators segment dominated the market in 2025.
- By drug class, the immunomodulators segment is expected to grow at the fastest CAGR in the market during the forecast period.
- By patient type, the adult segment dominated the market in 2025.
- By patient type, the geriatric segment is expected to grow at the fastest CAGR in the market during the forecast period.
S1P Receptor Modulator Drugs: Driven by Robust Clinical Research
The S1P receptor modulator drugs market is growing and expanding due to the increasing shift towards oral therapies and patients' strong preference for oral small-molecule drugs. The oral medications are highly preferred over traditional injectables and intravenous biologics due to a lack of immunogenicity and convenience. The market is driven by a transition from multiple sclerosis to ulcerative colitis and other autoimmune disorders. Research and safety innovations focus on the next-generation modulators with higher receptor selectivity to reduce side effects like lymphopenia and bradycardia. The leading players, such as Bristol Myers Squibb, Novartis, and Pfizer, are investing heavily in R&D to maintain competitiveness.
What is AI transforming the S1P Receptor Modulator Drugs Market?
Artificial Intelligence tools are transforming the pharmaceutical industry by accelerating the optimization of treatment outcomes and the identification of new therapeutic options. AI enables the integration and analysis of complex chemical, biological, and clinical data, which helps to expand the market globally. It also facilitates personalized therapeutic approaches, drug discovery, and patient stratification. AI helps researchers to develop precision medicine strategies and supports in identifying biomarkers. AI is used to predict drug-target interactions and match disease-specific molecular structures with drug-induced gene expression profiles.
S1P Receptor Modulator Drugs Market Trends
- Shift to Next-Generation Selectivity: The emerging advances across the S1P receptor modulator drugs market include safety improvements, selective targeting, and faster recovery. The industries are rapidly shifting from non-selective and first-generation drugs like Fingolimod due to the imposing cardiac side effects.
- Rapid Expansion into Non-Multiple Sclerosis (MS) Indications: There is an emerging class and a major oral alternative to biologics for ulcerative colitis. The European Medicines Agency (EMA) approved Etrasimod (Velsipity) for ulcerative colitis, followed by its FDA clearance. The ongoing research focuses on Crohn's disease, atopic dermatitis, and alopecia areata.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 3.16 Billion |
| Market Size in 2026 | USD 3.42 Billion |
| Market Size by 2035 | USD 6.96 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 8.22% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Application, Route of Administration, Drug Class, Patient Type, and region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segmental Insights
Application Insights
How does the Multiple Sclerosis Segment Dominate the S1P Receptor Modulator Drugs Market in 2025?
The multiple sclerosis segment dominated the market in 2025, owing to the U.S. Food and Drug Administration (FDA) approval for four sphingosine 1â€phosphate (S1P) receptor modulators (fingolimod, ozanimod, ponesimod, and siponimod) for the treatment of multiple sclerosis. The S1P receptor modulators are good therapeutic options for immune-mediated diseases, like multiple sclerosis. Researchers make efforts to evaluate the efficacy of S1P receptor modulators, like fingolimod, ozanimod, ponesimod, and Siponimod for multiple sclerosis in phase 2 and phase 3 clinical trials.
The cardiovascular diseases segment is expected to grow at the fastest CAGR in the S1P receptor modulator drugs market during the forecast period due to the importance of sphingosine-1-phosphate (S1P) as an integral sphingolipid molecule to regulate cardiovascular functions in physiological and pathological conditions. Research and clinical programs prioritize the integrated safety analysis of patients who are dealing with moderate to severe active ulcerative colitis. The S1P receptor modulator drugs deliver strong physiological effects in the treatment of bradyarrhythmia, hypertension, and other cardiovascular conditions.
Route of Administration Insights
What made Oral the Dominant Segment in the S1P Receptor Modulator Drugs Market in 2025?
The oral segment dominated the market in 2025, owing to the improved benefits of oral dosing over injectable or infusible biologics in enhancing patient adherence. Oral small molecules like S1P modulators have lower manufacturing costs than biologics. The oral formulations deliver a strong economic impact, which enables an easier distribution of drugs through retail pharmacies.
The injectable segment is estimated to grow at the fastest rate in the S1P receptor modulator drugs market during the predicted timeframe due to the high preference for injectables in patients who are intolerant to oral medication and need strict monitoring. Fingolimod is adaptable for use in combination with other injectable therapies in the treatment of multiple sclerosis. Research focuses on many delivery methods, including topical and transdermal fingolimod, which replace or complement traditional injectable needs.
Drug Class Insights
How did the Sphingosine 1-Phosphate Receptor Modulators Segment Dominate the S1P Receptor Modulator Drugs Market in 2025?
The sphingosine 1-phosphate receptor modulators segment dominated the market in 2025, owing to a rapid industrial shift from first-generation drugs to more selective and second-generation agents with improved safety profiles. The new indications, like ozanimod and etrasimod, are leading in the market due to dual indications in multiple sclerosis and ulcerative colitis. There are expanding therapeutic applications of these drugs across gastroenterology, which further offer an oral alternative to injectable biologics.
The immunomodulators segment is anticipated to grow at a notable rate in the S1P receptor modulator drugs market during the upcoming period due to patient compliance via oral formulations, where S1P modulators offer an oral alternative to injectable biologics. They enhance patient adherence and reduce hospital-based infusion costs. These drugs are primarily used for advanced therapies for the treatment of multiple sclerosis and ulcerative colitis.
Patient Type Insights
Why did the Adult Segment dominate the S1P Receptor Modulator Drugs Market in 2025?
The adult segment dominated the market in 2025, owing to the specific safety and monitoring requirements for the treatment of cardiac conditions and infections. The ongoing research focuses on Crohn's disease, Systemic Lupus Erythematosus (SLE), and Sjogren's syndrome by using S1P modulators and real-world data. Clinical trials prioritize rapid symptom relief, a fast onset of action, and reduction of symptoms.
The geriatric segment is predicted to grow at a rapid rate in the S1P receptor modulator drugs market during the studied period due to the expansion of major S1P modulators like ozanimod, etrasimod, and siponimod for older adults. These drugs are renowned as first-line advanced therapies and are most effective in patients who have adapted to biologics. The oral administration is ideal for the geriatric population, which often requires once-daily tablets over subcutaneous or intravenous therapies.
Regional Insights
How does North America dominate the S1P Receptor Modulator Drugs Market in 2025?
North America dominated the market in 2025, owing to the rising prevalence of autoimmune diseases, favorable reimbursement and regulatory environment, and strategic product launches. The robust clinical trials for next-generation modulators like Cenerimod reached efficacy and safety. There is an increased focus of healthcare systems on the improved diagnosis of autoimmune diseases. The government programs include Canada's National Pharmacare Act, National Strategy for Drugs for Rare Diseases in Canada, FDA Target Zero Campaign, and ARPA-H GLIDE Program in the USA. The Advanced Research Projects Agency for Health (ARPA-H) launched the Groundbreaking Lymphatic Interventions and Drug Exploration (GLIDE) program to promote and advance innovative drug delivery solutions.
U.S. S1P Receptor Modulator Drugs Market Analysis
The innovations in next-generation therapies have increased the preference of patients and clinicians for oral immunotherapies over traditional infused or injectable treatments. These major driving forces boost the adoption of S1P modulators in the U.S. and expand the market. In December 2024, Everest Medicines announced acceptance of VELSIPITY, a new drug application for the treatment of adult patients with moderate to severe ulcerative colitis.
What is the Potential of the S1P Receptor Modulator Drugs Market in the Asia Pacific?
Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period due to the shift to oral therapies, increased healthcare spending, expanding clinical research, and regulatory approvals. The leading Asian Pacific countries like Hong Kong, Japan, South Korea, and mainland China are supporting drug launches and expanded access in Asian Pacific hospitals. Japan's Ministry of Health, Labor, and Welfare (MHLW) introduced a 10-year government fund to support innovative drug development by targeting S1P modulators.
India S1P Receptor Modulator Drugs Market Trends
The rising pharmaceutical R&D investments and global capability centers for clinical trials and advanced analytics are positioning the market in India. In October 2025, the Indian government launched a ₹5,000 crore initiative, which aims to promote research and innovation in the medical technology and pharmaceutical sectors. There is a strong focus of the Indian pharmaceutical sector on generic drug manufacturing.
How does Europe advance Notably in the S1P Receptor Modulator Drugs Market?
Europe is expected to grow at a notable rate in the market, owing to new regulatory approvals, extended patent protection, and improved market access to targeted immune therapies. The government regulatory programs include the pharmaceutical strategy for Europe, the Priority Medicines scheme, and the Important Project of Common European Interest in health. The IPCEI Med4Cure project includes 14 R&D projects from 13 companies to boost medical progress and advance a resilient pharmaceutical value chain in Europe.
Germany S1P Receptor Modulator Drugs Market Analysis
The expanding therapeutic applications, supportive healthcare framework, and robust R&D and clinical trials are transitioning the market in Germany. In January 2026, the German government launched a new pharma and MedTech dialogue, which aims to develop regulatory reform proposals and new policies to recreate Germany as an innovation-friendly hub for life sciences R&D and manufacturing.
Value Chain Analysis
- R&D
In this stage, researchers integrate AI and deep learning to analyze molecular dynamics and predict drug-target interactions.
Key Players: Bristol Myers Squibb, Novartis, Pfizer, Johnson & Johnson, Vanda Pharmaceuticals, Priothera, Teva Pharmaceuticals, Viatris, Dr. Reddy's Laboratories, Biogen, Sanofi, Merck & Co. - Distribution to Hospitals, Pharmacies
This stage prioritizes high-level batch traceability, specialized labelling, and high-quality due to the different dosage strengths required for many treatment stages.
Key Players: Bristol Myers Squibb, Novartis, Pfizer Inc., Vanda Pharmaceuticals, Cencora, McKesson Corporation, Cardinal Health, DHL Life Sciences, Viatris, Dr. Reddy's Laboratories, Teva Pharmaceutical Industries. - Patient Support & Services
This stage encompasses clinical onboarding, safety monitoring, financial access, reimbursement hubs, and digital and AI-driven engagement.
Key Players: UnitedHealth Group, Infinitus, Bristol Myers Squibb, Novartis, Pfizer Inc., Vanda Pharmaceuticals, Cencora, McKesson Corporation.
S1P Receptor Modulator Drugs Market Companies
- Bristol Myers Squibb
- Novartis International AG
- Pfizer Inc.
- Johnson & Johnson
- Vanda Pharmaceuticals
- Teva Pharmaceutical Industries
- Viatris
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Idorsia Pharmaceuticals
- Mitsubishi Tanabe Pharma
- Immunic AG
Recent Developments
- In March 2024, Bristol Myers Squibb announced an update on the first phase 3 YELLOWSTONE trial, followed by the evaluation of oral zeposia (ozanimod) in patients with moderate to severe active Crohn's disease. YELLOWSTONE is a Phase 3 multicenter clinical trial program, which includes two 12-week induction studies, a 264-week open-label extension study, and a 52-week maintenance study. (Source: https://news.bms.com)
- In February 2024, Pfizer Inc. announced that the European Commission approved the S1P receptor modulator Velsipity (etrasimod) of Pfizer for patients aged 16 and older, who are dealing with moderate-to-severe active ulcerative colitis (UC), and who were less responsive to biological agents or traditional treatments, or who are intolerant to these treatments. Research suggested that once-daily oral medicine proves to offer an effective advanced treatment for people who are anxious about using injectable therapies such as biologics. (Source: https://firstwordpharma.com)
Segments Covered in the Report
By Application
- Multiple Sclerosis
- Cardiovascular Diseases
- Transplant Rejection
By Route of Administration
- Oral
- Injectable
- Topical
By Drug Class
- Sphingosine 1-Phosphate Receptor Modulators
- Immunomodulators
- Antiproliferatives
By Patient Type
- Pediatric
- Adult
- Geriatric
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content





 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
