What is the Contract Pharmaceutical Fermentation Services Market Size?
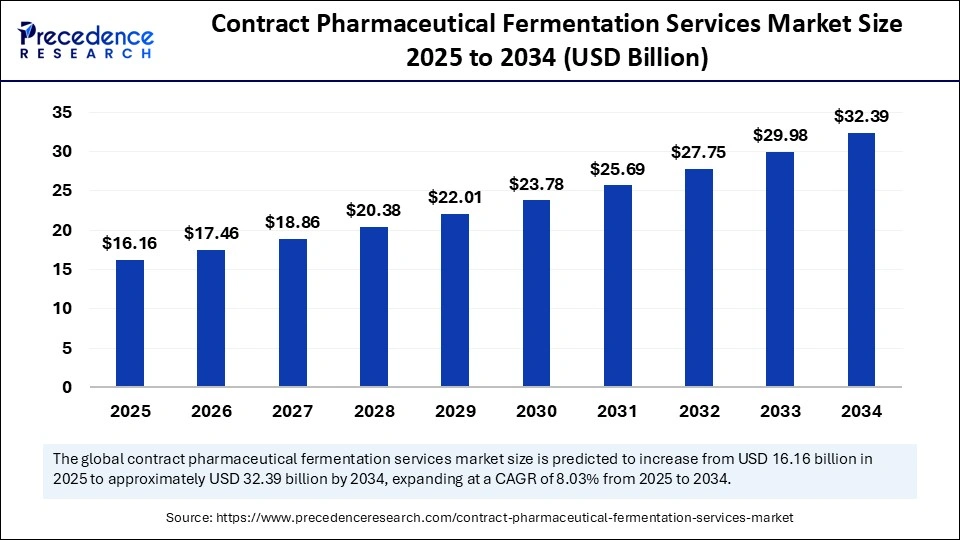
The global contract pharmaceutical fermentation services market size accounted for USD 16.16 billion in 2025 and is predicted to increase from USD 17.46 billion in 2026 to approximately USD 32.39 billion by 2034, expanding at a CAGR of 8.03% from 2025 to 2034. The market is driven by increasing demand for biologics, rising outsourcing among pharma companies, and advancements in microbial and cell-culture fermentation technologies.
Market Highlights
- North America dominated the market, with a 45.4% market share in 2024.
- The Europe is expected to grow at the fastest CAGR of 8.8 between 2025 and 2034.
- By service type, the commercial GMP manufacturing segment held the major market share of 37.4% in 2024.
- By service type, the process development & tech transfer segment is expected to grow at a solid CAGR of 8.2% between 2025 and 2034.
- By scale/capacity, the large-scale commercial segment contributed the highest market share of 54.4% in 2024.
- By scale/capacity, the lab/pilot scale segment is growing at a solid CAGR of 8.3% in between 2025 and 2034.
- By organism/host system, the mammalian cell culture segment generated the biggest market share of 52.10% in 2024.
- By organism/host system, the viral vector/cell therapy platforms is growing at a solid CAGR of 8.1% between 2025 and 2034.
- By product type, the mAbs segment Accounted for the major market share of 40.1% in 2024.
- By product type, the cell & gene therapy (viral vectors) segment is expected to expand at a strong CAGR of 8.0% between 2025 and 2034.
- By end-user, the large pharma & biotech segment held the largest share of 42.5% in the AI in telemedicine market during 2024.
- By end-user, the biotechnology companies/biotechs are set to grow at a remarkable share of 8.4% CAGR between 2025 and 2034.
Redefining Biologic Manufacturing: How Contract Fermentation Service is Driving the Next Pharmaceutical Innovation
Contract pharmaceutical fermentation services that offer specialized third-party services in biologics, vaccines, recombinant proteins, enzymes, and fermentation-derived API manufacturing. With microbial (bacterial and yeast) and mammalian cell systems, Contract Development and Manufacturing Organizations (CDMOs) provide a complete set of services in the upstream fermentation development, process optimization, scale-up, and a GMP-compliant manufacturing solution. CDMOs provide individualized fermentation capacity and technical expertise, which allow a pharmaceutical or biotech firm to speed up product pipelines with high-quality and compliance rates, without the financial investment of in-house infrastructure.
The upsurge in biologics and biosimilars, the increasing investment in R&D, and the rising outsourcing of the manufacturing process. Pharmaceutical companies are searching to find fast and low-cost remedies to handle sophisticated fermentation procedures and comply with world regulatory standards. Due to the continued reliance of biologic therapies on therapeutic pipelines, the necessity to have scalable, high-yield, and efficient fermentation is causing increased collaboration between drug developers and CDMOs. Moreover, automation, AI-enhanced process optimization, and fermentation systems are being deployed by service providers due to the increasing demand for personalized medicine and sustainable techniques of biomanufacturing.
Smart Fermentation: AI Redefines Biopharma Manufacturing
The contract pharmaceutical fermentation service market is transforming with the revolution of artificial intelligence that optimizes the operations, forecasts, and regulates quality. The AI-based algorithms can be used to monitor fermentation parameters, including the pH, temperature, and oxygen, in real-time and guarantee the right production yield and reduced variability. Firms such as Lonza, Thermo Fisher Scientific, and WuXi Biologics are implementing AI-based systems to manage bioprocesses to speed up process development and scale-up, and to minimize the cost of operation. In addition, the integration with robotics and automation increases the compliance with GMP, traceability, and reproducibility of batches. Through the integration of biotechnology and AI intelligence, CDMOs will be able to develop more sustainable, safe, and faster biologics production that will redefine the pharmaceutical fermentation processes.
Contract Pharmaceutical Fermentation Services Market Outlook
[[market_outlook]]
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 16.16 Billion |
| Market Size in 2026 | USD 17.46 Billion |
| Market Size by 2034 | USD 32.39 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 8.03% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Service Type, Scale/Capacity, Organism/Host System, Product Type, End-User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Contract Pharmaceutical Fermentation Services Market Segment Insights
[[segment_insights]]
Contract Pharmaceutical Fermentation Services Market Region Insights
[[regional_insights]]
Contract Pharmaceutical Fermentation Services Market Value Chain
[[value_chain]]
Contract Pharmaceutical Fermentation Services Market Companies
[[market_company]]
Other Contract Pharmaceutical Fermentation Services Market Companies
- Catalent, Inc.: Catalent provides advanced biologics and microbial fermentation services supported by end-to-end capabilities including process development, fill-finish, and regulatory support. The companys fermentation platforms are designed to ensure high yield, scalability, and compliance with global quality standards across therapeutic protein and enzyme production.
- Thermo Fisher Scientific (Patheon / Pharma Services): Thermo Fisher offers scalable microbial fermentation and biologics manufacturing solutions under its Patheon division. Its expertise spans from process development to commercial-scale production, supported by global regulatory compliance and analytical excellence to ensure reproducibility and quality in biopharmaceutical manufacturing.
- WuXi Biologics: WuXi Biologics operates a comprehensive biologics manufacturing network specializing in microbial and mammalian cell fermentation. The companys single-use bioreactor and continuous fermentation technologies support rapid, high-quality, and flexible biologics production, catering to global biopharma clients.
- Samsung Biologics: Samsung Biologics provides large-scale microbial and mammalian fermentation capabilities within its state-of-the-art facilities. The company focuses on speed, capacity, and global scalability, offering process development, clinical, and commercial biologics production services with full regulatory support.
- Boehringer Ingelheim (BioXcellence): Boehringer Ingelheim BioXcellence division offers contract manufacturing of biologics through advanced microbial and mammalian fermentation technologies. Its expertise covers the entire value chain, from strain development to large-scale production and fill-finish, emphasizing quality and regulatory compliance.
- Fujifilm Diosynth Biotechnologies: Fujifilm Diosynth Biotechnologies provides microbial fermentation and biologics contract manufacturing services for biopharmaceutical clients. The company global facilities are equipped for both batch and continuous fermentation, supporting vaccines, enzymes, and protein therapeutics production.
- Novasep: Novasep delivers integrated bioprocessing services combining microbial fermentation, purification, and downstream processing. The company strength lies in producing complex biomolecules, including enzymes and biopharmaceutical intermediates, through advanced chromatographic and separation technologies.
- AGC Biologics: AGC Biologics offers microbial fermentation services ranging from process development to GMP commercial production. The company facilities specialize in therapeutic proteins, plasmid DNA, and viral vectors, providing flexible and scalable solutions tailored to client requirements.
- Biocon Biologics: Biocon Biologics provides microbial fermentation-based manufacturing for biosimilars, enzymes, and biopharmaceutical intermediates. With strong expertise in process optimization and global regulatory compliance, Biocon supports cost-effective and large-scale bioproduction services.
- Rentschler Biopharma SE: Rentschler Biopharma delivers microbial and mammalian fermentation services focusing on therapeutic proteins and biopharmaceuticals. The company provides comprehensive CDMO solutions from cell line development to commercial manufacturing under GMP-compliant conditions.
- KBI Biopharma (Cytiva ecosystem): KBI Biopharma, now part of Cytiva, specializes in microbial fermentation and biopharmaceutical contract manufacturing. Its services include process development, analytical support, and clinical production, with a focus on precision, scalability, and quality assurance.
- Syngene International Ltd.: Syngene International provides microbial fermentation and biomanufacturing services as part of its integrated biologics contract research and manufacturing offerings. The company supports upstream and downstream process development for biologics and biosimilar production.
- Evonik Industries (BioProcess Division): Evoniks BioProcess Division provides contract fermentation services focused on producing amino acids, peptides, and biopolymers. The company combines microbial engineering expertise with large-scale industrial bioprocessing capabilities for both pharmaceutical and specialty applications.
- Merck KGaA (MilliporeSigma / BioReliance): Merck KGaA, through its BioReliance and MilliporeSigma divisions, provides end-to-end fermentation and biologics production services. Its capabilities include microbial strain development, bioprocess optimization, and analytical validation, serving the needs of global pharmaceutical and biotechnology clients.
Recent Developments
- In December 2024, Lonza decided to leave the capsules and health ingredients business to enhance its core Contract Development and Manufacturing Organization (CDMO) services. The relocation will enable it to increase its high-value biologics and advanced therapeutic modalities fermentation capacity.
- In December 2024, A joint venture of Zydus Lifescience and Perfect Day, named Sterling Biotech Limited, started building a precision fermentation-based dairy protein production plant in Bharuch, Gujarat. The new plant will be operational in Q1 2026, and it will also increase the biomanufacturing capacity of India to produce sustainable proteins.
- In October 2023, MycoTechnology introduced Fermentation-as-a-Service (FaaS) to assist startups and companies in accessing Indiefermentation infrastructure and expertise on a scale. The project assists firms in making bioproducts, including proteins, enzymes, and probiotics, to overcome the challenges of capacity and scale-up.
Contract Pharmaceutical Fermentation Services Market Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
