What is the Endotoxin Testing Market Size in 2026?
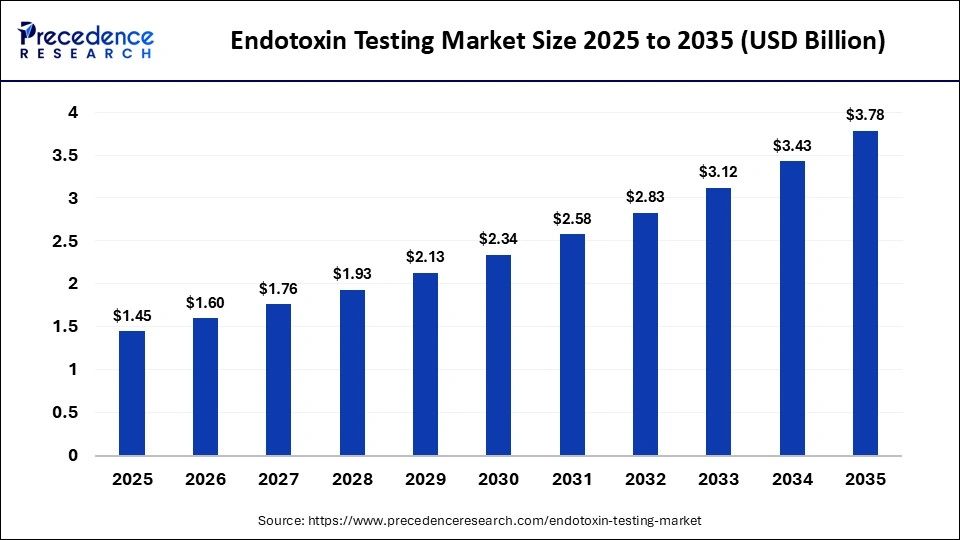
The global endotoxin testing market size accounted for USD 1.45 billion in 2025 and is predicted to increase from USD 1.60 billion in 2026 to approximately USD 3.78 billion by 2035, expanding at a CAGR of 10.05% from 2026 to 2035. The market is witnessing substantial growth due to increasing demand for safe pharmaceuticals, biologics, and medical devices, coupled with stringent regulatory standards for contamination control.
Key Takeaways
- North America dominated the market with a major market share in 2025.
- Asia Pacific is expected to grow at the fastest CAGR between 2026 and 2035.
- By testing method, the gel clot endotoxin test segment held a major market share in 2025.
- By testing method, the chromogenic endotoxin test segment is expected to expand at a notable CAGR from 2026 to 2035.
- By product type, the endotoxin detection products segment held the biggest market share in 2025.
- By product type, the PCR mycoplasma detection kits segment is expected to expand at the fastest CAGR between 2026 and 2035.
- By application, the pharmaceutical manufacturing segment accounted for the largest market share in 2025.
- By application, the medical device manufacturing segment is projected to grow at a solid CAGR between 2026 and 2035.
What is the Endotoxin Testing Market?
The global endotoxin testing market focuses on detecting and quantifying bacterial contaminants in pharmaceuticals, medical devices, and biologics. This is particularly crucial for ensuring sterility in injectables, implants, and vaccines to guarantee safety and comply with regulatory standards. The market is expanding due to the increasing prevalence of chronic diseases that require parenteral drugs, strict safety regulations for injectables, rising research and development efforts, and a shift towards animal-free testing methods, such as recombinant Factor C (rFC). These developments aim to reduce human error and provide faster turnaround times for high-volume testing.
How is AI Transforming the Endotoxin Testing Market?
Artificial Intelligence (AI) is transforming the endotoxin testing market by enhancing efficiency, accuracy, and compliance. It automates data analysis, shortens testing times, and improves pattern recognition to minimize human error. AI-powered systems enhance endotoxin detection by reducing false positives and ensuring adherence to stringent regulatory standards. Additionally, AI algorithms can predict potential issues and equipment failures, monitor production environments to prevent contamination, enhance data traceability, and automate audit trail reviews, all of which strengthen compliance with global regulatory standards and significantly improve laboratory throughput.
Major Trends in the Endotoxin Testing Market
- Shift to Animal-Free Testing: There is a rapid shift from horseshoe crab-derived Limulus Amebocyte Lysate (LAL) tests to rFC assays. This shift is largely driven by sustainability concerns, higher sensitivity, and, in some cases, lower costs associated with testing biological products.
- Automation and High-Throughput Technology: To enhance efficiency and accuracy, manufacturers are increasingly adopting automated robotic systems for sample handling, especially in pharmaceutical and medical device manufacturing.
- Stringent Regulatory Compliance: Regulatory bodies are intensifying their audits, requiring more rigorous testing for endotoxins in sterile injectables, vaccines, and medical devices. This has prompted investments in advanced, validated, and more precise testing methods.
- Rise of In-Process and Rapid Testing: There is a growing trend toward in-process testing, which aims to detect contamination earlier, thereby reducing waste. This method is increasingly accepted as a viable alternative to traditional animal tests.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.45 Billion |
| Market Size in 2026 | USD 1.60 Billion |
| Market Size by 2035 | USD 3.78 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 10.05% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Testing Method, Product Type, Application, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Testing Method Insights
What Made Gel Clot Endotoxin Test the Dominant Segment in the Endotoxin Testing Market?
The gel clot endotoxin test segment dominated the market with a major share in 2025. This dominance is attributed to its cost-effectiveness, simplicity, and established regulatory acceptance for routine qualitative analysis. The gel-clot technique is inexpensive and technically straightforward, making it highly appealing for routine testing applications. It is a validated, FDA-approved standard method that provides high confidence in safety assurance. This technique is extensively used for final product testing, raw material inspections, and specialized applications, including testing for 2-(18F) fluoro-2-deoxy-D-glucose (F18-FDG).
The chromogenic endotoxin test segment is projected to grow at the fastest rate during the forecast period. The growth of this segment is driven by its high sensitivity, suitability for high-throughput automation, and the ability to provide quantitative results in pharmaceutical quality control. This test offers precise quantitative data instead of just positive or negative results, which is crucial for strict pharmaceutical compliance. This significantly reduces turnaround times and labor costs for pharmaceutical manufacturers and increases regulatory pressure to adopt modern techniques to decrease reliance on LAL. Additionally, the expansion of biologics, vaccines, and injectable drug production is boosting the adoption of chromogenic assays for reliable endotoxin detection.
Type Insights
How Did the Endotoxin Detection Products Segment Lead the Endotoxin Testing Market?
The endotoxin detection products segment led the market in 2025. This is primarily due to their strong demand from the pharmaceutical and biotechnology industries for ensuring product safety and adhering to high-volume regulatory compliance. Additionally, the growing preference for animal-free, high-sensitivity testing, along with rapid advancements in biotechnology, biologics, and biosimilars, necessitates robust testing to prevent pyrogen contamination. The introduction of advanced detection methods, including rFC assays and chromogenic and turbidimetric LAL tests, has heightened sensitivity and accuracy in endotoxin detection. The growing production of biologics and injectable therapeutics has further amplified the need for reliable endotoxin detection kits and reagents, strengthening this segment's market dominance.
The PCR mycoplasma detection kits segment is expected to grow at the fastest CAGR over the forecast period. This is mainly due to their ideal sensitivity, rapid results, and strong compliance with FDA/EMA regulatory requirements for biologics. Developments in qPCR and digital PCR have enhanced quantitative accuracy and specificity. The increase in biopharmaceutical production and the growing necessity to test for mycoplasma in cell cultures are driving this segment's growth. Major regulatory bodies favor PCR-based methods to ensure the safety of biopharmaceuticals, vaccines, and cell therapies.
Application Insights
Why Did the Pharmaceutical Manufacturing Segment Dominate the Endotoxin Testing Market?
The pharmaceutical manufacturing segment dominated the market in 2025, driven by strict regulatory mandates requiring checks for safety in parenteral drugs, vaccines, and biologics. The surge in the production of vaccines, biopharmaceuticals, and recombinant DNA molecules necessitates extensive testing with LAL reagents to guarantee sterility. Given that parenteral and oral drugs must comply with stringent safety protocols, endotoxin testing is critical for quality control in manufacturing. The need for faster results is driving the adoption of advanced rapid testing devices in pharmaceutical production.
The medical device manufacturing segment is expected to experience the fastest growth in the forecast period, primarily due to escalating regulatory requirements for product sterility and a growing demand for biocompatibility testing. There is an increasing focus on managing bacterial endotoxin contamination in devices to prevent infections, particularly in intensive care units, which drives this demand. Advancements in rapid testing technologies enable more efficient, precise, and automated quality control, accelerating the testing process emphasized the need for stringent quality control in the medical product manufacturing sector.
Regional Insights
What Made North America the Dominant Region in the Endotoxin Testing Market?
North America dominated the endotoxin testing market while holding the largest share in 2025. The dominance of the region is attributed to stringent FDA regulations, robust biotechnology and pharmaceutical industries, and significant investment in drug development. The U.S. FDA enforces strict guidelines on the safety of pharmaceuticals and medical devices, for extensive endotoxin testing. The region is home to a large number of pharmaceutical, biotechnology, and contract research organizations, which drives demand for testing services. Additionally, there is a rapid adoption of advanced, high-end, and automated testing kits and systems, along with substantial investment in research and development, vaccine development, and complex biologics for quality control.
U.S. Endotoxin Testing Market Trends
The U.S. holds a dominant position in the market due to stringent FDA quality control standards. It is leading in the adoption of modern, non-animal-derived technologies like recombinant Factor C and recombinant Cascade Reagents to replace older horseshoe crab-based methods, aligning with the standards set by the U.S. Pharmacopeia. Major companies providing specialized testing services, such as Nelson Laboratories, are either based in or heavily active in the U.S., supporting pharmaceutical and medical device manufacturers.
Why is Asia Pacific Considered the Fastest-Growing Region in the Endotoxin Testing Market?
Asia Pacific is expected to experience the fastest growth during the forecast period, driven by the expansion of biopharmaceutical manufacturing, stringent quality standards, and high demand for medical devices. The increase in vaccine production and biologic therapies, particularly in China, India, and South Korea, necessitates rigorous, specialized endotoxin testing for raw materials and final product release. Also, the growing trend of outsourcing pharmaceutical manufacturing to CMOs accelerated the adoption of efficient, standardized endotoxin testing services. Transitioning from traditional methods to faster, automated, and more accurate technologies improves efficiency and reduces costs.
India Endotoxin Testing Market Trends
India is emerging as a significant player in the region, bolstered by increasing investments in biotechnology and pharmaceutical manufacturing. As a global pharmacy, India's substantial production of sterile injectables and biotech products demands high-volume, cost-effective endotoxin testing, making it a critical growth area. Indian manufacturers are also increasingly focusing on international regulatory compliance, raising the demand for advanced endotoxin detection methods.
How is the Opportunistic Rise of Europe in the Market?
Europe is experiencing opportunistic growth in the endotoxin testing market due to its strong pharmaceutical manufacturing base and expanding biologics production across countries such as Germany, France, and the UK. Stringent regulatory oversight by the European Medicines Agency and harmonized quality standards have increased the adoption of advanced endotoxin detection methods. Additionally, rising investments in research and development, along with growing contract manufacturing activities, are supporting market expansion in the region.
Endotoxin Testing Market Value Chain analysis

Endotoxin Testing Market Companies
- Charles River Laboratories International, Inc.
- Lonza Group Ltd.
- Merck KGaA
- Thermo Fisher Scientific Inc.
- bioMérieux SA
- Eurofins Scientific SE
- Associates of Cape Cod, Inc.
- FUJIFILM Wako Pure Chemical Corporation
- WuXi AppTec Co. Ltd.
- Sartorius AG
- Bio-Rad Laboratories, Inc.
- SGS Société Générale de Surveillance SA
- Nelson Laboratories, LLC
- GenScript Biotech Corporation
- Maravai LifeSciences Holdings, Inc.
Recent Developments
- In January 2026, BETMAT Biotechnology LLC launched High-Performance Sustainable Endotoxin Testing Services, offering animal-free, biosynthetic solutions using Recombinant Factor C (rFC) and Recombinant Cascade Reagent (rCR) technologies for next-generation quality control labs.
- In October 2024, LGM Pharma expanded its Analytical Testing Services at its Irvine, CA facility to include endotoxin and rapid sterility testing, offering faster turnaround, improved quality control, and cost savings for drug developers.(Source: https://lgmpharma.com)
- In January 2024, Charles River Laboratories launched the Endosafe Trillium rCR cartridge, combining its flagship endotoxin testing technology with a recombinant cascade reagent. This animal-free solution enhances bacterial endotoxin testing efficiency and aligns sustainability goals. The rCR cartridges become pre-loaded, reducing technician error and integrating seamlessly with existing Endosafe instrumentation.(Source: https://ir.criver.com)
Segments Covered in the Report
By Testing Method
- Gel Clot Endotoxin Test
- Chromogenic Endotoxin Test
- Turbidimetric Endotoxin Test
By Product Type
- Endotoxin Detection Products
- LAL Test Reagents
- Mycoplasma Detection and Removal
- PCR Mycoplasma Detection Kit
- Mycoplasma Elimination Cocktail
- Mycoplasma Detection and Elimination Custom Services
- Accessories
- Instrumentation
- Endotoxin Removal Products
- Low Endotoxin Recovery (LER) Products
- Lipopolysaccharides
By Application
- Medical Device Manufacturing
- Pharmaceutical Manufacturing
- Packaging Manufacture
- Raw Materials Production
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content







 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
