What is the Lentivirus Manufacturing for Advanced Therapies Market Size?
The global lentivirus manufacturing for advanced therapies market is growing as demand rises for viral vectors used in gene therapy, cell therapy, and cutting-edge regenerative treatments.The expansion of gene therapy pipelines primarily drives the lentivirus manufacturing for advanced therapies market.
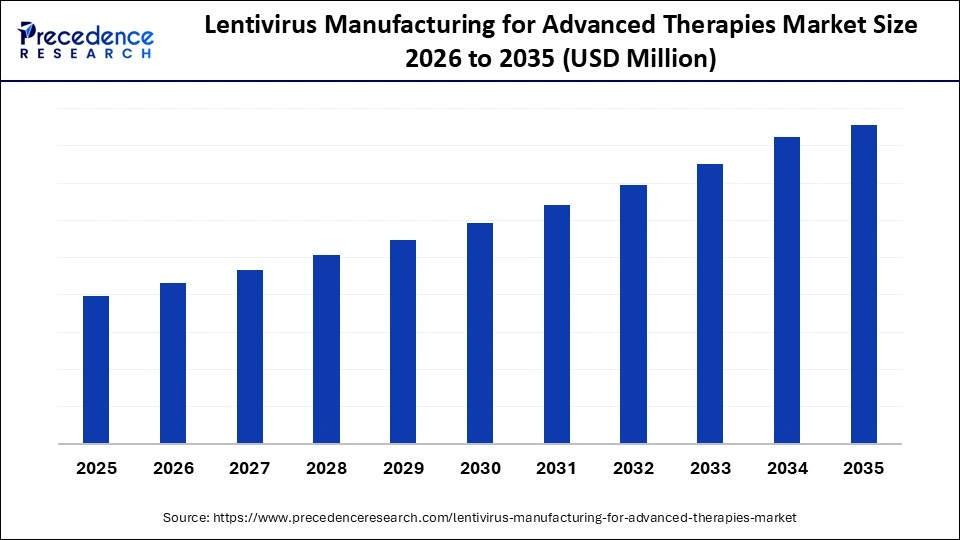
Market Highlights
- North America dominated the market, holding the largest market share of 42.40% in 2025.
- The Asia Pacific is expected to expand at the fastest CAGR of 13.5% between 2026 and 2035.
- By application, the gene therapy segment contributed the highest market share of 45.60% in 2025.
- By application, the cancer immunotherapy segment is projected to grow at a notable CAGR of 11.1% from 2026 to 2035.
- By end-user, the pharmaceutical & biotechnology companies segment captured the highest market share of 43.5% in 2025.
- By end-user, the research institutes & academic institutions segment is growing at a strong CAGR of 11.3% between 2026 and 2035.
- By scale of operation, the clinical / pilot-scale production segment held the major market share of 62.4% in 2025.
- By scale of operation, the commercial/large-scale production segment is set to grow at a healthy CAGR of 11.6% between 2026 and 2035.
Lentivirus Manufacturing for Advanced Therapies Market Overview
The lentivirus manufacturing for the advanced therapies market comprises the development and GMP-compliant production of lentiviral vectors used in gene and cell therapies . These vectors serve as essential delivery systems that transport therapeutic genes into target cells, resulting in stable, long-lasting expression. The market spans the full lifecycle of vector production. This begins with vector design, where developers optimize promoter selection, transgene configuration, and safety features to ensure high performance in clinical applications.
Upstream manufacturing includes cell culture, transfection, and vector generation using producer or packaging cell lines. Each step requires controlled bioreactor conditions, precise nutrient management, and validated transfection protocols to achieve high yield and consistent vector potency. Downstream processes follow with purification steps designed to remove impurities, host cell proteins , residual plasmid DNA, and other contaminants. Chromatography, filtration, and concentration methods are used to obtain clinical-grade material with defined purity and safety attributes.
The market also includes fill-finish operations, in which vectors are formulated, aliquoted, and packaged into vials under aseptic conditions. Quality control activities span the entire workflow and include assays for infectivity, transduction efficiency, sterility, identity, and genomic integration profiles. These processes ensure that vectors meet regulatory expectations for both clinical and commercial use.
How Are AI-Driven Innovations Reshaping the Lentivirus Manufacturing for Advanced Therapies Market?
In today's rapidly evolving technological landscape, the integration of artificial intelligence (AI) is a transformative force. It holds great potential to accelerate the growth of the lentivirus manufacturing for advanced therapies market by optimizing the entire workflow, from vector design to quality control. The integration of AI allows companies to make data-driven decisions. This leads to reducing CAR-T cell manufacturing timelines, lower costs, and greater patient access to advanced treatments. AI-integrated bioprocessing platforms enable real-time quality control, predictive analytics, and error detection. AI-based computational modeling is less expensive than traditional laboratory methods for optimizing an effective and safe gene delivery carrier.
Lentivirus Manufacturing for Advanced Therapies Market Outlook
[[market_outlook]]
Market Scope
| Report Coverage | Details |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Application, End-User, Scale of Operation, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Lentivirus Manufacturing for Advanced Therapies Market Segmental Insights
[[segment_insights]]
Lentivirus Manufacturing for Advanced Therapies Market Regional Insights
[[regional_insights]]
Key players in the Lentivirus Manufacturing for Advanced Therapies Market
- Thermo Fisher Scientific
- Oxford Biomedica
- Miltenyi Biotec
- Charles River Laboratories
- Catalent Inc.
- Fujifilm Diosynth Biotechnologies
- Cytiva
- Takara Bio Inc.
- Vigene Biosciences (part of Charles River)
- Bluebird Bio
- Cobra Biologics Limited
- Sirion Biotech GmbH
- Merck KGaA
- Sartorius AG
Recent Developments
- In June 2024, Charles River Laboratories International, Inc. and the Gates Institute at the University of Colorado Anschutz Medical Campus announced an agreement to establish a lentiviral vector contract development and manufacturing organization (CDMO). Gates Institute will leverage Charles River's premier cell and gene therapy CDMO expertise to develop Good Manufacturing Practice (GMP)-grade lentiviral vectors (LVVs) for use in novel chimeric antigen receptor (CAR) T-cell therapies for hematological cancers.(Source: https://www.nasdaq.com )
- In November 2025, AGC Biologics announced a new manufacturing agreement with the biotechnology company, marking AGC Biologics' latest advancement in the adeno-associated virus market. Under this agreement, AGC Biologics will provide Good Manufacturing Practice manufacturing for AAVantgarde's two novel candidates designed to address progressive and irreversible vision loss, where there are currently no approved therapies, such as AAVB-039 for Stargardt disease and AAVB-081 for retinitis pigmentosa (caused by Usher syndrome type 1B).(Source: https://www.businesswire.com )
Lentivirus Manufacturing for Advanced Therapies Market Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content


 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
