What is the Medical Batteries Market Size?
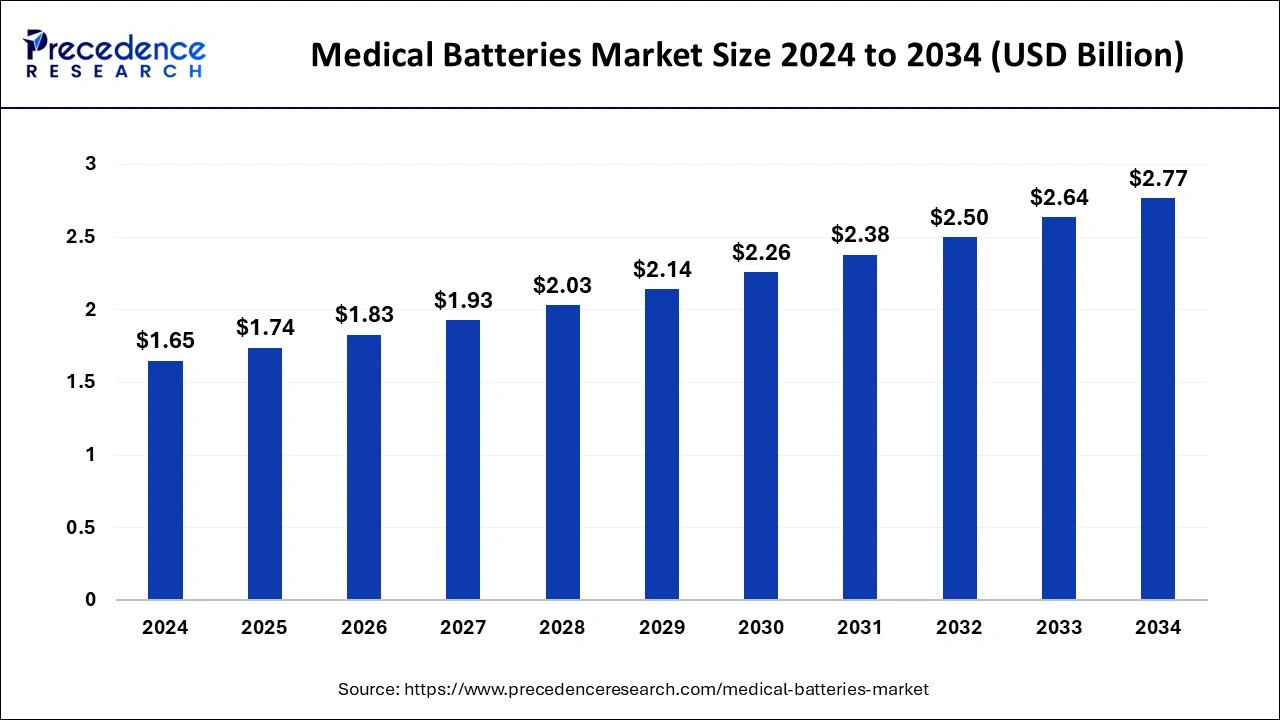
The global medical batteries market size is calculated at USD 1.74 billion in 2025 and is predicted to increase from USD 1.83 billion in 2026 to approximately USD 2.91 billion by 2035, expanding at a CAGR of 5.28% from 2026 to 2035.
Medical Batteries Market Key Takeaways
- In terms of revenue, the global medical batteries market was valued at USD 1.65 billion in 2025.
- It is projected to reach USD 2.91 billion by 2035.
- The market is expected to grow at a CAGR of 5.28% from 2026 to 2035
- North America dominated the global medical batteries market with a revenue share of 37% in 2025.
- Asia Pacific is projected to host the fastest-growing market over the forecast period.
- By battery type, the lithium-ion batteries segment dominated the market in 2025.
- By battery type, the nickel metal hydride batteries segment is expected to witness the fastest growth in the market over the forecast period.
- By application, in 2023, the implantable medical devices segment dominated the market.
- By application, the portable & wearable medical devices segment is expected to show significant growth during the studied period.
- By end-user, the hospitals & clinics dominated the market in 2025.
- By end-user, the ambulatory surgical center (ASCs) segment will show rapid growth over the projected period.
Market Overview
Medical batteries are designed to provide extended operational time and higher energy output for healthcare devices. These specialized power sources meet the specific requirements of medical applications, powering a wide range of devices from basic portable tools to complex, life-saving equipment. Unlike conventional batteries, medical batteries are engineered for reliability, safety, and longevity, which are essential for the proper functioning of critical healthcare instruments.
Medical Batteries Market Growth Factors
- The growing trend towards wearable healthcare technology is expected to fuel the growth of the medical batteries market.
- One significant advantage of medical batteries is their compact size, which enables seamless integration into devices like portable infusion pumps, blood glucose monitors, and electronic thermometers.
- Medical batteries are available in different forms, including disposable and rechargeable options, designed to meet the specific requirements of medical equipment and procedures.
- The rising utilization of remote patient monitoring systems can boost the medical batteries market growth during the forecast period.
- The increasing use of telemedicine, along with the growing requirement for portable, battery-powered diagnostic gadgets, can drive market growth shortly.
- Key players are constantly engaged in R&D activities, which will likely have a positive impact on market growth.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.74 Billion |
| Market Size in 2026 | USD 1.83 Billion |
| Market Size by 2035 | USD 2.91 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 5.28% |
| Largest Market | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Battery Type, Application, End-user, and Regions |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Driver
Advancement in medical device technology
The global rise in chronic diseases such as diabetes, cardiovascular diseases, and respiratory disorders requires continuous monitoring and management facilitated by battery-powered medical devices. Devices like glucose monitors, cardiac monitors, and nebulizers rely on batteries for portability and sustained operation. Additionally, the aging global population, which often requires ongoing healthcare and monitoring, further drives the demand for the medical batteries market.
Advancements in medical device technology, including the integration of advanced features, increased automation, connectivity, and miniaturization, aim to enhance the performance, precision, and efficiency of these devices. Hence, the medical batteries market is expected to expand rapidly in the coming years.
- In October 2023, Enovix Corporation, a silicon battery company, announced its standard IoT and Wearable-sized batteries have been chosen by Accurate Meditech for their Class II FDA-Approved “Mini” multi-vital sign monitor. The Accurate Mini continuously monitors blood pressure, pulse rate, and other vital signs.
Restraint
Battery failures
Balancing the demand for smaller batteries with the need for adequate energy storage is a delicate challenge that researchers and engineers continue to address. The risk of leakage or rupture remains a significant concern for medical batteries. Although technological advancements have reduced these risks, battery failures still occur. A battery leak in implanted devices can cause serious tissue damage and potentially endanger the patient's life. These factors are expected to constrain the growth of the medical batteries market in the coming years.
Opportunity
Government initiatives
Although ventilators typically run on electricity, battery backups ensure their continuous operation during power outages. Medical batteries are crucial for ventilators. Electric Fuel, a smart battery and charger company, was selected to supply rechargeable Li batteries to an Israeli company for automatic ventilators to support COVID-19 efforts. Furthermore, state initiatives to improve healthcare infrastructure in many countries are creating significant growth opportunities for the medical batteries market. Programs encouraging medical device start-ups are also providing substantial growth prospects for the medical batteries market.
- In July 2023, Resonetics entered into a strategic partnership with Resonant Link with the aim of integrating Resonetics' capabilities in rechargeable batteries and Resonant Link's innovative wireless charging technology to develop innovative rechargeable medical devices to improve patient outcomes.
Segment Insights
[[segment_insights]]
Regional Insights
[[regional_insights]]
Value Chain Analysis
- Raw Material Sourcing
Medical batteries mainly rely on high purity materials such as lithium metal, lithium cobalt oxide and nickel based chemistries. Its sourcing must meet strict medical regulatory standards, compelling suppliers to be highly specialized and certified. There is an increasing demand for safer, longer life chemistries.
Key Players: Umicore, 3M, Targray - Manufacturing Process
Manufacturing requires producing highly stable cells that can operate for years without any failure, especially with it comes to implantable categories. Its manufacturing process includes precise electrode coating, hermetic sealing, sterilization compatibility and long cycle validation. Manufacturers are seen adopting solid state designs, safer electrolytes and miniaturized cells.
Key Players: Saft, UltraLife Corporation, Renata - Quality Control and Testing
Medical batteries undergo rigorous compliance processes and testing in order to ensure efficiency, safety and reliability. Quality control includes electrical stability testing, leak proof casing validation, high temperature cycling and even vibration or shock checks. There is an increasing emphasis on increasing thermal stability, traceability and remote monitoring capabilities.
Medical Batteries Market Companies

- EaglePicher Technologies
- KAYO Battery Company Limited
- Ultralife Corp
- Panasonic
- STMicroelectronics N.V
- Tadiran Batteries Ltd.
- Arotech Corporation
- Saft Groupe S.A.
- Texas Instruments
- Shenzhen Kayo Battery Co
- Quallion LLC
- Vitec Group Plc
- Liberating Technologies
- Electrochem Solutions
- Maxim Integrated
- Others
Recent Developments
- In November 2025, Researchers at McGill University have come up with a new battery that's both stretchable and biodegradable, which are perfect for powering wearables and medical implants. It is made from gelatin mixed with magnesium and molybdenum electrodes, plus a splash of citric or lactic acid to boost voltage and lifespan. Unlike regular batteries that stick around as waste, this one breaks down in a phosphate-buffered saline solution within two months, which makes it a safer option for the environment and could help reduce e-waste from gadgets.
(Source: www.newsbytesapp.com ) - In April 2025, Abbott announced late-breaking data from the AVEIR™ Conduction System Pacing (CSP) acute clinical feasibility study, which demonstrated the safety and performance of the investigational AVEIR CSP leadless pacemaker technology. The AVEIR CSP acute clinical feasibility study is the world's first assessment of a leadless pacemaker delivering conduction pacing to the heart's left bundle branch (LBB) area. CSP is a novel pacing approach that targets the LBB area by enabling pacing that mimics the heart's natural electrical rhythm.
(Source: abbott.mediaroom.com ) - In August 2023, Ilika entered into a 10-year licensing agreement with Cirtec Medical to produce the Stereax range of mm-scale batteries at Cirtec's facility.
- In March 2022, Reliance New Energy Limited (Reliance) is a subsidiary of Reliance Industries Ltd. They acquired Lithium Werks for $61 million USD, along with manufacturing facilities in China and key business contacts.
- In October 2022, Turtle & Hughes, one of the nation's leading electrical and industrial distributors, today announced a strategic investment in Cadenza Innovation, a growth-stage company that has developed a patented technology platform – the ‘superCell' – which is enabling a new generation of safe, high-performing, low-cost lithium-ion (Li-ion) batteries and energy storage solutions.
Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content







 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
