What is mRNA Synthesis and Manufacturing Market Size in 2026?
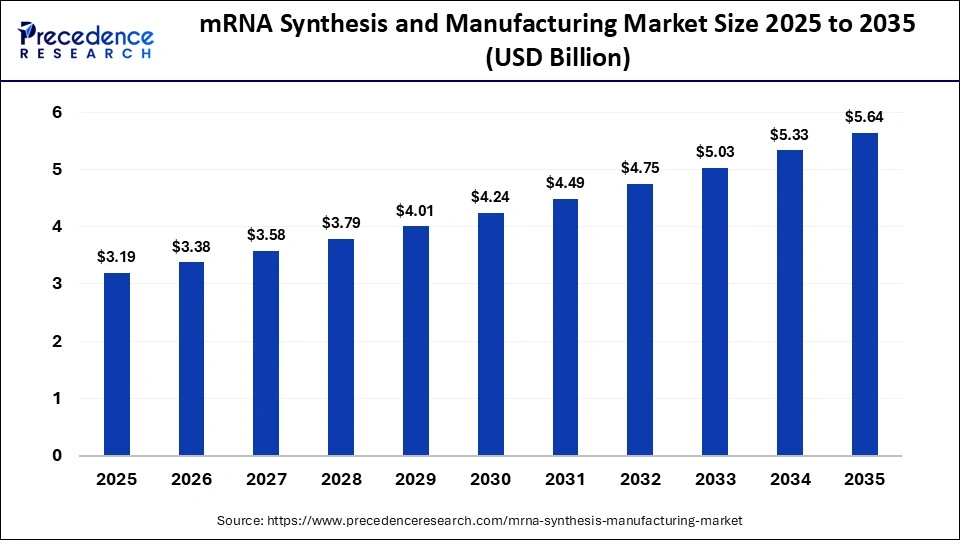
The global mRNA synthesis and manufacturing market size was calculated at USD 3.19 billion in 2025 and is predicted to increase from USD 3.38 billion in 2026 to approximately USD 5.64 billion by 2035, expanding at a CAGR of 5.85% from 2026 to 2035. The market is influenced by the increasing need to develop a vaccine faster and produce treatments tailored to the needs of a patient. Increasing investment in infectious disease prevention, oncology pipelines, and higher biologics production is driving the large-scale adoption of mRNA platforms.
Key Takeaways
- North America dominated the global mRNA synthesis and manufacturing market with a share of approximately 42.4% in 2025.
- Asia-Pacific is expected to grow at the fastest CAGR of approximately 7.8% in the market during the forecast period.
- By product type, the drug substance (APIs) segment held a dominant position in the market with a share of approximately 60% in 2025.
- By product type, the drug product (finished formulations) segment is expected to grow at the fastest CAGR of approximately 8.1% in the market between 2026 and 2035. 
- By service type, the GMP-grade manufacturing segment led the market with a share of approximately 46% in 2025
- By service type, the custom mRNA synthesis segment is expected to grow with the highest CAGR of approximately 9.1% in the market during the studied years.
- By application, the vaccine production segment dominated the mRNA synthesis and manufacturing market with a share of approximately 67% in 2025.
- By application, the therapeutics (oncology/rare disease) segment is expected to expand rapidly in the market with a CAGR of approximately 10.7% in the coming years.
- By scale of operation, the commercial scale segment held the largest market share of approximately 85% in 2025.
- By scale of operation, the preclinical/clinical scale segment is expected to show the fastest growth with a CAGR of approximately 3.4% over the forecast period.
- By end-user, the pharmaceutical and biotech companies segment contributed the biggest market share of approximately 51% in 2025.
- By end-user, the CROs and CDMOs segment is expected to gain the highest market share with a CAGR of approximately 6.8% between 2026 and 2035.
What is the mRNA Synthesis and Manufacturing Market?
The mRNA manufacturing and manufacturing industry has become an innovative cornerstone of a contemporary biopharmaceutical strategy. After the positive results of mRNA-based vaccines, pharmaceutical firms are moving into oncology, rare, and protein replacement therapies pipelines. Commercialization is being enhanced by scalable technology of in vitro transcription and lipid nanoparticle delivery systems. Biotech companies and contract development and manufacturing organizations (CDMOs) are also engaging in strategic alliances that are augmenting their production capacity globally.
What is the Role of AI in the mRNA Synthesis and Manufacturing Market?
Artificial intelligence (AI) plays a crucial role in mRNA design, sequence optimization, and process scalability. Codon optimization is quickened by machine learning (ML) models to increase protein expression and stability. AI-based predictive analytics enable manufacturers to detect potential errors and make effective decisions, aiding in high yield, purity, and quality control. Digital twins and automated bioreactor systems also make manufacturing efficiency easier, and the risk of batch failure lower. AI and ML can analyze vast datasets and help develop appropriate, personalized therapeutics based on patients' conditions.
mRNA Synthesis and Manufacturing Market Trends
- Technological Advancements: Manufacturing capabilities are changing because of the rapid increase in mRNA synthesis technologies. Stability, scalability, and product consistency are being improved by the improvement of in vitro transcription (IVT), lipid nanoparticle (LNP) delivery systems, and advanced purification processes.
- Partnering with CDMOs: Biopharmaceutical companies are turning to CDMOs to hasten the development and commercialization of mRNA products. CDMOs offer detailed infrastructure, regulatory capabilities, and scalable manufacturing.
- Increasing RNA Vaccine Requirement: The effectiveness of mRNA-based COVID-19 vaccines has further increased the desire to use mRNA vaccine platforms. Pharmaceutical firms are developing pipelines for infectious diseases, cancer, and rare genetic disorders.
- Demand for Manufacturing Efficiency: Manufacturers are focusing more on lowering costs, optimizing yield, and reducing turnaround time. The use of single-use bioreactors, modular facilities, and continuous manufacturing methods is enhancing the flexibility of operations.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 3.19 Billion |
| Market Size in 2026 | USD 3.38 Billion |
| Market Size by 2035 | USD 5.64 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 5.85% |
| Dominating Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, Service Type, Application, Scale of Operation, End-User, and region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segmental Insights
Product Type
Which Product Type Segment Dominated the mRNA Synthesis and Manufacturing Market?
The drug substance (APIs) segment held a dominant revenue share of approximately 60% in 2025, as core active pharmaceutical ingredients (APIs) are essential for the therapeutic effect. This segment is based on large-scale in vitro transcription and purification. High-yield, high-purity mRNA production is highly valuable to pharmaceutical companies for its effectiveness and safety. The need for bulk mRNA, mainly used in vaccines and pipeline therapeutics, has been increasing, and it remains a driving force behind dominance.
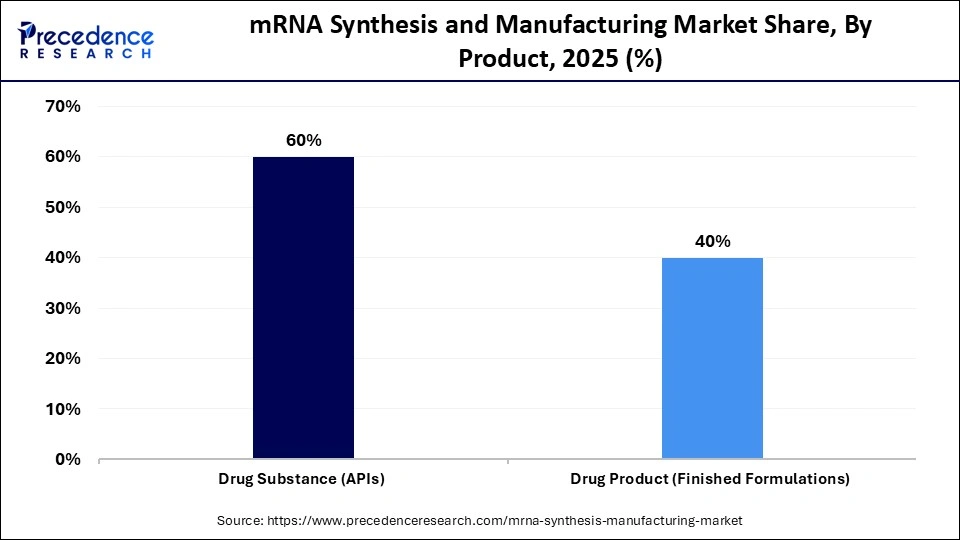
The drug product (finished formulations) segment is expected to show the fastest growth with a CAGR of approximately 8.1% over the forecast period because of increased investments in the lipid nanoparticle (LNP) encapsulation and fill-finish services. The demand in the field of final dosage formulation is increasing faster due to growth in personalized medicine and oncology pipelines. Overproduction of sterile and ready-to-deliver forms also promotes growth due to regulatory attention. The demand for finished drug products is soaring as more candidate drugs proceed to the later trials.
Service Type Insights
How the GMP-Grade Manufacturing Segment Dominated the Market?
The GMP-grade manufacturing segment dominated the mRNA synthesis and manufacturing market with a share of approximately 46% in 2025 because the production of clinical and commercial supplies is required to comply with regulations. There is consistency and traceability in the form of stringent quality standards and verified production facilities. Scalability and approval of global distribution are examples of advantages of large pharmaceutical companies that depend on the GMP certification. The growing commercial mRNA vaccine manufacturing further promotes the segment's growth.
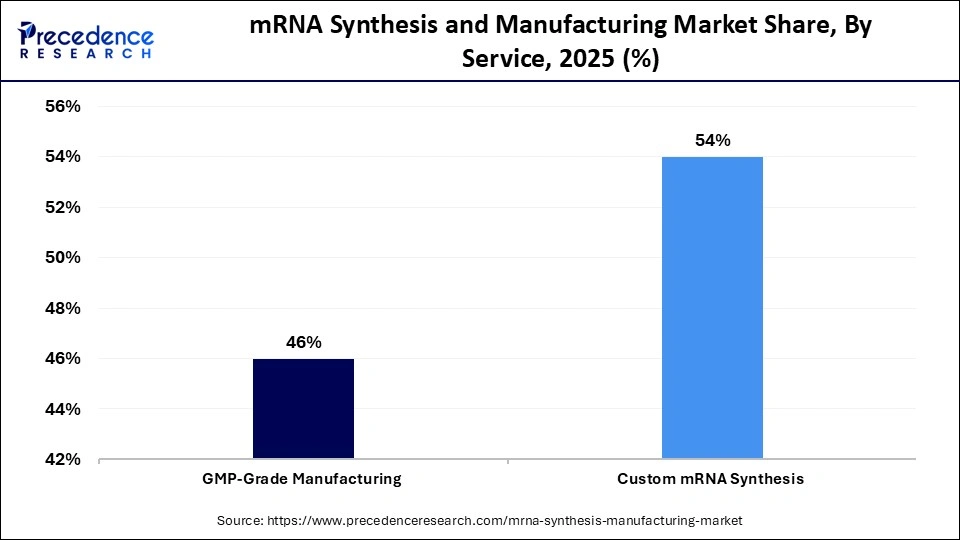
The custom mRNA synthesis segment is expected to gain the highest market share with a CAGR of approximately 9.1% between 2026 and 2035 because of the rising need to customize a sequence to use in research and therapeutic development. Academic institutes and new biotech companies need a niche to use specialized constructs. Flexibility is increased by the development of rapid prototyping and small-volume synthesis. Individualized immunization in cancer and uncommon disease therapeutics are significant growth drivers.
Application Insights
How the Vaccine Production Segment Led the mRNA Synthesis and Manufacturing Market?
The vaccine production segment contributed the biggest revenue share of approximately 67% in 2025, due to global preventive measures against infectious diseases. Scalability in platforms and the ability to respond quickly in a short time were proven by the success of mRNA-based COVID-19 vaccines. Governments are still investing in pandemic preparedness and stockpiling. Growing research on influenza, RSV, and the next generation of vaccines potentiates the need for mRNA vaccines.
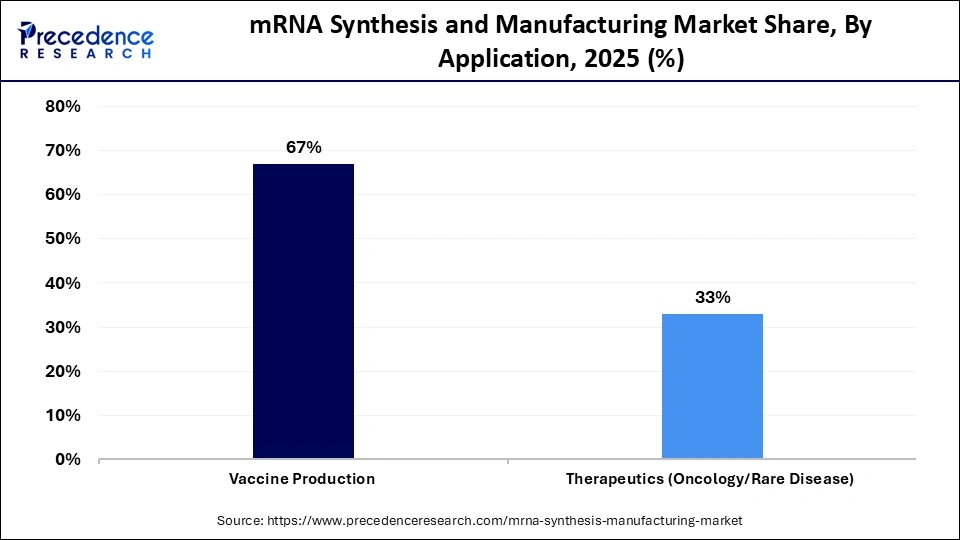
The therapeutics (oncology/rare disease) segment is expected to witness the fastest growth in the market with a CAGR of approximately 10.7% over the forecast period, driven by oncology or rare genetic diseases, and mRNA-based protein replacement therapies and cancer immunotherapies are in clinical pipelines. The platform is flexible, which allows for identifying the target quickly and optimizing the design. It is believed that the growth of this segment will be greatly boosted by the increasing activity in the clinical trials.
Scale of Operation Insights
What Made Commercial Scale the Dominant Segment in the Market?
The commercial scale segment registered its dominance over the global market with a share of approximately 85% in 2025, driven by the growing use of mRNA diagnostics and therapeutics for clinical applications. New product approvals and rapidly evolving regulatory landscapes enhance the market availability of mRNA products for commercial use. Overall, the data reflects the segment's considerable influence and growth. Advancements in manufacturing technologies enhance efficiency, accuracy, and reproducibility, augmenting the segment's growth.
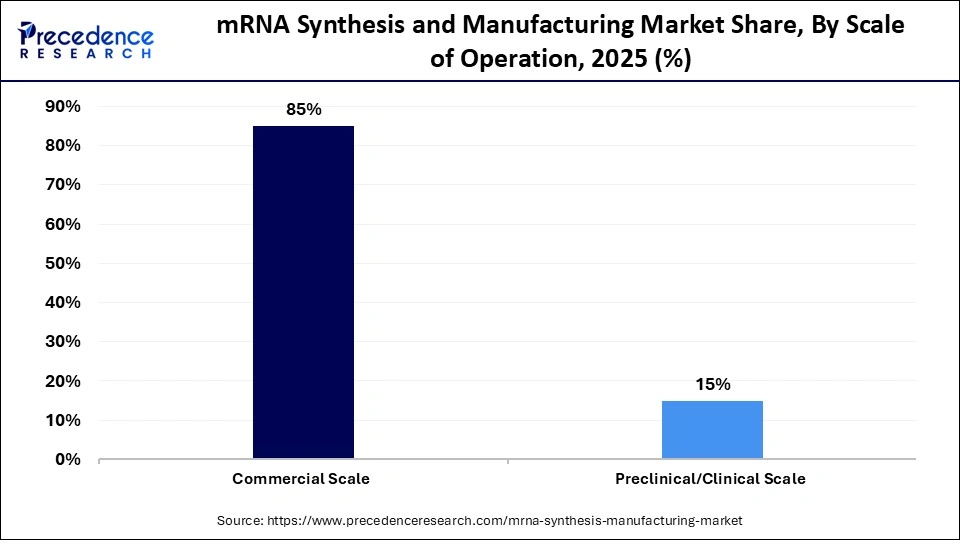
The preclinical/clinical scale segment is expected to expand rapidly in the market with a CAGR of approximately 3.4% in the coming years, due to many mRNA candidates being developed early. Phase I and II trials are being supported with smaller batch production facilities being constructed. Platforms of flexible modular manufacturing increase the flexibility of pipeline diversification. The fast development of biotech startups is one of the primary contributors to this segment.
End-User Insights
Which End-User Segment Led the mRNA Synthesis and Manufacturing Market?
The pharmaceutical and biotech companies segment led the market with a share of approximately 51% in 2025, driven by high R&D expenditures and its own mRNA pipelines. Such companies have progressive research facilities and control knowledge. There are also strategic alliances and licensing, which increase production capacity. Their market leadership is maintained with large-scale commercialization.
The CROs and CDMOs segment is expected to grow with the highest CAGR of approximately 6.8% in the market during the studied years, driven by increasing trends of outsourcing of small and mid-sized biotech companies. CDMOs offer GMP facilities, which are scalable and have technical expertise. The active movement of clinical trials has a strong impact on the growth trend of this segment.
Regional Insights
North America mRNA Synthesis and Manufacturing Market Size and Growth 2026 to 2035
The North America mRNA synthesis and manufacturing market size is estimated at USD 1.35 billion in 2025 and is projected to reach approximately USD 2.40 billion by 2035, with a 5.92% CAGR from 2026 to 2035.
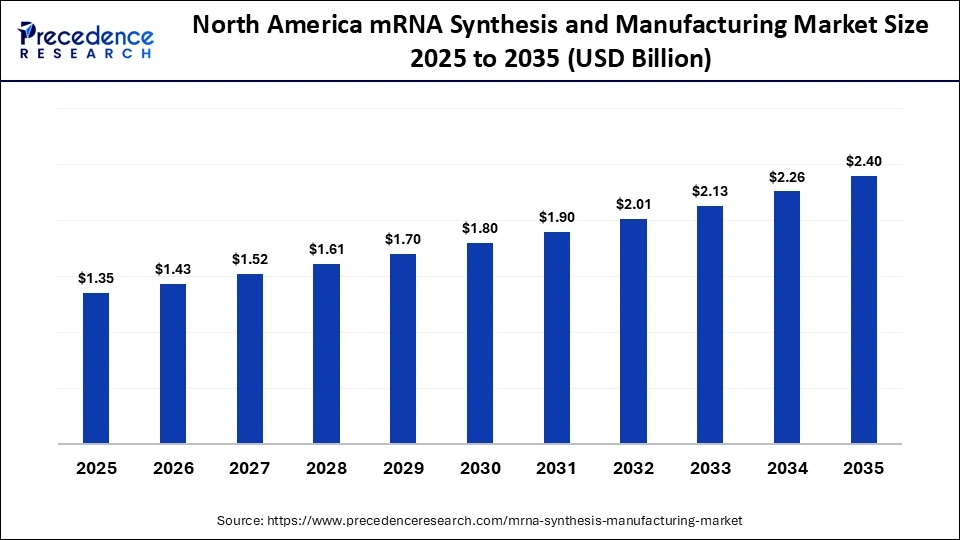
Why North America Dominated the mRNA Synthesis and Manufacturing Market?
North America held a major revenue share of approximately 42.4% in the market in 2025, because of well-developed biotechnology and early adoption of platforms. The region has been enjoying great research and development expenditure and strong intellectual property systems. The domestic production capacity is enhanced by government funding for the development of vaccines and biosecurity preparedness.
Enhanced competitiveness is also promoted by the higher level of manufacturing technologies and digital bioprocessing systems. Large pharmaceutical companies create strategic alliances with biotech inventors to speed up commercialization. The existence of well-established CDMOs and integrated supply chains guarantees efficiency in production and distribution.
U.S. mRNA Synthesis and Manufacturing Market Size and Growth 2026 to 2035
The U.S. mRNA synthesis and manufacturing market size is calculated at USD 1.01 billion in 2025 and is expected to reach nearly USD 1.81 billion in 2035, accelerating at a strong CAGR of 6.01% between 2026 to 2035.
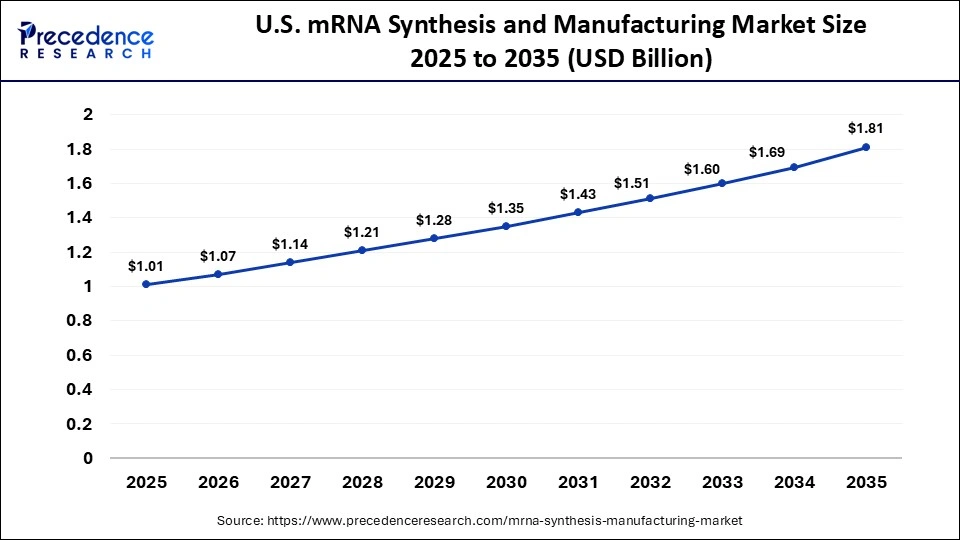
Country-Level Analysis
The U.S. has the greatest investment in the mRNA vaccine and therapeutic pipelines in the region. Large biotech hubs facilitate innovation, cooperation, and high-speed clinical development. Canada is increasing home mRNA manufacturing capacity to increase healthcare resilience. Federal programs promote the development of biomanufacturing and personnel.
Universities and other academic research institutions make a significant contribution towards early-stage innovation. The combination of these makes North America have a strong leadership in the world.
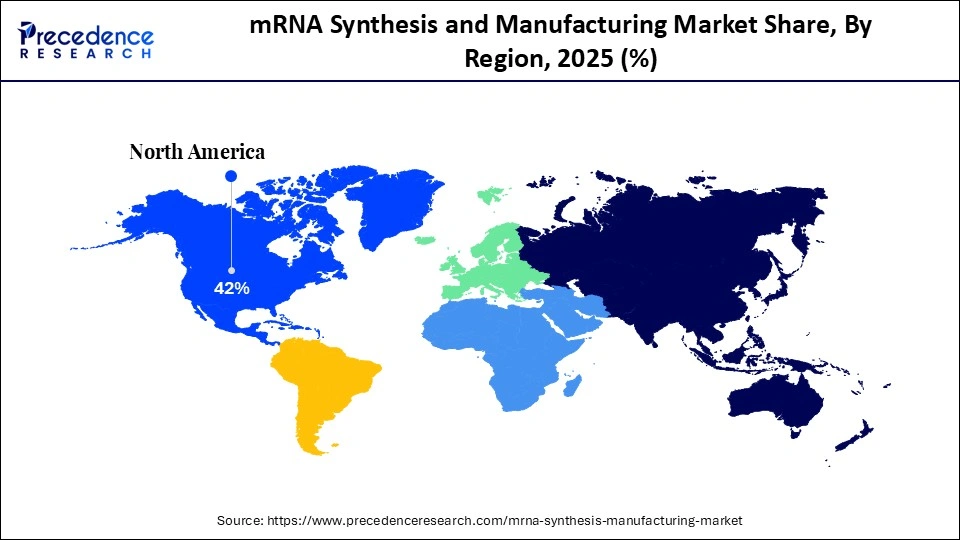
Why is Asia-Pacific the Fastest-Growing Region in the Market?
Asia-Pacific is expected to host the fastest-growing mRNA synthesis and manufacturing market with a CAGR of approximately 7.8% in the coming years, due to the increasing pharmaceutical production capacity and rising healthcare spending. Governments are also focusing on producing mRNA domestically to minimize the use of imports. Growing clinical trial operations and biotechnology investments facilitate market growth.
Facility development is being accelerated through infrastructure upgrades and incentive policies. The industry has potential in the market due to increasing demand for vaccines and new forms of treatment. The local partnerships are increasing the transfer of technology and the scalability of production.
Country-Level Analysis
China is making a heavy investment in mRNA R&D and large-scale production plants. Japan is promoting mRNA-based therapy research in the case of social-corporate associations. South Korea is increasing its capacity in CDMO to cater to international customers. India is reinforcing the production capacity and regulatory systems of vaccines. Australia has become a research centre in the mRNA platform technologies. All these country-specific projects make the Asia Pacific an epicentre of high growth.
Will Europe Grow in the mRNA Synthesis and Manufacturing Market?
Europe is expected to witness notable growth in the market due to strong regulatory support for advanced biologics and vaccine innovation. The region's emphasis on healthcare sovereignty and localized biomanufacturing capacity has accelerated infrastructure investments. Strategic public–private partnerships are strengthening research translation from academic institutions to commercial production.
Expansion of GMP-certified facilities and cold-chain logistics capabilities is enhancing supply reliability. The growing oncology and rare disease therapeutic pipelines are further stimulating demand for mRNA platforms. Additionally, sustainability-driven pharmaceutical manufacturing initiatives are encouraging modernization of production technologies across the region.
Country-Level Analysis
At the country level, Germany leads growth with robust biotech clusters and advanced bioprocess engineering expertise. France is expanding state-supported vaccine innovation programs and biomanufacturing hubs. Belgium plays a critical role as a European life sciences gateway with a strong CDMO presence.
The U.K. is advancing mRNA research through strategic academic–industry collaborations and regulatory agility. Italy and Spain are strengthening domestic pharmaceutical manufacturing to enhance regional resilience. Collectively, these country-specific advancements position Europe as a strategically expanding mRNA manufacturing powerhouse.
mRNA Synthesis and Manufacturing Market Value Chain Analysis

Top Companies in the mRNA Synthesis and Manufacturing Market
- Thermo Fisher Scientific Inc.
- Danaher Corporation (Aldevron/IDT)
- Merck KGaA
- Lonza Group AG
- Maravai LifeSciences (TriLink BioTechnologies)
- BioNTech SE
- Moderna, Inc.
- CureVac SE
- GenScript Biotech Corporation
- Samsung Biologics
- WuXi Biologics
- New England Biolabs
- Sartorius AG
- ST Pharm Co., Ltd.
Recent Developments in the mRNA Synthesis and Manufacturing Market
- In November 2025, Moderna announced that it had invested more than $140 million to increase the production of drug products in its Norwood, Massachusetts, plant. The growth enhances its wholly owned domestic mRNA manufacturing network to facilitate a smooth end-to-end provision to support both clinical development programs and large-scale commercial provision of its expanding therapeutic platform.
(source: https://in.investing.com) - In October 2025, Medicines Patient Pool and the World Health Organization launched Phase 2.0 of their global mRNA Technology Transfer Program to increase the capacity to produce sustainably in low and middle-income countries. The program is planned to be implemented between 2026-2030 and make the regions more self-reliant and be able to produce up to 1.9 billion doses of resistance to the pandemic.
- In May 2025, GenScript launched an advanced GMP-like mRNA manufacturing solution tailored for phase-specific drug development, enhancing its end-to-end synthesis and formulation capabilities. The initiative strengthens support for biopharmaceutical research and early-stage production, enabling academic institutions and biotech companies to streamline development timelines and accelerate progression of mRNA therapeutic pipelines.
(source: https://www.fiercebiotech.com)
Segments Covered in the Report
By Product
- Drug Substance (APIs)
- Drug Product (Finished Formulations)
By Service
- GMP-Grade Manufacturing
- Custom mRNA Synthesis
By Application
- Vaccine Production
- Therapeutics (Oncology/Rare Disease)
By Scale of Operation
- Commercial Scale
- Preclinical/Clinical Scale
By End-User
- Pharmaceutical and Biotech Companies
- CROs and CDMOs
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
