What is Next Generation Vector Engineering Market Size?
The global next generation vector engineering market is expanding rapidly as advances in gene therapy and synthetic biology drive demand for innovative vector design solutions. Market growth is driven by the rising prevalence of genetic disorders and the increasing adoption of viral and non-viral vector-based gene therapies for targeted, personalized treatments.
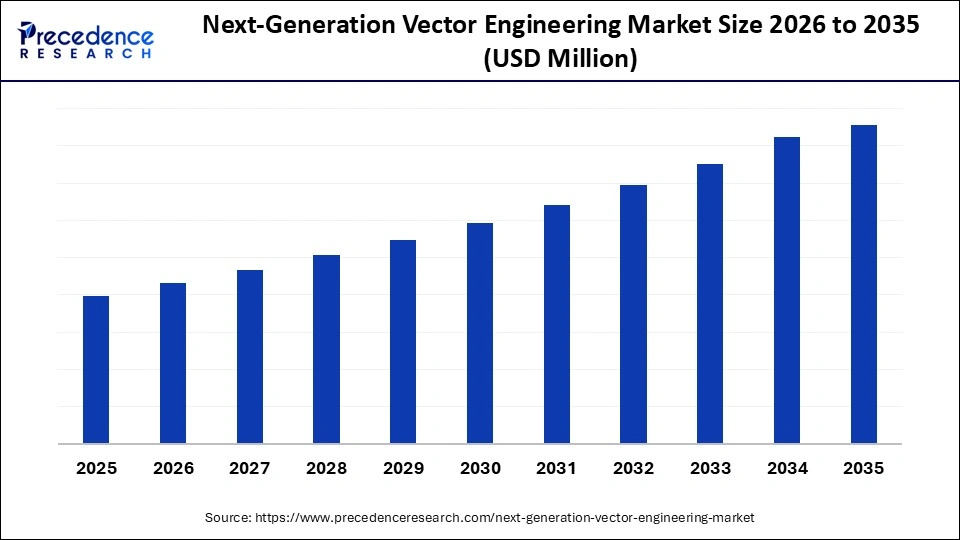
Market Highlights
- North America accounted for the largest market share of 47% in 2025.
- The Asia Pacific segment is expected to grow at the fastest CAGR of 16.5% from 2026 to 2035.
- By vector type, the viral vectors segment contributed for a considerable 56% share in 2025.
- By vector, the synthetic & engineered vectors segment is projected to experience the highest CAGR of 18.5% between 2026 and 2035.
- By technology approach, the capsid engineering segment led the market with a 30% market share in 2025.
- By technology approach, the AI/ML-guided optimization systems segment is projected to grow at a strong CAGR of 19.8% from 2026 to 2035.
- By application, the gene therapy segment dominated the market, accounting for 41% of the market in 2025.
- By application, the cell therapy segment is expected to grow at the highest CAGR from 2026 to 2035.
- By workflow stage, the preclinical development segment contributed the highest market shar of 38% in 2025.
- By workflow stage, the discovery & design segment is projected to expand rapidly with an 18% CAGR from 2026 to 2035.
- By end user, the biopharmaceutical companies segment maintained a leading position in the market with a 48% share in 2025.
- By end user, the CDMOs segment is growing at a notable CAGR of 16.2% from 2026 to 2035.
- By delivery route, the Ex vivo segment captured a significant market share of 52% in 2025.
- By delivery route, the In vivo delivery segment will grow at a 17.8% CAGR from 2026 to 2035.
Market Overview of Next-Gen Vector Engineering: Therapeutic Applications, Design Tools, and Production Needs
Rapid growth in demand for treatments of rare genetic disorders and cancers drives expansion in this vector based therapy sector. It involves the use of engineered viral or non-viral vectors, such as AAV, lentivirus, or plasmid-based vectors, that transfer therapeutic genes into patient cells to correct, replace, or regulate malfunctioning genes. By the beginning of March 2023, the international database had registered approximately 3,900 clinical trials in gene therapy in 46 different countries.
Viral vector methodologies are highly popular for their efficient delivery. Rapid regulatory approvals and increasing trial volume highlight the growing ecosystem for complex vector-based therapeutics. Furthermore, the growing number of at-risk populations due to genetic and rare diseases is expected to drive widespread use of these technologies in the coming years.
Impact of Artificial Intelligence on the Next-Generation Vector Engineering Market
Artificial intelligence (AI) plays a significant role in improving the design, optimization, and manufacturing in the next-generation vector engineering market. Leading biotechnology companies are using AI-based algorithms to process complex genomic , proteomic , and transcriptomic datasets, allowing them to identify optimal vector configurations with far greater speed and accuracy than traditional experimental methods. AI-driven sequence engineering supports the discovery of improved capsid variants, enhances tissue tropism, increases transduction efficiency, and reduces immunogenic risk by predicting how the immune system may respond to specific vector designs.
Furthermore, AI tools are increasingly used to model vector host interactions at the molecular level, enabling researchers to anticipate off-target effects and refine payload stability before entering preclinical studies. In manufacturing, AI supports real-time process optimization, yield prediction, and quality control monitoring, which improves batch consistency for viral and non-viral vectors. AI-enabled predictive modeling of clinical outcomes also guides patient stratification, dosing strategies, and trial design, helping companies reduce clinical failure rates and accelerate the development of next-generation gene therapies.
Next-Generation Vector Engineering Market Growth Factors
- Rising Adoption of Personalized Medicine: Growing demand for patient-specific gene therapies is propelling the development of tailored vector platforms.
- Advancements in CRISPR and Gene-Editing Technologies: Breakthroughs in precise genome editing are driving innovation in vector design and delivery efficiency.
- Expanding Clinical Trial Pipeline: An Increasing number of ongoing trials for rare and complex diseases is boosting the need for scalable vector production.
- Growing Biopharma-CDMO Collaborations: Strategic partnerships between biotech firms and contract manufacturing organizations are fuelling access to advanced vector engineering expertise.
- Enhanced Regulatory Support for Gene Therapies: Evolving frameworks in the U.S., EU, and Asia are propelling faster approval and commercialization of vector-based therapies.
Market Scope
| Report Coverage | Details |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Vector Type, Technology Approach, Application, Workflow Stage, End User, Delivery Route, and by Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Global Metrics, Innovation Drivers, and Competitive Landscape in Next-Generation Vector Platforms
- Japan continues to scale high-volume bioreactor capacity suitable for vector production, spurred by AGC Biologics' Yokohama site, which is being outfitted with two 5,000-L single-use bioreactors, making it one of the largest single-site mammalian/vector-capable facilities in the Asia-Pacific region.
- South Korea is positioning itself as a large-capacity biologics and vector-supporting manufacturer, with Samsung Biologics adding over 180,000 L of biologics capacity at Bio Campus II. Although primarily for proteins, this scale enables rapid conversion toward large-scale viral-vector support for global gene-therapy supply chains.
- The U.K. continues to lead Europe, hosting more than 5,000 active clinical trials , boosted by the NHS's integrated data systems and the MHRA's recent rapid-review framework. This regional growth directly contributes to faster Phase II/III progressions and improved cross-border recruitment efficiency.
- In 2025, Azalea Therapeutics secured USD 82 million to build dual-vector and in vivo editing platforms. This level of early-stage capital signals accelerating investor confidence in next-gen vector platforms that can bypass current delivery limitations.
- Atsena Therapeutics closed a USD 150 million round backed by Bain and Sofinnova for AAV-based retinal vector engineering. Large late-stage funding rounds strengthen vector optimization pipelines and push more programs toward commercial-scale manufacturing.
- Fuse Vectors secured ~5 M (USD $5.2 M) for its next-gen, cell-free AAV platform. Early funding for manufacturing-focused startups shows growing global demand for scalable, low-contamination vector production technologies.
- In 2025, over 115 new patents for AAV vector technologies were published, with ~15% already granted according to recent industry reporting. This demonstrates that innovations in vector design and delivery continue at a rapid pace. Continuous patent grants encourage companies to invest in next-gen vector R&D, locking in IP protection for novel capsids and delivery methods.
- Shift toward non-viral and hybrid delivery technologies is underway, as evidenced by PROGEN launching lipid nanoparticle (LNP) kits in 2025, expanding beyond AAV analytics to offer customizable LNP-based delivery systems. Emergence of non-viral delivery options diversifies the vector engineering landscape, driving innovation in both viral and non-viral platforms.
Next Generation Vector Engineering Market Segment Insights
[[segment_insights]]
Next Generation Vector Engineering Market Regional Insights
[[regional_insights]]
Next-Generation Vector Engineering Market Value Chain
[[value_chain]]
Next-Generation Vector Engineering Market Companies
[[market_company]]
Other Next-Generation Vector Engineering Market Companies
- Charles River Laboratories
- Fujifilm Diosynth Biotechnologies
- AGC Biologics
- Sartorius Stedim Biotech
- Merck KGaA (MilliporeSigma)
- Takara Bio
- Vigene Biosciences
- Vector BioPharma
- Cellares
- Aldevron
Recent Developments
- In October 2025, Asimov launched its AAV Edge Stable Producer system, aiming to overcome limitations of traditional transient-transfection workflows and raise AAV manufacturing reliability to standards closer to antibody-based biologics. Alec Nielsen, PhD, co-founder and CEO, stated that the platform is expected to enhance stability, reproducibility, and scalability for next-generation vector production. (Source: https://www.genengnews.com )
- In July 2025, Lonza announced the launch of the 4D-Nucleofector LV Unit PRO, its next-generation large-scale electroporation system engineered for delivering clinically relevant cargos into high-volume T-cell batches. The new unit expands Lonza's non-viral transfection portfolio and is optimized for CRISPR genome-engineering programs and advanced cell-therapy manufacturing applications. (Source: https://www.lonza.com )
- In April 2025, CellFE announced a strategic collaboration with the Harbottle Lab at the German Cancer Research Center (DKFZ) and its spin-out TcellTech. The partnership integrates CellFE's Ryva Mechanoporation System with TcellTech's nanoSMAR gene-expression DNA vector platform to enhance the performance of non-viral DNA vectors. The collaboration is expected to accelerate personalized cell-therapy development by addressing key challenges in delivery and expression. (Source: https://www.cellfebiotech.com )
Next-Generation Vector Engineering MarketSegments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content
.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
