Why is the Preclinical and Clinical Trials Market Expanding Rapidly?
The global preclinical and clinical trials market is expanding as pharmaceutical and biotech companies accelerate drug development and invest in advanced testing solutions.The market is expanding rapidly due to the impending threat of antibiotic-resistant pathogens, a rapid shift towards outsourcing, the active presence of biotechnology and pharmaceutical companies for R&D to develop new drugs, and supportive government regulations.
Market Highlights
- North America held the largest market share of 38% in 2025.
- The Asia Pacific is projected to grow at a solid CAGR from 2026 to 2035.
- By phase/study type, the preclinical studies segment held the largest market share of 30% in 2025.
- By phase/study type, the Phase II clinical trials segment is expected to grow at a notable CAGR from 2026 to 2035.
- By therapeutic area, the oncology segment contributed the biggest market share of 40% in 2025.
- By therapeutic area, the rare/orphan disease is expanding at a strong CAGR from 2026 to 2035.
- By technology/mode of action, the electronic data capture platforms segment captured the highest market share of 25% in 2025.
- By technology/mode of action, the AI & machine learning in trials segment is growing at a CAGR of 30% from 2026 to 2035.
- By end-user, the pharmaceutical & biotechnology companies held the largest market share of 45% in 2025.
- By end-user, the contract research organization segment is poised to grow at a CAGR of 30% from 2026 to 2035.
Evolving Landscape of Preclinical and Clinical Trials
The preclinical and clinical trials market refers to the global industry that supports every stage of drug development, beginning with laboratory-based discovery studies and extending through phased human trials. The market includes a wide range of services and technologies used to evaluate safety, efficacy, dosing, and long-term performance of new therapeutics. Preclinical testing covers both in vitro and in vivo studies that examine toxicity, pharmacokinetics, target engagement, and disease model response. These studies generate the data packages required for regulatory submissions such as Investigational New Drug applications in the United States and Clinical Trial Applications in the European Union.
Clinical development spans Phase I to Phase IV trials. Phase I studies assess safety and tolerability in small groups of volunteers, while Phase II trials evaluate therapeutic activity and early efficacy signals. Large-scale Phase III trials confirm safety and efficacy in broader patient populations, and Phase IV post-marketing surveillance monitors long-term outcomes, rare adverse events, and real-world effectiveness.
The market also includes patient recruitment services, site management, trial monitoring, pharmacovigilance programs, and regulatory consulting to support compliance with agencies such as the FDA, EMA, and national drug authorities. Advanced digital platforms are now essential to this market. Electronic data capture systems, remote monitoring tools, electronic patient-reported outcome systems, and clinical trial management platforms support real-time data collection and centralized oversight.
AI Shifts in Preclinical and Clinical Trials Market
The integration of AI with the preclinical and clinical trials market is transforming the market on a large scale by speeding up drug discovery , making trials highly efficient, and focusing on developing more patient-centric solutions. Experts predict that the AI healthcare market will witness a huge growth of nearly 40% annually, along with clinical trials and AI integration.
A key benefit of AI in clinical trials includes accelerating drug development, reducing clinical trial costs, enhancing data quality, developing personalized treatments, and offering real-time expertise for smart trials. AI can handle time-consuming tasks like finding trial participants, safety data tracking and management of workflow which allows researchers to focus more on identifying new compounds quickly and planning maximum trial design within a shorter time frame.
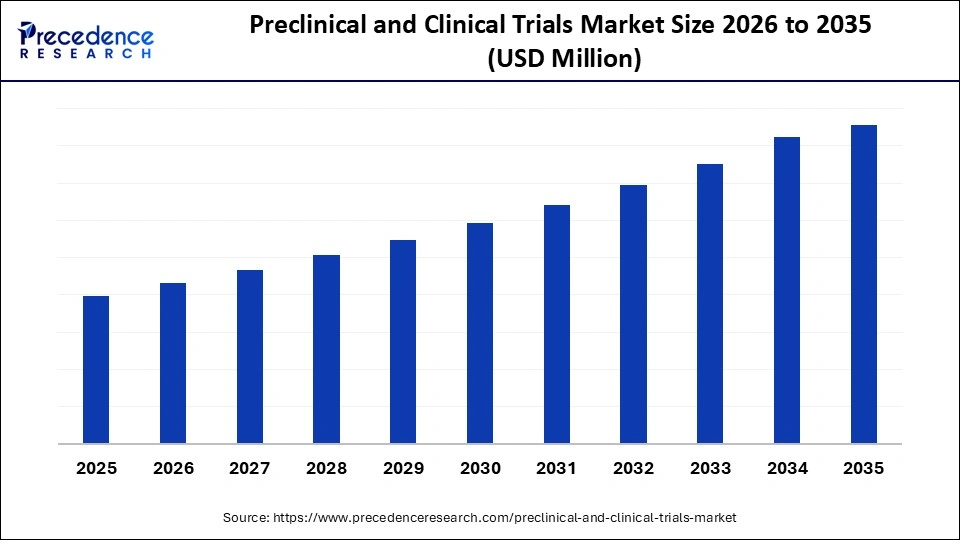
Preclinical and Clinical Trials Market Outlook
[[market_outlook]]
Market Scope
| Report Coverage | Details |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Phase/Study Type, Therapeutic Area, Technology/Mode of Action, End-User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Preclinical and Clinical Trials Market Segmental Insights
[[segment_insights]]
Preclinical and Clinical Trials MarketRegional Insights
[[regional_insights]]
Preclinical and Clinical Trials Market Value Chain

Top Companies in Preclinical and Clinical Trials Market & their Offerings
- IQVIA
- LabCorp Drug Development (Covance)
- Charles River Laboratories
- ICON plc
- Parexel International
- PPD, Inc. (Thermo Fisher)
- Syneos Health
- Medpace
- WuXi AppTec
- PRA Health Sciences
- Pharmaceutical Product Development, LLC
- SGS Life Sciences
- Eurofins Scientific
- Pharmaron
- Celerion
- Envigo
- BioReliance (Merck KGaA)
- KCR Group
- Frontage Laboratories
- Medpace Holdings
Recent Developments
- In April 2025, Atsena Therapeutics, a leading pharmaceutical company, well known for its working on gene therapies for inherited retinal diseases, secured a $150 million oversubscribed series of C financing. This therapy is currently in phase I/II clinical trials.(Source: https://www.fightingblindness.org )
- In November 2025, Oxford has joined a national initiative with funding of 15.9 million euros, aiming to transform through the development of highly advanced human tissue models, where the project will use samples of live human tumors to improve preclinical research and its precision.(Source: https://www.ox.ac.uk )
Preclinical and Clinical Trials MarketSegments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content
.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
