In Vivo CAR-T Therapy Market Revenue to Attain USD 10,841.07 Mn by 2035
In Vivo CAR-T Therapy Market Revenue and Trends 2026 to 2035
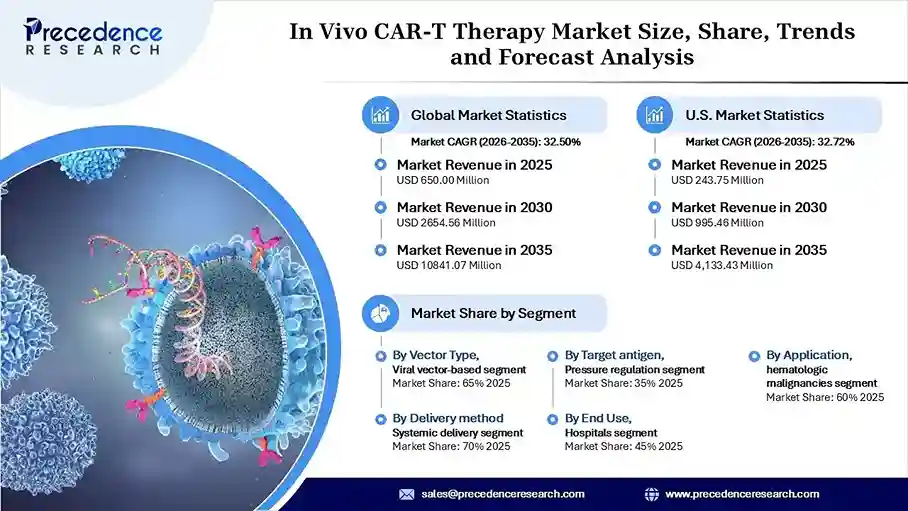
The global in vivo CAR-T therapy market revenue reached USD 650.00 million in 2025 and is predicted to attain around USD 10,841.07 million by 2035 with a CAGR of 32.50%. The market is growing due to the rising development of targeted cancer therapies and increasing R&D investments.
Market at a Glance
The in vivo CAR-T therapy market is an emerging cell therapy approach in which a patient’s T cells are genetically modified directly inside the body, eliminating the need for external cell extraction and laboratory-based engineering. It uses delivery systems such as lipid nanoparticles, viral vectors, and RNA-based platforms to enable faster, more scalable, and potentially more cost-effective treatment production.
This market spans oncology, autoimmune disease treatment, gene delivery technologies, clinical validation pipelines, and future commercial applications. Its growth is driven by increasing collaboration between biotech firms and pharmaceutical companies, integration with hospital systems, and evolving regulatory frameworks aimed at enabling faster and more accessible alternatives to traditional CAR-T therapies.
What Drives the Growth of the In Vivo CAR-T Therapy Market?
Manufacturing Simplicity
Traditional CAR-T therapies require multiple time-intensive steps, including cell harvesting, ex vivo engineering, transport, and reinfusion. In-vivo CAR-T eliminates most of these steps by programming immune cells directly inside the patient, which can reduce treatment delays, infrastructure requirements, and overall cost per patient. This operational efficiency is expected to be a key driver of market adoption.
Expanding Area of Applications
The application of CAR-T therapy is expanding beyond oncology into chronic immune conditions, particularly B-cell-driven autoimmune disorders. In February 2026, Eli Lilly agreed to acquire Orna Therapeutics for up to US$2.4 billion, with its lead candidate ORN-252 targeting CD19, highlighting strong interest in non-cancer applications of in-vivo CAR-T platforms.
Growing Collaborations
Growing collaborations between biotechnology companies, pharmaceutical firms, and gene delivery technology providers are accelerating the development of in-vivo CAR-T therapies by combining expertise in vector design, immunology, and clinical development. These partnerships also help share high R&D costs and regulatory risks, enabling faster clinical translation and scaling of innovative in-body cell engineering approaches.
In April 2026, Eli Lilly and Company entered a definitive agreement to acquire Kelonia Therapeutics, a clinical-stage biotech company focused on in vivo gene delivery. Kelonia has developed its proprietary iGPS platform, which uses engineered lentiviral particles to generate CAR-T cells directly inside the patient’s body.
Market Segmentation Overview
- By vector type, the viral vector-based in vivo CAR-T segment accounted for a revenue share of 65% in the in vivo CAR-T therapy market in 2025, due to their well-established gene delivery mechanisms and early clinical success. The long history of viral transduction has given developers confidence, making these vectors the preferred choice for advancing candidates into human trials.
- By vector type, the non-viral vector-based in vivo CAR-T segment is expected to grow at the fastest CAGR in the market between 2026 and 2035. This growth is attributed to improved safety profiles, reduced immunogenicity, and better scalability, positioning them as a promising alternative for broader market adoption.
- By target antigen, the CD19 segment held a major revenue share of 35% in the market in 2025, due to its proven success in treating B-cell malignancies. Established clinical outcomes, well-understood biology, and strong physician confidence have reinforced CD19 as the primary target for early in vivo CAR-T development.
- By target antigen, the solid tumor targets segment is steadily growing in the market between 2026 and 2035. This expansion is driven by access to larger patient populations and advancements in tumor targeting, antigen discovery, and tumor microenvironment modification.
- By application, the hematologic malignancies segment dominated the in vivo CAR-T therapy market with a 60% in 2025, due to the proven effectiveness of CAR-T therapies in the treatment of blood cancers, where immune cells have a greater route of penetration into affected cells. Leukemia, lymphoma, and myeloma have become dominant targets of in vivo innovation owing to this clinical experience.
- By application, the solid tumors segment is expected to expand rapidly in the market in the coming years, due to enormous unmet clinical needs and growing research intensity. As delivery precision improves, developers are likely to chase opportunities in lung, breast, ovarian, and gastrointestinal cancers.
- By delivery method, the systemic delivery segment accounted for a revenue share of 70% in the in vivo CAR-T therapy market in 2025, due to ease of administration and widespread applicability. Its ability to reach circulating immune cells without specialized procedures made it the practical early option for most developers and treatment centers.
- By delivery method, the localized delivery segment is expected to grow at the fastest CAGR in the market between 2026 and 2035, as it enables more targeted tumor activity while minimizing systemic side effects, making it particularly valuable for complex solid tumors.
- By end-use, the hospitals segment held a major revenue share of 45.0% in the in vivo CAR-T therapy market in 2025, as they are equipped to administer advanced therapies, monitor patients, and manage complications. Their infrastructure and multidisciplinary teams make them the primary centers for CAR-T treatment.
- By end-use, the specialized cancer treatment centers segment is expected to grow at the fastest CAGR between 2026 and 2035, driven by their focused expertise, advanced infrastructure, and participation in clinical trials.
- By development stage, the clinical stage segment accounted for a considerable revenue share of 50% in the in vivo CAR-T therapy market in 2025, as multiple therapies advance through trials. A crowded pipeline across early and mid-stage studies reflected the market’s experimental yet rapidly maturing character.
Regional Analysis
North America dominated the global in vivo CAR-T therapy market with a share of 50% in 2025. This dominance is supported by strong biotech innovation, extensive funding, and a deep clinical pipeline. The U.S. leads the market due to its concentration of advanced research institutions, venture-backed companies, and active regulatory pathways, while Canada contributes through academic and translational research initiatives.
Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period. This growth is driven by increasing clinical trials and rising biotech investments. China leads with strong manufacturing capabilities and supportive innovation policies, while Japan, South Korea, and India are advancing through precision medicine initiatives, improved infrastructure, and expanding R&D investments.
In Vivo CAR-T Therapy Market Coverage
| Report Attribute | Key Statistics |
| Market Revenue in 2025 | USD 650.00 Million |
| Market Revenue by 2035 | USD 10,841.07 Million |
| CAGR from 2026 to 2035 | 32.50% |
| Quantitative Units | Revenue in USD million/billion, Volume in units |
| Largest Market | North America |
| Base Year | 2025 |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and the Middle East & Africa |
Top Companies in the In Vivo CAR-T Therapy Market
Capstan Therapeutics, Interius BioTherapeutics, and Umoja Biopharma are developing systems aimed at manufacturing immune cells inside the body, challenging conventional production techniques. Several companies, such as Sana Biotechnology, Cellectis, Beam Therapeutics, Precision BioSciences, Poseida Therapeutics, CRISPR Therapeutics, Intellia Therapeutics, and Caribou Biosciences, have established strong expertise in gene editing and cell programming that could revolutionize therapeutic precision. Novartis AG, Gilead Sciences (Kite Pharma), Bristol Myers Squibb Company, and Legend Biotech Corporation all utilize their experience in commercial CAR-T therapies together with substantial global networks and regulatory capacity.
Segments Covered in the Report
By Vector Type
- Viral Vector-based In Vivo CAR-T (AAV, Lentiviral)
- Non-Viral Vector-based In Vivo CAR-T (LNPs, Electroporation, Nanoparticles)
By Target Antigen
- CD19
- BCMA
- CD20
- Solid Tumor Targets (HER2, EGFR, Mesothelin, etc.)
By Application
- Hematologic Malignancies (Leukemia, Lymphoma, Myeloma)
- Solid Tumors
- Autoimmune Diseases
By Delivery Method
- Systemic Delivery (Intravenous)
- Localized Delivery (Intratumoral, Targeted Delivery)
By End-Use
- Hospitals
- Specialized Cancer Treatment Centers
- Academic & Research Institutes
By Development Stage
- Preclinical Stage
- Clinical Stage (Phase I/II/III)
- Commercialized Therapies
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
Get this report to explore global market size, share, CAGR, and trends, featuring detailed segmental analysis and an insightful competitive landscape overview @ https://www.precedenceresearch.com/sample/8380
You can place an order or ask any questions, please feel free to contact us at [email protected] |+1 804 441 9344