What is the Transplant Diagnostics Market Size in 2026?
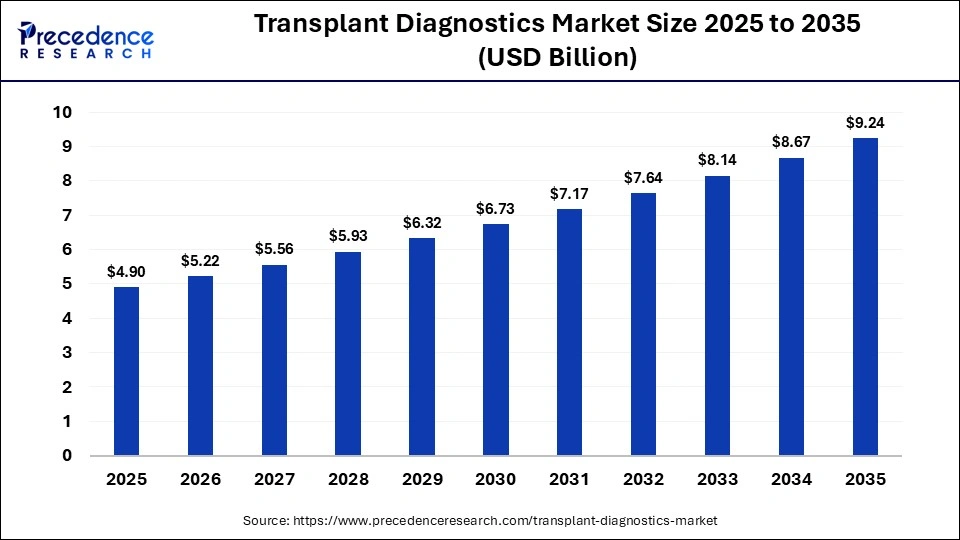
The global transplant diagnostics market size accounted for USD 4.90 billion in 2025 and is predicted to increase from USD $5.22 billion in 2026 to approximately USD 9.24 billion by 2035, expanding at a CAGR of 6.55% from 2026 to 2035. The market is witnessing substantial growth, driven by rising organ failure rates and the demand for advanced, non-invasive molecular testing and AI-driven compatibility tools that boost post-transplant survival rates.
Key Takeaways
- North America dominated the market with a major market share in 2025.
- Asia Pacific is expected to grow at the fastest CAGR between 2026 and 2035.
- By product and service, the instruments segment contributed the highest market share in 2025.
- By product and service, the reagents and consumables segment is growing at a strong CAGR between 2026 and 2035.
- By screening type, the pre-transplant segment held a major market share in 2025.
- By screening type, the post-transplant segment is expected to expand at a notable CAGR from 2026 to 2035.
- By organ type, the kidney segment captured the highest market share in 2025.
- By organ type, the liver segment is poised to grow at a healthy CAGR between 2026 and 2035.
Market Overview
The transplant diagnostics market comprises medical tests, primarily molecular assays and antibody screenings, used to assess donor-recipient compatibility, monitor post-transplant immune responses, and reduce graft rejection risk. The market encompasses instruments, reagents, consumables, and software used for Human Leukocyte Antigen (HLA) typing, crossmatching, and donor-specific antibody (DSA) detection. There is a strong, rapid shift from traditional serological assays toward NGS and PCR-based molecular assays for higher-resolution HLA typing.
How is AI Transforming the Transplant Diagnostics Market?
Artificial intelligence (AI) is transforming the transplant diagnostics market by shifting from reactive to predictive, data-driven, and non-invasive methods. AI analyzes high-dimensional data to identify optimal matches, reducing waitlist mortality and improving long-term graft survival. AI algorithms analyze blood samples for donor-derived cell-free DNA and molecular markers to detect rejection early. AI integrates multi-omics data to tailor drug regimens, minimizing side effects. AI-powered systems analyze Whole-Slide Images (WSI) of tissue biopsies, offering higher accuracy than manual pathology in detecting cellular rejection.
Major Trends in the Market
- Rapid Shift Toward Molecular Diagnostics: There is a strong transition from traditional serology to high-precision molecular techniques, specifically NGS, for HLA typing to minimize mismatch and reduce organ rejection.
- Immersive Adoption of Non-Invasive Monitoring: Non-invasive, biomarker-based tests, such as donor-derived cell-free DNA, are rapidly replacing traditional, painful tissue biopsies for post-transplant surveillance and real-time monitoring of graft health.
- AI and Machine Learning for Optimization: AI and machine learning are being integrated into diagnostic platforms to analyze complex transplant data, enhancing the accuracy of HLA matching and predicting the risk of rejection.
- Personalized Immunosuppression and Molecular Profiling: The industry are moving towards personalized medicine, where diagnostic tests help tailor immunosuppressive therapies to the individual patient's immune risk profile.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 4.90 Billion |
| Market Size in 2026 | USD 5.22 Billion |
| Market Size by 2035 | USD 9.24 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 6.55% |
| Dominating Region | Asia Pacific |
| Fastest Growing Region | North America |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product and Service, Screening Type, Organ Type, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Product and Service Insights
What Made Instruments the Dominant Segment in the Transplant Diagnostics Market?
The instruments segment dominated the market in 2025. This leadership is primarily fueled by the growing demand for advanced, high-throughput, and rapid-processing molecular technologies. The transition from serological to molecular techniques necessitates high-precision, automated, and specialized instruments, enhancing the market position of instrument manufacturers. There is an increasing need for rapid and reliable donor-recipient matching and post-transplant surveillance using molecular assay technologies. Automated systems are being increasingly adopted in hospitals and transplant centers.
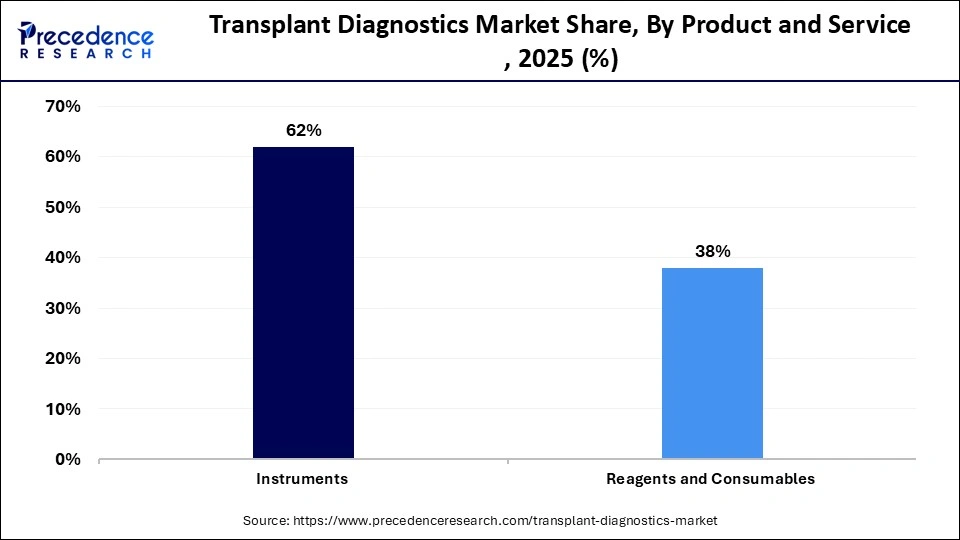
The reagents and consumables segment is expected to experience the fastest growth during the forecast period. This growth is largely due to the high volume of recurring diagnostic procedures required for donor-recipient matching. These reagents must be continuously replenished for each transplant procedure, thereby driving long-term revenue. The rise in global organ transplants and the need for pre-transplant HLA typing and post-transplant monitoring boost the demand. The adoption of molecular assay techniques necessitates the use of specialized, high-resolution kits, thereby increasing the market value of these products.
Screening Type Insights
How Did the Pre-Transplant Segment Lead the Transplant Diagnostics Market?
The pre-transplant segment led the market in 2025. This is mainly due to the critical need for high-precision HLA typing, cross-matching, and antibody screening to ensure donor-recipient compatibility. These measures are essential for maximizing long-term graft survival and minimizing rejection risks. Pre-transplant testing is crucial in preventing acute rejection. The shift from traditional serological methods to molecular techniques allows for faster, more accurate, and high-throughput screening. Advanced molecular assays, including donor-specific antibody detection, help identify immunological risks before the transplant occurs.
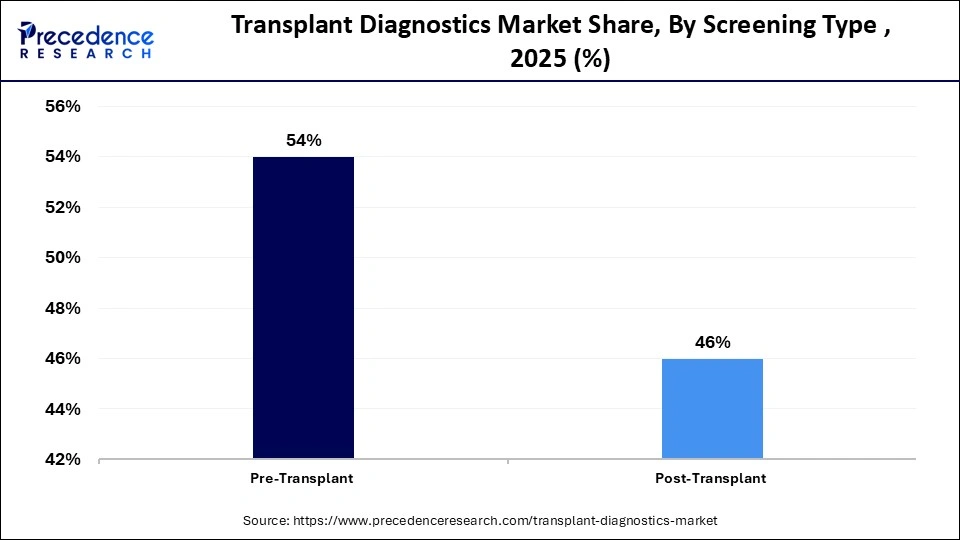
The post-transplant segment is expected to grow at the fastest rate during the forecast period. This is largely driven by the increasing demand for long-term, non-invasive monitoring to manage immunosuppression and prevent graft rejection. As the number of surviving transplant recipients rises, there will be a growing need for regular, non-invasive, and highly accurate monitoring to detect early rejection indicators. The move from invasive biopsies to liquid biopsy-based monitoring enables more frequent and safer assessments of organ function.
Organ Type Insights
Why Did the Kidney Segment Dominate the Transplant Diagnostics Market?
The kidney segment dominated the market in 2025, primarily due to the high global prevalence of end-stage renal disease, which requires continuous, high-volume HLA typing and post-transplant monitoring. Kidney transplantation entails extensive routine diagnostic procedures, such as HLA typing, cross-matching, and donor-specific antibody (DSA) detection, both pre- and post-transplant, sustaining a consistently high market share. The increased availability of both living and cadaveric donors, supported by government initiatives, has led to a rise in transplant numbers, further boosting the demand for diagnostic services.
The liver segment is anticipated to experience the fastest growth during the forecast period. This growth is attributed to a disturbing increase in chronic liver diseases, which has spurred greater demand for liver transplants and advanced molecular diagnostics. An aging population and sedentary lifestyles led to a rise in liver failure cases, escalating the need for transplantation. The growing use of molecular assays and biomarker based tests enables precise compatibility assessments, reducing rejection risks and increasing demand for diagnostic kits. The surge in living donor liver transplants also necessitates rapid and precise diagnostic testing.
Regional Insights
How Did North America Dominate the Transplant Diagnostics Market?
North America dominated the transplant diagnostics market with a major share in 2025, driven by a high volume of transplants, advanced healthcare infrastructure, and a swift adoption of molecular diagnostics. There is a strong preference for molecular assays, including NGS and donor-derived cell-free DNA, which provide high precision for HLA typing and allow for non-invasive rejection monitoring. The presence of specialized hospitals and well-funded laboratory networks supports rapid, complex testing. Many leading diagnostic companies, such as Thermo Fisher Scientific, CareDx, and Illumina, are based in or operate extensively within the U.S., driving innovation in the field.
U.S. Transplant Diagnostics Market Trends
The U.S. is the major contributor to the North American market, driven by a high prevalence of chronic diseases, specialized transplant centers, and robust adoption of molecular assays and NGS. Additionally, it hosts major companies like Thermo Fisher Scientific, Bio-Rad Laboratories, and CareDx, which are leading the way in innovation in histocompatibility and HLA typing. Strong regulatory frameworks and initiatives like the Organ Procurement and Transplantation Network further support the market.
Why is Asia Pacific Considered the Fastest-Growing Region in the Transplant Diagnostics Market?
Asia Pacific is expected to grow at the fastest rate during the forecast period. This growth is mainly driven by increasing adoption of stem cell therapies, high volumes of chronic organ failure, and improving healthcare infrastructure. Significant investments in hospital infrastructure, combined with lower-cost surgical procedures, have made countries like China, India, Thailand, and Japan hubs for medical tourism, boosting transplant demand. Governments in the region are establishing standardized national guidelines for organ donation, implementing reimbursement schemes for diagnostic procedures, and facilitating market expansion.
India Transplant Diagnostics Market Trends
India plays an emerging role within the region, characterized by rising healthcare investment, an increasing burden of end-stage organ diseases, expanding transplant programs, and growing awareness. Rising adoption of cost-effective, sophisticated diagnostic technologies, along with partnerships with international companies and major local players like Dr. Lal Path Labs and Metropolis Healthcare, is making high-end diagnostics more accessible. Managed by the National Organ and Tissue Transplant Organisation.
Why is Europe Considered a Notable Region in the Transplant Diagnostics Market?
Europe is a notable region in the market, mainly characterized by high organ transplant rates, advanced healthcare infrastructure, and rapid adoption of molecular diagnostic technologies for HLA typing. High rates of organ transplants, particularly in Germany, France, and the U.K., drive demand for sophisticated pre- and post-transplant diagnostics. Organizations such as Eurotransplant and Scandiatransplant facilitate efficient organ sharing, thereby boosting demand for standardized diagnostics. Strong presence of key industry players such as Roche, bioMérieux, and Abbott ensures the availability of advanced diagnostic tools.
Transplant Diagnostics Market Value Chain Analysis

Transplant Diagnostics Market Companies
- Thermo Fisher Scientific
- F. Hoffmann-La Roche
- Bio-Rad Laboratories
- Immucor
- CareDx
- Illumina
- Qiagen
- Abbott Laboratories
- Becton, Dickinson and Co
- DiaSorin
Recent Developments
- In February 2026, Thermo Fisher Scientific launched the TacroType™ Pharmacogenetic Test to assist clinicians in determining the optimal dosing of tacrolimus, an immunosuppressive drug for transplant recipients. Correct dosing is crucial; too low a dose may lead to organ rejection, while too high a dose increases risks of complications. Dr. Keith Melancon of the George Washington Transplant Institute emphasized the test's role in minimizing variability in patient responses and enhancing initial dosing decisions.(Source: https://www.businesswire.com)
- In November 2025, CareDx, Inc. unveiled new data at ASN Kidney Week 2025, highlighting the launch of HistoMap™ Kidney. This tissue-based test characterizes rejection types and integrates molecular data with biopsy results to improve diagnostic insights. CareDx's innovations aim to provide clinicians with actionable information for better kidney transplant management.(Source: https://investors.caredx.com)
- In October 2025, Thermo Fisher introduced the Molecular Microscope Diagnostic System for Lung (MMDx Lung), designed to enhance lung transplant rejection detection via molecular analysis of biopsies. By employing machine learning to assess gene expression data, this system addresses the challenges of inconsistent rejection diagnoses among pathologists, potentially improving treatment guidelines for lung transplants.(Source: https://clpmag.com)
Segments Covered in the Report
By Product and Service
- Instruments
- Reagents and Consumables
By Screening Type
- Pre-Transplant
- Post-Transplant
By Organ Type
- Kidney
- Liver
- Heart
- Lung
- Pancreas
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
