What is the Albinism Drug Market Size in 2026?
The global albinism drug market size accounted for USD 280.00 million in 2025 and is predicted to increase from USD 292.88 million in 2026 to approximately USD 439.01 million by 2035, expanding at a CAGR of 4.60% from 2026 to 2035. The market focuses on therapies addressing vision impairment, skin protection, and genetic complications associated with albinism. Growing awareness, early diagnosis, and advances in ophthalmic treatments drive demand. Supportive care medications, sunscreen formulations, and research into gene-based therapies are shaping innovation and expanding treatment accessibility worldwide.
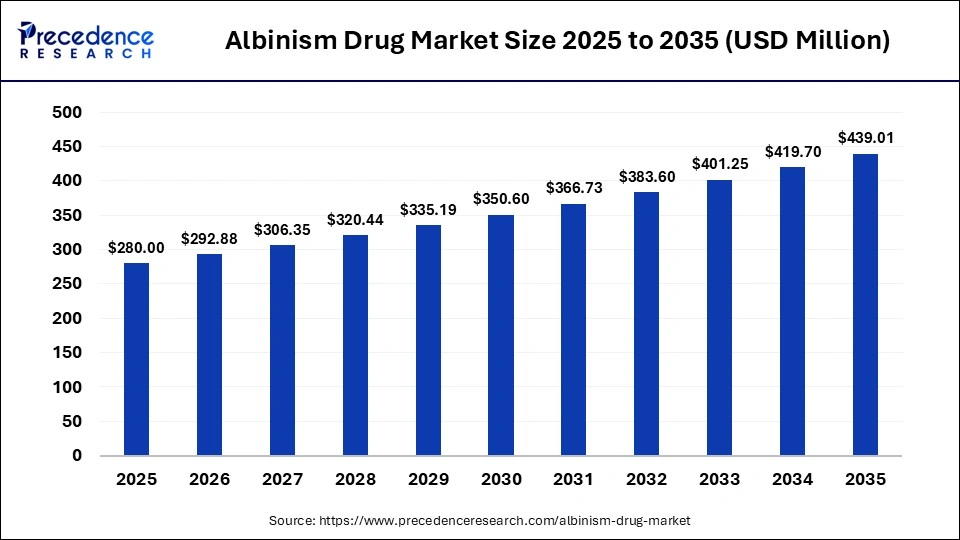
Key Takeaways
- North America led the global albinism drug market in 2025.
- Asia-Pacific is expected to expand at the highest CAGR in the market between 2026 and 2035.
- By indication, the oculocutaneous albinism segment held a dominant revenue share in the market in 2025.
- By indication, the X-linked ocular albinism segment is expected to grow at the highest CAGR between 2026 and 2035.
- By treatment, the hormone drugs segment led the market in 2025.
- By treatment, the photo-sensitive drugs segment is expected to expand at the fastest CAGR from 2026 to 2035.
- By distribution channel, the hospital pharmacy segment held the largest market share in 2025.
- By distribution channel, the online pharmacy segment is expected to grow at a solid CAGR between 2026 and 2035.
How Will New Therapies Transform the Albinism Drug Market?
Albinism drugs refer to therapeutic and supportive treatments designed to manage symptoms of albinism, a genetic condition characterized by reduced or absent melanin production, affecting the skin, hair, and eyes. These include ophthalmic medications, vision-enhancing therapies, dermatological protectants, and emerging gene-based interventions. New therapies such as targeted molecular treatments and gene editing technologies are expected to transform the industry by addressing underlying genetic causes, improving visual outcomes, reducing skin complications, and shifting care from symptomatic management to precision-based treatment approaches.
How Can AI Integration Transform the Albinism Drug Market?
Artificial intelligence integration can significantly transform the global albinism drug industry by accelerating how treatments are discovered and developed. By using advanced machine learning and generative models, researchers can rapidly identify genetic targets specific to albinism, design optimized drug molecules, and predict safety and efficacy far earlier in the process compared with traditional methods.
AI tools also help integrate complex genomic and clinical data to support precision therapies and reduce early-stage attrition. Major AI breakthroughs in protein structure prediction and large-scale data analysis are reshaping rare disease R&D, making tailored and efficient albinism solutions more attainable and cost-effective.
Primary Trends Influencing the Albinism Drug Market
- Advancements in Genetic and Molecular Research: Increasing research into the genetic mutations responsible for albinism is driving innovation in targeted therapies. Scientists are exploring gene therapy, RNA-based treatments, and molecular correction techniques to address the root cause rather than only managing symptoms. This shift toward precision medicine is encouraging pharmaceutical companies to invest in long-term, disease-modifying treatment solutions.
- Growing Focus on Vision-Enhancing Therapies: Since visual impairment is a major complication of albinism, there is a rising demand for advanced ophthalmic drugs and supportive vision therapies. Research into retinal development, optic nerve function, and neuro-visual correction technologies is expanding. Improved diagnostic tools and early intervention strategies are further accelerating the development of specialized eye-related pharmaceutical treatments.
- Rising Awareness and Early Diagnosis Initiatives: Global awareness campaigns and better newborn screening programs are leading to earlier detection of albinism. Early diagnosis allows timely medical intervention, increasing demand for preventive dermatological drugs, visual aids, and supportive therapies. Patient advocacy groups and healthcare organizations are also promoting access to treatment, particularly in underserved regions.
- Expansion of Dermatological Protection Solutions: Individuals with albinism are highly susceptible to sun damage and skin cancer, increasing the need for medicated sunscreens, UV-protective formulations, and skin-repair treatments. Pharmaceutical companies are developing advanced dermatological products with higher efficacy and longer-lasting protection, positioning skin protection as a key segment within the albinism drug landscape.
- Government and Non-Profit Support Programs: Public health initiatives and support from international organizations are improving access to care and funding research activities. Policies promoting rare disease treatment development, incentives for orphan drug designation, and global health collaborations are encouraging pharmaceutical manufacturers to enter and expand within the market.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 280.00 Million |
| Market Size in 2026 | USD 292.88 Million |
| Market Size by 2035 | USD 439.01 Million |
| Market Growth Rate from 2026 to 2035 | CAGR of 4.60% |
| Dominating Region | North America |
| Fastest Growing Region | Asia pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Indication, Treatment, Distribution Channel, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Indication Insights
Why Was the Oculocutaneous Albinism Segment Dominant?
The oculocutaneous albinism (OCA) segment dominated the global albinism drug market due to its higher prevalence and broader clinical manifestations affecting skin, hair, and vision. Patients require continuous dermatological protection, ophthalmic treatments, and long-term medical monitoring. Increased genetic awareness, early diagnosis programs, and supportive therapy demand significantly strengthen treatment adoption within this segment.
The X-linked ocular albinism segment is expected to be the fastest-growing in the coming years, due to increased genetic screening and improved diagnostic capabilities, leading to higher detection rates. Focused research on targeted ophthalmic therapies and personalized interventions has accelerated treatment development. Growing awareness among healthcare providers and patients, along with the expansion of specialty care services and supportive visual solutions, further boosts demand in this specific segment.
Treatment Insights
How the Hormone Drugs Segment Dominated the Albinism Drug Market?
The hormone drugs segment held the largest revenue share of the market in 2025, due to increasing research on melanin-stimulating pathways and hormonal regulation of pigmentation. Therapies targeting melanocortin receptors and related endocrine mechanisms show potential in enhancing residual pigmentation and managing associated complications. Growing clinical interest, supportive trials, and broader applications of hormone-based treatments in rare genetic disorders further strengthen this segment's therapeutic relevance.
The photosensitive drugs segment is expected to grow with the highest CAGR in the market during the studied years, due to rising focus on managing extreme UV sensitivity and preventing skin damage in individuals with albinism. Increasing awareness about skin cancer risks, advancements in photoprotective formulations, and the development of improved light-responsive therapies are accelerating demand. Expanding dermatological research, better access to medicated sunscreens, and supportive public health initiatives further contribute to the rapid adoption of photosensitive treatment solutions.
Distribution Channel Insights
Which Distribution Channel Segment Led the Albinism Drug Market?
The hospital pharmacy segment led the market in 2025, due to the requirement for specialized prescriptions, controlled dispensing of hormone and ophthalmic therapies, and close clinical supervision. Albinism patients often receive treatment through multidisciplinary hospital settings, ensuring proper dosage management and monitoring. Strong supply chain integration, access to rare-disease medications, and coordinated patient care programs further strengthen hospital pharmacies' leading role.
The online pharmacy segment is expected to witness the fastest growth in the market over the forecast period, due to increased digital adoption, easier access without geographical constraints, competitive pricing, and home delivery convenience. Enhanced user experience, telemedicine integration, and broader availability of generic albendazole formulations online attract more consumers. Privacy, 24/7 access, and streamlined prescription fulfillment further drive rapid uptake of online pharmaceutical channels.
Regional Insights
Why North America Dominated the Albinism Drug Market?
North America held a major revenue share of the market in 2025, due to advanced healthcare infrastructure, strong rare disease research funding, and early adoption of innovative therapies. The region benefits from robust genetic testing capabilities, well-established ophthalmic and dermatology care networks, and supportive regulatory pathways for orphan drugs. Active patient advocacy groups, higher awareness levels, and the presence of leading pharmaceutical and biotechnology companies further strengthen treatment development and accessibility across the region.
U.S. Market Trends
In the U.S., albinism drug development is shaped by targeted genetic research and community-driven initiatives. Organizations like the National Organization for Albinism and Hypopigmentation (NOAH) are funding focused studies on ocular pigmentation pathways, enabling new therapeutic candidates to emerge.
Academic and non-profit collaborations support preclinical work that could translate into clinical trials. Meanwhile, dedicated eye research groups at major U.S. institutions are exploring regenerative strategies for vision deficits, reflecting a shift toward precision and patient-informed albinism treatment approaches.
Why is Asia-Pacific Experiencing the Fastest Growth of the Albinism Drug Market?
Asia-Pacific is expected to grow at the fastest CAGR in the market during the predicted timeframe, driven by expanding rare-disease clinical research, rising genetic testing initiatives, and stronger healthcare investment across China, India, and Japan. Increased government support for biotechnology and rare disease programs is enhancing early diagnosis and access to treatment. Growing regional manufacturing capabilities, regulatory reforms that streamline innovative drug approvals, and rising patient awareness are collectively boosting market momentum in 2026.
China Market Trends
In China, albinism drug development is being shaped by the country's rare disease policy reforms and healthcare system innovations. China has expanded its national rare disease list and insurance coverage, significantly lowering costs and improving access to treatments, including rare genetic conditions like albinism.
A nationwide hospital network and digital health platforms facilitate faster diagnosis, data sharing, and clinical research, while government research funding and collaborations between academia and biotech accelerate new therapeutic progress. Patient advocacy and policy pilots also help bring international rare disease drugs into the Chinese market sooner.
How Big is the Opportunity for the Growth of Europe in the Albinism Drug Market?
Europe is expected to grow at a notable CAGR in the foreseeable future, due to strong rare disease collaboration frameworks and research initiatives like the European Rare Diseases Research Alliance (ERDERA), which unites over 170 partners to accelerate diagnostics and therapy development across member states. Supportive policies, orphan drug incentives from the European Medicines Agency, and cross-border clinical networks enhance innovation and patient access. Active patient advocacy and coordinated funding ecosystems further strengthen Europe's potential to lead rare disease drug advancement.
UK Market Trends
The rising prevalence of albinism, growing research activities, and favorable government support propel the market in the UK. Public and private institutions conduct several research programs to develop novel albinism drugs, serving a larger patient population. This is supported by sufficient funding from various organizations. The National Organization for Albinism and Hypopigmentation (NOAH) aims to develop new drugs and lead to new treatments for albinism.
Albinism Drug Market Value Chain Analysis

Albinism Drug Market Companies
- Verisk Health
- Zeomega, Inc.
- Mckesson Corporation
- Cerner Corporation
- Epic Systems Corporation
- Aetna, Inc.
- IBM Corporation
- NextGen Healthcare
- Allscripts Healthcare Solutions
- UnitedHealth Group, Inc.
- eClinicalWorks, Inc
- Optum, Inc
Recent Developments
- In January 2026, an early Phase 1 clinical trial evaluating JWK010, an investigational gene therapy for OCA1, initiated patient recruitment at West China Hospital in Sichuan. The therapy involves a single suprachoroidal injection delivering a functional tyrosinase gene using an AAV vector platform. This marks one of the first clinical-stage gene therapy efforts specifically targeting the genetic root cause of albinism.(Source: https://clinicaltrials.gov)
- In August 2025, the Medical Research Council awarded approximately £542,000 to researchers at the University of Southampton to investigate early eye development abnormalities in infants with albinism. The project focuses on understanding disrupted retinal pathway formation and exploring therapeutic interventions that may enhance visual acuity during critical developmental windows.(Source: https://research.uhs.nhs.uk)
- In March 2025, the University of Southampton received a £135,000 donation from a Guernsey-based charitable trust to accelerate laboratory research focused on inherited retinal disorders, including albinism. The funding supports advanced cellular modeling to test existing approved drugs on genetically engineered retinal cells carrying albinism mutations. Researchers aim to identify repurposable compounds that could improve retinal development and visual signaling.(Source: https://www.islandfm.com)
Segments Covered in the Report
By Indication
- Oculocutaneous Albinism
- X-Linked Ocular Albinism
- Hermansky-Pudlak Syndrome
- Chediak-Higashi Syndrome
By Treatment
- Photo-Sensitive Drugs
- Hormone Drugs
- Others
By Distribution Channel
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
