What is the Antimicrobial Resistance Diagnostics Market Size?
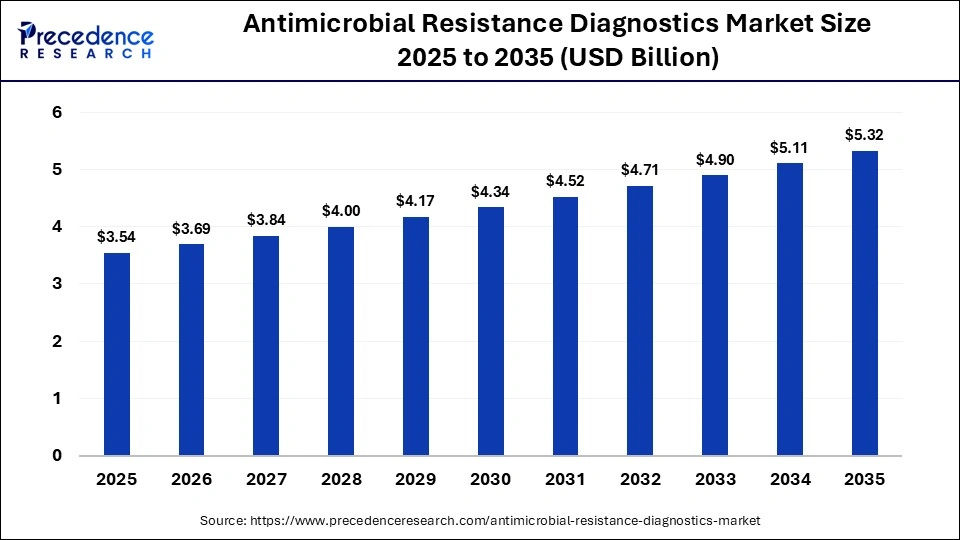
The global antimicrobial resistance diagnostics market size accounted for USD 3.54 billion in 2025 and is predicted to increase from USD 3.69 billion in 2026 to approximately USD 5.32 billion by 2035, expanding at a CAGR of 4.16% from 2026 to 2035. The antimicrobial resistance diagnostics market is expanding steadily, driven by rising drug-resistant infections, improved diagnostic awareness, faster testing technologies, and a stronger emphasis on early, accurate treatment decisions.
Market Highlights
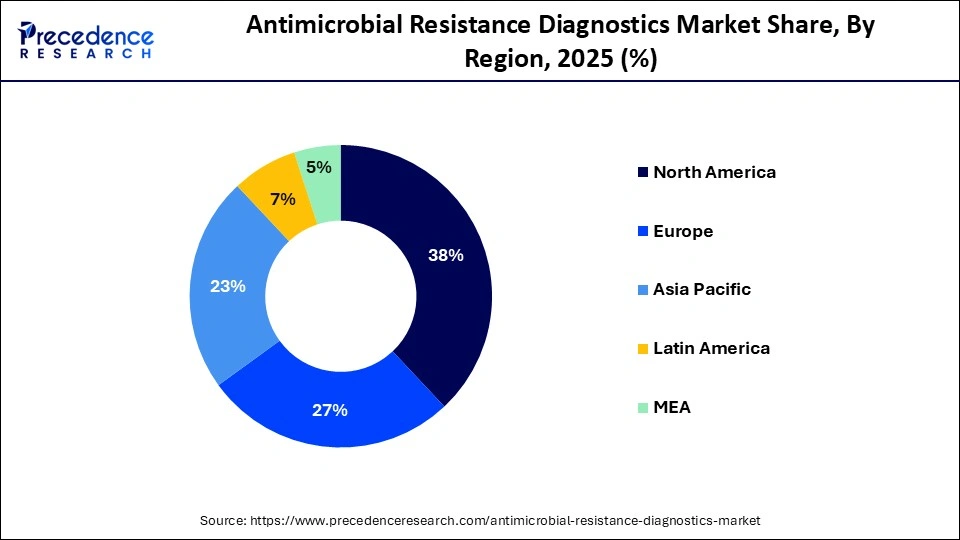
- North America led the antimicrobial resistance diagnostics market with a 38% share in the global market in 2025.
- Asia Pacific is estimated to expand at the fastest CAGR in the market between 2026 and 2035.
- By technology, the phenotypic AMR diagnostics segment captured approximately 46% market share in 2025.
- By technology, the molecular AMR diagnostics segment is growing at a strong CAGR between 2026 and 2035.
- By test type, the antimicrobial susceptibility testing (AST) segment held around 52% market share in 2025.
- By test type, the resistance gene detection segment is projected to grow at a solid CAGR between 2026 and 2035.
- By sample type, the blood & plasma samples segment led the market and held approximately 34% market share in 2025.
- By sample type, the respiratory samples segment is expected to expand at a notable CAGR from 2026 to 2035.
- By application, the clinical diagnostics segment led the market and held approximately 63% market share in 2025.
- By application, the veterinary diagnostics segment is expected to expand at a notable CAGR from 2026 to 2035.
- By end-user, the hospitals & clinical laboratories segment held around 49% market share in 2025.
- By end-user, the high public health & research institutes segment is projected to grow at a solid CAGR between 2026 and 2035.
What Diagnostic Tools Are Capable of Minimizing Antimicrobial Resistance (AMR)?
The diagnostic market is focused on developing products that will provide healthcare professionals with the ability to identify bacterial pathogens, determine if they are resistant to antimicrobial agents, and then make the appropriate choice of treatment based on this information.
Currently, there is significant demand in this market driven by the increased incidence of multi-drug-resistant infections, increased burden of hospital-acquired infections, and more emphasis on antimicrobial stewardship programs. Advanced molecular diagnostic techniques, as well as automated susceptibility testing, are quickly becoming preferred methods due to their rapid identification of resistant organisms and their far greater accuracy than traditional culture-based diagnostic systems. Furthermore, the application of artificial intelligence, machine learning, and data analytics will continue to enhance diagnostic accuracy and improve workflow efficiencies. Therefore, the continued development of antimicrobial resistance diagnostics will facilitate the development of sustainable infection management.
Emerging Technologies and Artificial Intelligence Are Transforming the Future of Antimicrobial Resistance Diagnostics
Artificial Intelligence (AI) has made strides toward revolutionizing the way antimicrobial resistance diagnostics are performed through the application of advanced technology and utilizing AI to provide a quicker and more accurate way to detect specific resistant pathogens and develop predictive analytics on resistance development. Current research shows that AI models have improved the accuracy of resistance detection through example studies conducted on Mycobacterium tuberculosis and Staphylococcus aureus; such studies have shown that AI models provide a greater accuracy of recognition than traditional methods and decrease the chances of misdiagnosis.
There are studies currently underway that are investigating the use of Machine Learning models in predicting resistance patterns within E. coli, and these studies might one day provide valuable insight into how to best monitor and prevent outbreaks from E. coli. Additionally, beyond diagnostics, AI is also playing an integral role in creating more effective antibiotic stewardship and developing new therapeutic approaches to address AMR globally.
Key Trends Driving the Evolution of Diagnostics for AMR
- Rise in molecular testing: Healthcare providers are using PCR and isothermal molecular tests more frequently, allowing for an improved ability to identify and treat resistant pathogens in a timely manner, thereby decreasing the use of unneeded/ineffective antibiotics and improving patient outcomes.
- AI in Resistance Detection: AI is being utilized in molecular diagnostics platforms to enhance the interpretation of complex resistance-related information and to improve diagnostic performance while providing support to physicians in selecting more specific/effective antimicrobial therapy.
- Growth in Point-of-Care Testing: Portable POC testing for AMR is becoming increasingly available in hospitals and various nonhospital settings, thus supporting early identification of resistant infections and improving access to timely clinical decision-making.
- Increased syndromic panels demand: Molecular syndromic panels, which test for multiple pathogens and resistance markers, are experiencing a high adoption rate because they provide improved efficiency and diagnostic confidence to the clinician managing infectious diseases.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 3.54 Billion |
| Market Size in 2026 | USD 3.69 Billion |
| Market Size by 2035 | USD 5.32 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 4.16% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Technology, Test Type, Sample Type, Application, End-User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segmental Insights
Technology Insights
Why Are Phenotypic AMR Diagnostics Dominating the Market?
The phenotypic AMR diagnostics are the dominant segment with a 46% share, because they measure the direct effect of antibiotic activity on microbial growth. This makes them the most reliable test for determining how to treat an infection. Culture and sensitivity testing and automated phenotypic antistaphylococcal tests (AST) systems have been standard practice within hospitals, particularly when diagnosing patients with bloodstream and serious infections. In conjunction with clinical guidelines, all phenotypic antimicrobial resistance diagnostics deliver clinically informative susceptibility profiles and continue to be in high demand, although these tests typically take longer for results to be reported back.
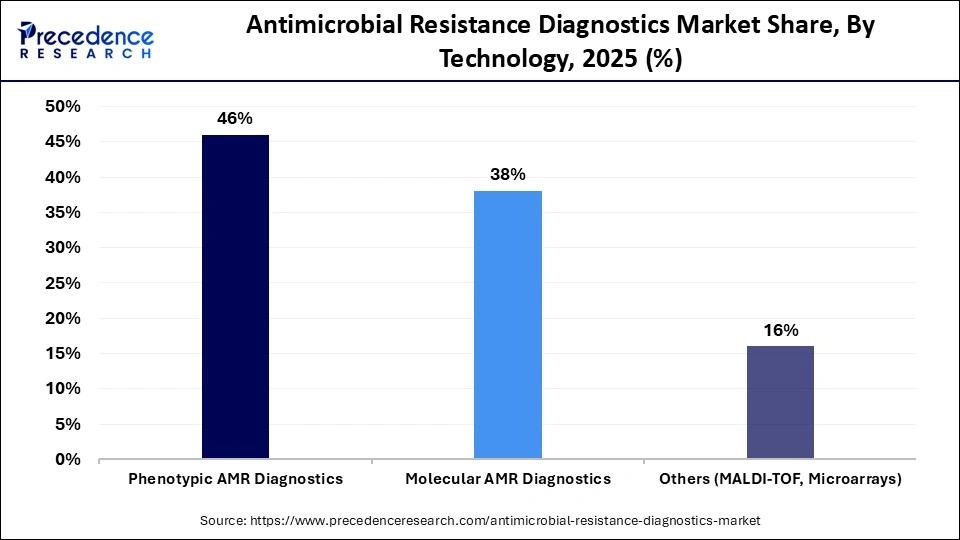
Molecular-based AMR diagnostics are set to be the most rapidly expanding segment during the forecasted period for antimicrobial resistance diagnostic technology. The speed and versatility of these tests are a major reason for their continued growth in popularity for evaluating patients with time-critical infections. The capacity of molecular tests to swiftly identify and characterize antimicrobial resistance genes enables more timely and appropriate decisions for antimicrobial agents to be made. Advancements in the development of multiplex assays, reductions in the cost of sequencing technologies, and the increasing usage of molecular-based AMR tests in decentralized laboratory settings will continue to most likely catapult the transition from phenotypic to molecular-based antimicrobial resistance testing.
Test Type Insights
Why Are Antimicrobial Susceptibility Tests Still the Leading Form of Antimicrobial Resistance Diagnostics?
Antimicrobial susceptibility tests segment led the market with a 52% share during 2025, because they give direct evidence of how microbes respond to a specific set of antibiotics. Antimicrobial susceptibility tests provide important information to guide clinicians in choosing the right therapy, to limit unnecessary therapy, and to slow down the development of antimicrobial resistance. As the technology around antimicrobial susceptibility tests continues to develop, companies are developing automated platforms that can integrate with the minimum inhibiting concentrations, allowing hospitals to use them consistently and throughout acute care, intensive care, and infectious disease management areas.
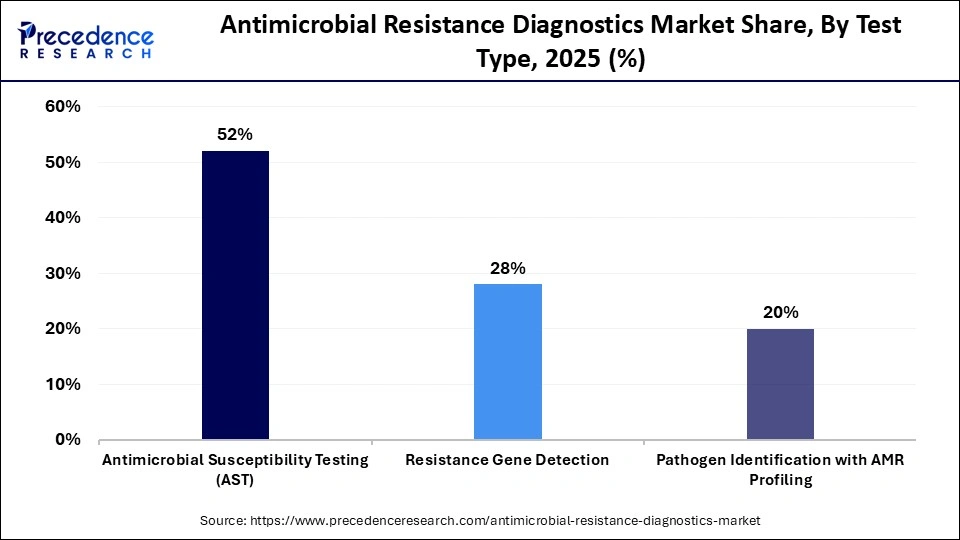
The fastest growing type of antimicrobial resistance diagnostics is the detection of resistance genes. These tests identify genetic markers that correlate with antimicrobial resistance so that clinicians can make rapid decisions about patient treatment. Additionally, they are used in the tracking of outbreaks and support strategies for controlling the spread of infections. The growing use of molecular panels, syndromic testing, and strategic programs will continue to drive the adoption of resistance gene tests, particularly in areas where there is a strong emphasis on early intervention and precision diagnostics.
Sample Type Insights
What Causes Increased Demand for Antimicrobial Resistance Diagnostics with Blood and Plasma Biopsies?
Blood and plasma samples segment dominated the market with a 34% share, because these sample types are pivotal in identifying bloodstream infection (BSI), as well as identifying pathogens associated with sepsis. Therefore, timely and accurate detection of resistance profiles based on blood sample methods is critical to improving outcomes for patients with high-mortality infections in hospitals. Furthermore, the latest automated blood culture technologies, as well as the continuing rise in the incidence of hospital-acquired infections, are further supporting this growth trend.
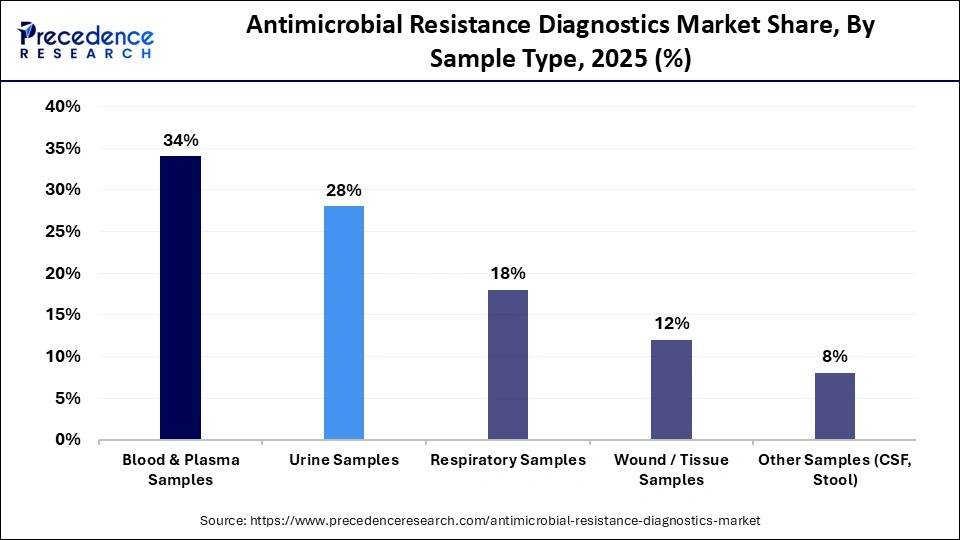
The respiratory samples are the fastest-growing sample types for antimicrobial resistance diagnostics. Respiratory infection incidence continues to rise, with increases in pneumonia, ventilator-associated infections (VAIs), and drug-resistant respiratory pathogens. Increased molecular diagnostic testing using respiratory panels and increased surveillance of hospital or airborne transmission of infections, along with increased use of AMR testing for respiratory infections in both inpatient and outpatient environments, expand the opportunities for antimicrobial resistance testing using respiratory specimen types.
Application Insights
Clinical diagnostics held the largest share of 63% in the antimicrobial resistance diagnostic market in 2025, as AMR tests are a critical component of patient care, infection prevention and control, and antimicrobial stewardship initiatives. Hospitals, diagnostic testing companies, and other healthcare providers use antimicrobial resistance assays to support their selection of appropriate medications, decrease the chances that therapy will fail, and manage patients who have developed resistant infections. Regulations continue to stress the importance of accurate diagnosis, and growing awareness by physicians of the significance of AMR testing only serves to solidify the leadership position that clinical diagnostics fill within the overall antimicrobial resistance diagnostics market.
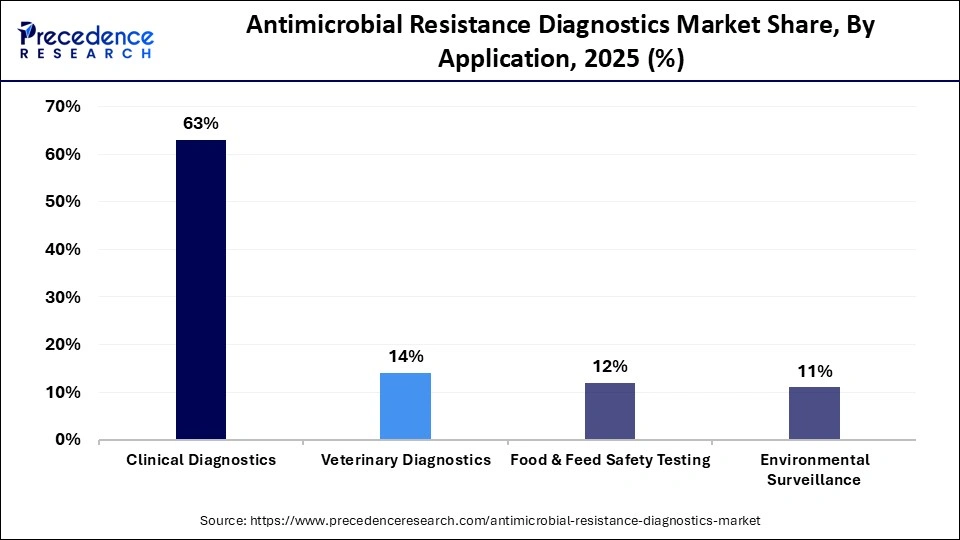
Veterinary diagnostics represent the fastest-growing application category and will reflect continued growth as more people recognize the risk of transmission of zoonotic diseases and the inappropriate use of antibiotics within the veterinary medicine community. In addition, the surveillance of resistance patterns within commercial livestock, companion animals, and poultry will continue to grow and subsequently fuel the demand for AMR testing throughout the veterinary community, as guidelines and regulations regarding the use of antibiotics become increasingly strict.
End-User Insights
How Hospitals and Clinical Laboratories Shape Antimicrobial Resistance Diagnostics Testing?
The hospitals and clinical laboratories lead the antimicrobial resistance diagnostic market with a 15% share as hospitals and clinical laboratories handle complex cases of infections where time-sensitive antimicrobial selection is crucial. In addition, hospitals' infrastructure, including automated testing equipment and links to hospital information systems, further establish their continued centrality as laboratories that conduct routine and advanced antimicrobial resistance testing.
The public health and research institutions also comprise the biggest group of end users of AMR testing. Driven by new AMR surveillance efforts from countries worldwide and through government-funded research programs, public health and research institutions are fully engaged in supporting the tracking of resistant bacteria, validating new AMR diagnostic methods, and providing input into national and international antimicrobial resistance policies. With the increasing interest in national and global AMR monitoring systems, this market will continue to expand.
Regional Insights
How Big is the North America Antimicrobial Resistance Diagnostics Market Size?
The North America antimicrobial resistance diagnostics market size is estimated at USD 1.35 billion in 2025 and is projected to reach approximately USD 2.03 billion by 2035, with a 4.16% CAGR from 2026 to 2035.
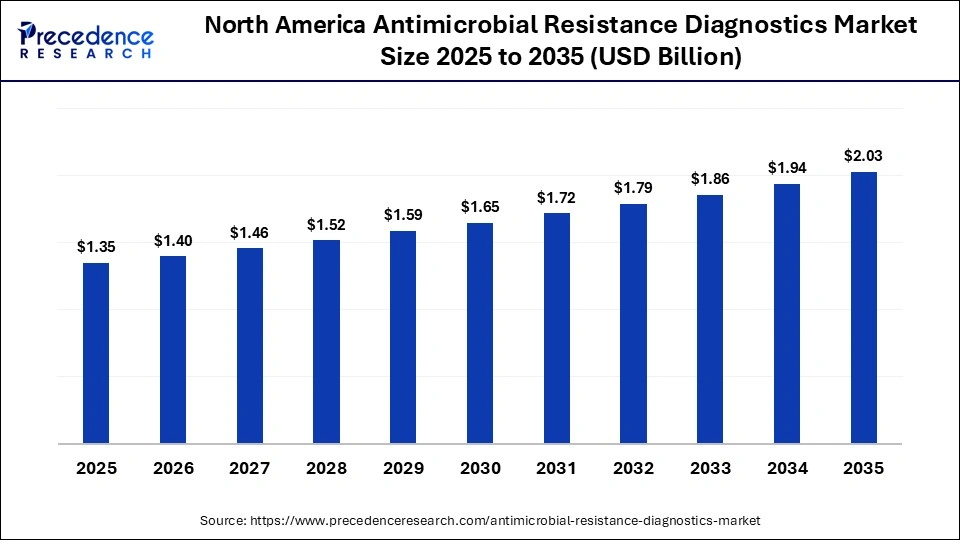
Why is the North American region dominating the Antimicrobial Resistance Diagnostics Market?
North America has a strong infrastructure for the development of new diagnostics to combat antimicrobial resistance, including advanced laboratory networks, a significant number of independent laboratories using molecular diagnostics, and an extensive integration of healthcare data systems. The growth of rapid pathogen identification and antimicrobial resistance detection tools continues to generate innovative approaches for the clinical care of patients, while partnerships between academic institutions, public health agencies, and hospitals support ongoing surveillance and educational initiatives. High levels of awareness and continued investment in the development of new diagnostic technologies have made North America the leader in dealing with complex antimicrobial resistance problems in a timely, precise manner.
What is the Size of the U.S. Antimicrobial Resistance Diagnostics Market?
The U.S. antimicrobial resistance diagnostics market size is calculated at USD 1.01 billion in 2025 and is expected to reach nearly USD 1.53 billion in 2035, accelerating at a strong CAGR of 4.24% between 2026 and 2035.
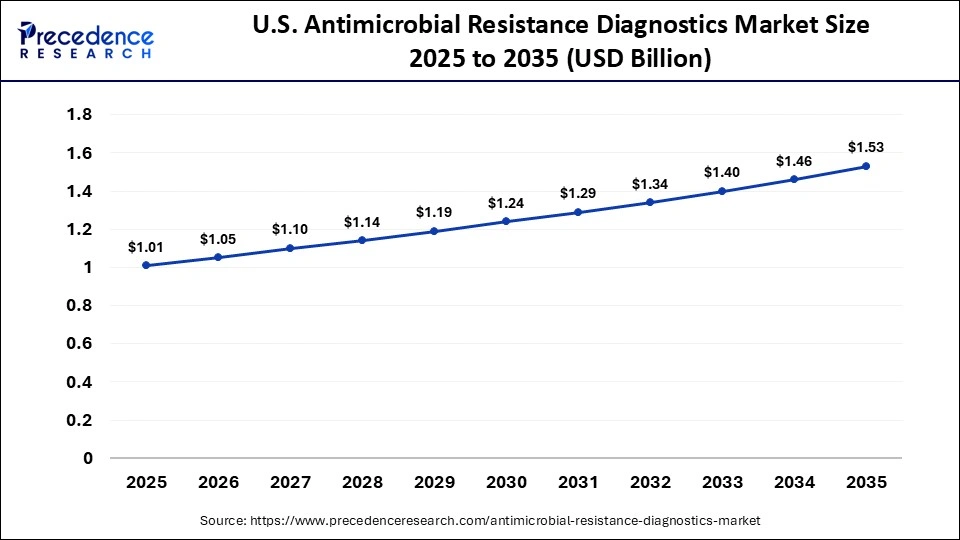
U.S. Antimicrobial Resistance Diagnostics Market Trends
The United States leads in the development and use of diagnostics for antimicrobial resistance. Robust research capacity, innovative biotech companies, and a supportive regulatory environment foster the rapid approval of new diagnostic products. A substantial ecosystem of clinical trials and focus on precision medicine provides the impetus for rapid adoption of new diagnostic technologies. As genomic insights become integrated into routine diagnostic testing, it will provide improved identification of resistant strains and an improved pathway for improved patient care delivery across the United States.
What is the Asia Pacific Antimicrobial Resistance Diagnostics Market Size?
The Asia Pacific antimicrobial resistance diagnostics market size is expected to be worth USD 1,230 million by 2035, increasing from USD 810 million by 2025, growing at a CAGR of 4.27% from 2026 to 2035.
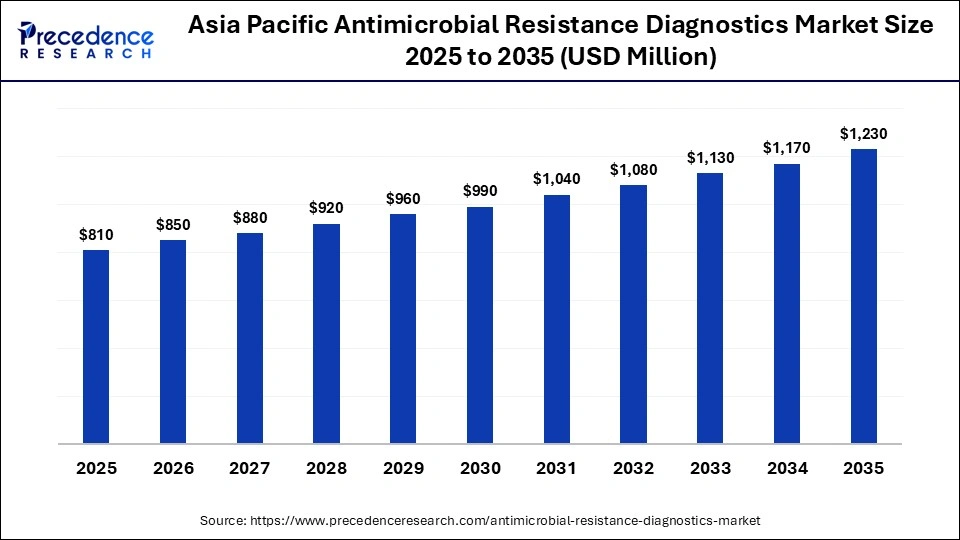
Why Is Asia Pacific Experiencing the Fastest Growth of the Antimicrobial Resistance Diagnostics Market?
Antimicrobial resistance diagnostics are on the rise due to fast-growing access to healthcare, improved diagnostics, and increased awareness of antimicrobial-resistant infections across Asia-Pacific. Health systems and governments are expanding the number of laboratories available, becoming more sophisticated in using molecular or POC testing, and strengthening their regional surveillance systems. The population of ASIA-PACIFIC is very diverse, and many regions have a high burden of infectious diseases, creating an established incentive for the growth of diagnostics. The expanding coalitions of knowledge and investments in workforce development will continue to positively impact the scale of diagnostics utilized and prepared to respond effectively to the overall need.
India Antimicrobial Resistance Diagnostics Market Trends
India has made great strides in developing diagnostics for Antimicrobial Resistance (AMR), and this trend is continuing. National Policy strategies, the growing number of Laboratories across India, expanding access to Diagnostic Services beyond tier-1 cities due to developing low-cost rapid testing devices, and the continuing development of new Rapid Tests through academic and clinical partnerships have led to ongoing refinement of Diagnostics Development to meet specific regional needs.
What is the Europe Antimicrobial Resistance Diagnostics Market Size and Growth Rate?
The Europe antimicrobial resistance diagnostics market size has grown strongly in recent years. It will grow from USD 960 million in 2025 to USD 1,450 million in 2035, expanding at a compound annual growth rate (CAGR) of 4.21% between 2026 and 2035.
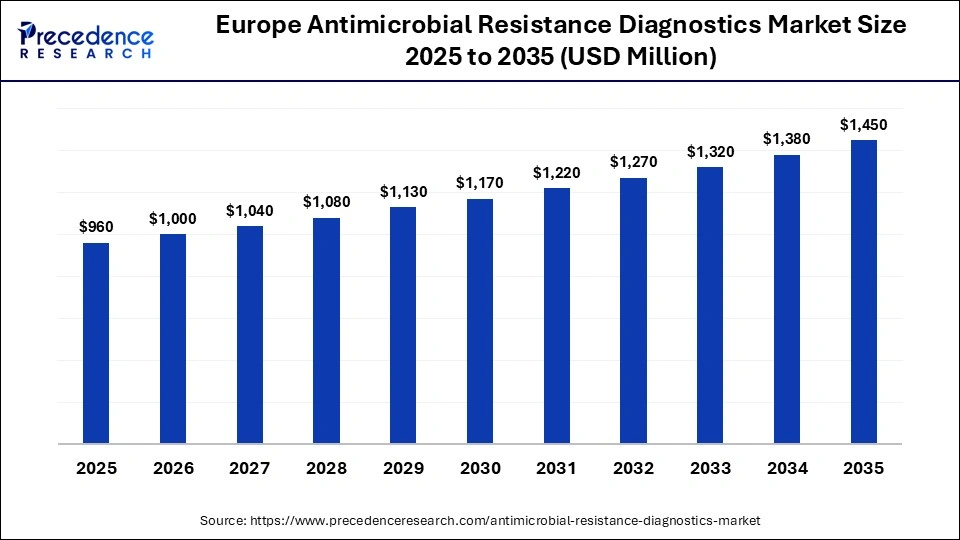
How is the Opportunistic Rise of Europe in the Antimicrobial Resistance Diagnostics Market?
Europe is expected to experience an opportunistic rise in the market, driven by strong public health initiatives, increased government funding, and coordinated surveillance programs aimed at combating drug-resistant infections. The region benefits from advanced healthcare infrastructure, rapid adoption of molecular and point-of-care diagnostics, and supportive regulatory frameworks, particularly under EU-led action plans on AMR. Additionally, growing collaboration between research institutions, diagnostic companies, and healthcare providers is accelerating innovation and market expansion across the region.
Who are the Major Players in the Global Antimicrobial Resistance Diagnostics Market?
The major players in the antimicrobial resistance diagnostics market include BD, BIOMERIEUX, Abbott, Accelerate Diagnostics, Inc., Danaher, Hologic Inc. (Gen Probe), Molsid, F. Hoffmann-La Roche, Ltd., Vela Diagnostics, Visby Medical, Inc., OpGen, Seegene Inc, EliTechGroup, and CERTEST BIOTEC.
Recent Developments
- In June 2025, A new global collaboration to accelerate access to and effective use of diagnostics to fight antimicrobial resistance (AMR) launched in Accra, focusing on innovation and equity in low- and middle-income countries.(Source: https://gardp.org)
- In January 2026, ShanX Medtech raised €24 million to develop its ultra-rapid point-of-care antibiotic diagnostic system, aiming to deliver same-day results and reduce reliance on slow traditional culture tests.(Source: https://ioplus.nl)
- In September 2025, Molecular Designs launched the Urogenital Microbiota with ABX 53 Simplicity Panel, a multiplex PCR panel detecting 28 organisms and 25 resistance genes with same-day results for research labs.(Source: https://www.prnewswire.com)
Segments Covered in the Report
By Technology
- Phenotypic AMR Diagnostics
- Culture & Sensitivity Assays
- Automated Phenotypic AST Systems
- Molecular AMR Diagnostics
- PCR & qPCR-based Assays
- Next-Generation Sequencing
- Others (MALDI-TOF, Microarrays)
By Test Type
- Antimicrobial Susceptibility Testing (AST)
- Resistance Gene Detection
- Pathogen Identification with AMR Profiling
By Sample Type
- Blood & Plasma Samples
- Urine Samples
- Respiratory Samples
- Wound/Tissue Samples
- Other Samples (CSF, stool)
By Application
- Clinical Diagnostics
- Veterinary Diagnostics
- Food & Feed Safety Testing
- Environmental Surveillance
By End-User
- Hospitals & Clinical Laboratories
- Reference & Specialized Laboratories
- Veterinary Clinics & Labs
- Public Health & Research Institutes
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
