What is the cGMP Manufacturing Market Size?
The global cGMP manufacturing market is growing as pharma and biotech companies invest in compliant, high-quality production systems for drugs, biologics, and advanced therapies. The cGMP manufacturing market is driven by rising demand for biologics and stringent global regulatory compliance requirements.
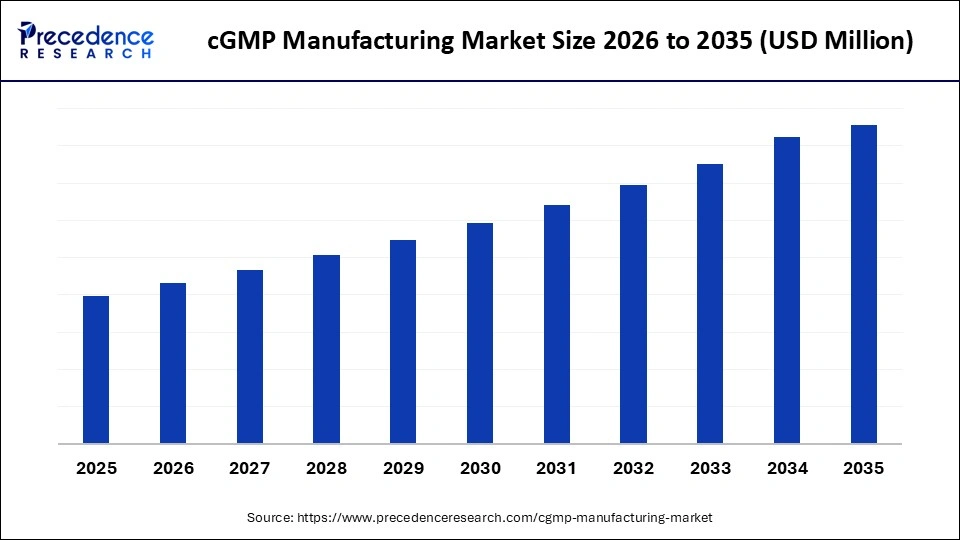
Market Highlights
- North America dominated the market, holding the largest market share of 44.6% in 2025.
- The Asia Pacific is expected to grow at the fastest CAGR of 8.4% between 2026 and 2035.
- By product type, the biologics segment held the major market share of 45.8% in 2025.
- By product type, the cell & gene therapies segment is expected to grow at a CAGR of 7.5% between 2026 and 2035.
- By process type, the upstream processing segment contributed the highest market share of 53.4% in 2025.
- By process type, the fill-finish segment is growing at a strong CAGR of 7.8% between 2026 and 2035.
- By scale of operation, the clinical segment captured the highest market share of 54.7% in 2025.
- By scale of operation, the commercial is set to grow at a remarkable share of 7.5% CAGR between 2026 and 2035.
- By service type, the manufacturing services segment accounted for the largest market share of 46.8% in 2025.
- By service type, the packaging & fill-finish services segment is expected to grow at a solid CAGR of 7.6% between 2026 and 2035.
- By therapeutic area, the oncology segment held the largest share of 42.8% in the market during 2025.
- By therapeutic area, the rare diseases is expanding at a healthy CAGR of 7.5% between 2026 and 2035.
- By end-user, the pharmaceutical and biotechnology companies segment contributed the highest market share of 43.7% in 2025.
- By end-user, the CDMOS / CMOS segment is expected to expand at a remarkable growth rate of 7.7% CAGR between 2026 and 2035.
Redefining Biopharmaceutical Production: How cGMP Manufacturing Is Powering the Next Wave of Advanced Therapies
The cGMP manufacturing market is the heart of the current biopharmaceutical manufacturing, which guarantees that drugs, biologics, cell and gene therapies, and vaccines are manufactured to global standards of quality, safety, and consistency. The market is at the interfaces of a broad spectrum, both upstream and downstream processing, as well as formulation, aseptic filling, analytical testing, and regulatory documentation. With the increasingly complex therapeutic pipelines and tightened controls of regulatory authorities, the presence of cGMP-certified facilities and technologies is becoming a necessity.
The expansion of the cGMP manufacturing market is driven by the rapid expansion of biologics, which are monoclonal antibodies, recombinant proteins, and biosimilars that necessitate highly controlled environments to manufacture them. Increased cell and gene therapies have further exacerbated the need to supply specialized cleanrooms, viral vectors, and advanced single-use systems. Pharmaceutical firms are gaining more and more exposure to outsourcing to CDMOs to cut down on capital, speed up the schedules, and gain access to the latest capabilities. Moreover, the FDA, EMA, and other international regulatory bodies are increasing regulatory demands, which compel producers to resort to automation, digital quality assurance/quality control solutions, real-time oversight, and streamlined procedures.
Key AI Integration in the cGMP Manufacturing Market
The adoption of AI in the cGMP production market is redefining the state of biopharmaceutical production, control, and release by making it automatable, predictive, and real-time throughout the entire chain of value. Quality control and real-time release testing based on AI will be used to identify deviations in the initial stages and allow for maintaining closer control over important process parameters and minimizing failures in batches. AI can be applied in process development, wherein the data obtained through the process history is analyzed to improve cell culture conditions, purification workflows, and yield consistency, and minimize experimentation time.
AI is also able to simplify regulatory documentation by use of intelligent batch records, automatic SOP updates, and audit readiness checks by use of data integrity. As the production process is shifted towards sophisticated biologics and cell therapy and gene therapies, AI increases efficiency, reliability, and compliance, becoming one of the key sources of digital transformation in the modern GMP plants.
CGMP Manufacturing Market Outlook
[[market_outlook]]
Market Scope
| Report Coverage | Details |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product Type, Process Type, Scale of Operation, Service Type, Therapeutic Area, End-User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
cGMP Manufacturing Market Segment Insights
[[segment_insights]]
cGMP Manufacturing Market Region Insights
[[regional_insights]]
cGMP Manufacturing Market Companies
- Lonza Group
- Thermo Fisher Scientific / Patheon
- Catalent Pharma Solutions
- Samsung Biologics
- Fujifilm Diosynth Biotechnologies
- Boehringer Ingelheim BioXcellence
- WuXi AppTec / WuXi Biologics
- AbbVie Contract Manufacturing
- AGC Biologics
- Novartis Gene Therapies
- Baxter BioPharma Solutions
- Recipharm
- Jubilant Biosys/Jubilant Pharmova
- Lonza's Cell & Gene Therapy Unit
- Rentschler Biopharma
Recent Developments
- In September 2025, NFIL inaugurated Phase 1 of the new cGMP-4 plant at its plant in Dewas, Navin Molecular, with the participation of the European partner companies. It is a large acquisition for the global presence of the company in the specialty chemicals and CDMO markets.(Source: https://www.indiapharmaoutlook.com)
- In August 2025, ProBio proclaimed its manufacturing services of cGMO Adeno-Associated Virus (AAV) in a new 128000 sq. ft. facility in Hopewell, New Jersey. The expansion will be in response to a growing supply requirement in viral vectors and will make ProBio stronger in gene and cell therapy development.(Source: https://www.biospectrumasia.com)
- In July 2025, The Flamma Group announced the opening of another cGMP production plant in Dalian, China. The facility enhances its ability to aid in the synthesis and manufacturing of New Chemical Entities and late-stage intermediates for the worldwide pharmaceutical innovators.(Source: https://www.contractpharma.com)
cGMP Manufacturing Market Segments Covered in the Report
[[segment_covered]]
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content
.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
