What is the Clinical AI Model Governance Market Size in 2026?
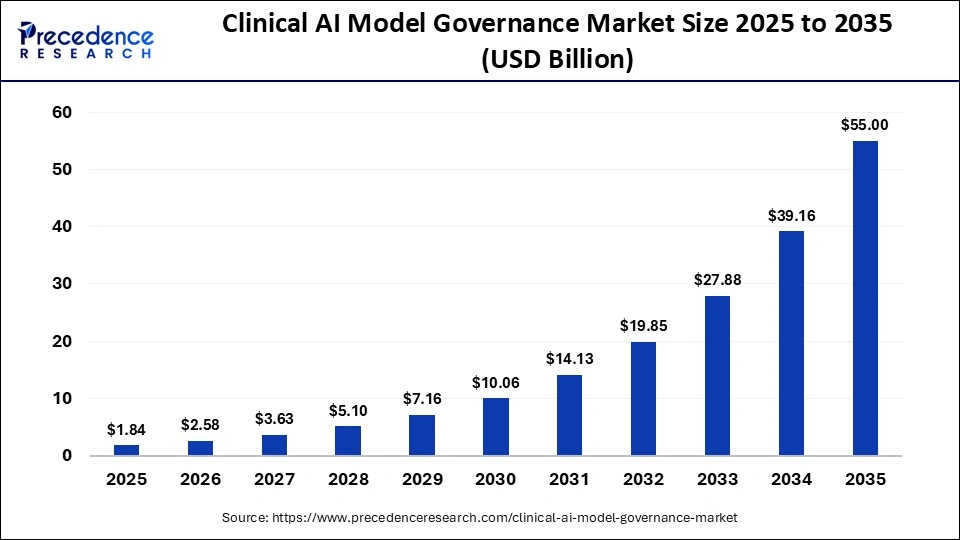
The global clinical AI model governance market size accounted for USD 1.84 billion in 2025 and is predicted to increase from USD 2.58 billion in 2026 to approximately USD 55.00 billion by 2035, expanding at a CAGR of 40.46% from 2026 to 2035. The market growth is attributed to the rapid adoption of artificial intelligence in healthcare and the increasing need for governance frameworks that ensure transparency, safety, and regulatory compliance for clinical AI models.
Key Takeaways
- North America dominated the market with the major market share in 2025.
- Asia-Pacific is expected to grow at the fastest CAGR between 2026 and 2035.
- By component, the software platforms segment contributed the highest clinical AI model governance market share in 2025.
- By component, the services segment is expected to grow at the fastest CAGR between 2026 and 2035.
- By deployment mode, the cloud-based segment held a dominant revenue share in the market in 2025 and is expected to sustain its position in the upcoming period.
- By application, the risk and compliance management segment captured the highest market share in 2025.
- By application, the model monitoring and performance management segment is poised to grow at the highest CAGR between 2026 and 2035.
- By end user, the hospitals and health systems segment generated the biggest market share in 2025.
- By end user, the pharmaceutical and biotechnology companies segment is expected to expand at the fastest CAGR between 2026 and 2035.
Which Factors Drive the Clinical AI Model Governance Market?
The rapid integration of artificial intelligence (AI) into healthcare systems is a major driver accelerating the clinical AI model governance industry. Hospitals and regulators increasingly require frameworks that ensure safe, transparent, and compliant deployment of clinical AI models. Clinical AI model governance technologies and processes to monitor, validate, audit, and manage AI algorithms utilized in healthcare applications.
Such applications include disease prediction, analysis of diagnostic imaging, treatment recommendation, and patient risk assessment tools that are applied in modern healthcare systems. U.S. Department of Health and Human Services HealthIT program shows that 71% of U.S. hospitals were using predictive AI integrated with electronic health record systems in 2024. Furthermore, the use of AI model governance solutions is growing at a high pace, with healthcare providers implementing predictive AI in clinical processes.
Clinical AI Model Governance Market Trends
- Emergence of Federated Learning for Privacy-Preserving Clinical AI: Healthcare organizations are increasingly adopting federated learning to train AI models across multiple hospitals without sharing sensitive patient data. This strategy allows shared model creation and ensures that there are stringent policies for data privacy and other governance compliance. Research indicates that federated learning promotes secure analytics across institutions, as it is an essential governance trend for the scalability of implementing clinical AI.
- Regulatory Expansion of High-Risk AI Frameworks in Healthcare: Global regulators are introducing stricter compliance requirements for AI systems used in medical decision-making and diagnostics. The AI Act of the European Union, which came into effect in 2024, identifies a large number of medical uses of AI as high-risk systems. These regulatory trends are motivating the need to develop governance platforms that can assist healthcare organizations in staying compliant in the complicated AI environments.
Growth Factors
- Increasing Regulatory Oversight of Clinical AI Systems: Strengthening global regulatory scrutiny of medical AI technologies is driving demand for structured governance platforms, ensuring safety and compliance.
- Rising Deployment of AI-Powered Diagnostic Tools: The growing use of AI in radiology, pathology, and clinical decision support is fuelling the need for continuous model monitoring frameworks.
- Increasing Investment in Responsible and Ethical AI Development: Rising funding for trustworthy AI technologies is driving the development of governance platforms focused on bias detection and ethical compliance.
The Rising Wave of Regulated Clinical AI Driving Governance Transformation in Healthcare
- The U.S. Food and Drug Administration (FDA) remains the global leader in regulating clinical AI systems, with more than 1,356 AI-enabled medical devices authorized for clinical use by 2025, covering applications such as diagnostic imaging, cardiovascular monitoring, and clinical decision support tools.
- In 2024 alone, the FDA cleared 168 machine learning enabled medical devices, with approximately 94.6% approved through the 510(k) regulatory pathway and the remainder receiving De Novo authorization under risk-based regulatory evaluation
- Hospital adoption of predictive AI tools has increased significantly, with approximately 71% of non-federal acute care hospitals reporting AI use in clinical or operational functions by 2024, reinforcing the need for governance frameworks that manage model safety, transparency, and regulatory compliance.
- The U.S. Food and Drug Administration reported that the number of AI- and machine learning-enabled medical devices authorized in the United States exceeded 950 by 2024, reflecting the rapid expansion of regulated clinical AI technologies that require ongoing governance, safety monitoring, and regulatory compliance controls.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.84 Billion |
| Market Size in 2026 | USD 2.58 Billion |
| Market Size by 2035 | USD 55.00 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 40.46% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Component, Deployment Mode, Application, End User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segmental Insights
Component Insights
Which Component Segment Dominated the Clinical AI Model Governance Market?
The software platforms segment dominated the market in 2025, as the growing number of healthcare organizations are turning to integrated digital systems to oversee more complex clinical algorithms. These systems facilitate automatic performance and audit trails needed in regulated healthcare settings.
Increased use of machine learning in electronic health record systems has further boosted the use of these systems. Furthermore, enterprise healthcare institutions increasingly prioritize scalable governance systems that manage AI models across multiple departments and clinical workflows.
The services segment is expected to grow at the fastest CAGR in the coming years. Owing to the growing demand for specialized expertise to implement and manage clinical AI governance frameworks.
The implementation of AI governance software is associated with complicated tasks, such as risk analysis, planning regulatory compliance, and ongoing monitoring of performance. Furthermore, managed services and support positions are also expected to grow at a high rate as the healthcare systems scale their AI implementation in clinical processes.
Deployment Mode Insights
Why Did the Cloud-Based Segment Dominate the Clinical AI Model Governance Market?
The cloud-based segment registered its dominance in the market in 2025 and is expected to expand rapidly in the market in the coming years, due to the high need for scalable infrastructure to monitor huge numbers of clinical AI models. Hospitals and research networks have various algorithms in imaging diagnostics, predictive analytics , and clinical decision systems. The use of cloud platforms offers central environments to support the continuous monitoring and validation of records.
The increasing amount of electronic health record-generated and connected medical device-generated digital health data is also expected to drive the need for scalable cloud governance environments. The Organisation for Economic Co-operation and Development claims that over 38 nations had already implemented national AI strategies by 2024. Moreover, the decrease in the complexity of infrastructure and the ability to scale AI monitoring functions quickly are expected to fuel the segment growth in the coming years.
Application Insights
Which Application Segment Led the Clinical AI Model Governance Market?
The risk and compliance management segment led the market in 2025, as healthcare regulators increasingly emphasized the responsible and safe deployment of clinical AI systems.
Compliance-related governance platforms allow hospitals to record the development of algorithms and monitor regulatory approvals within clinical settings. Furthermore, regulatory trends have increased the need to rely on governance platforms based on compliance in hospitals, research institutions, and digital health developers.
The model monitoring and performance management segment is expected to show the fastest growth over the forecast period, owing to the fact that clinical AI models continue to be introduced into hospital routines and diagnostic systems. Monitoring solutions aid automated performance validation and assist clinical teams in identifying algorithmic bias or reliability issues during implementation.
Research into AI lifecycle monitoring models to be deployed in a healthcare system is still being pursued in academic research institutions. Such frameworks focus on ongoing assessment, dataset management, and automatic retraining pipelines that facilitate safe AI deployment, thus facilitating the segment's growth.
End User Insights
How the Hospitals and Health Systems Segment Dominated the Market?
The hospitals and health systems segment held the largest revenue share in the clinical AI model governance market in 2025, as clinical AI applications are primarily deployed within hospital care environments. Medical services are becoming increasingly interested in AI systems in radiological analysis, clinical decision-making, and risk prediction of multiple diseases.
Governance platforms thus contribute towards monitoring the safety, accuracy, and ethical responsibility of algorithms in complicated medical systems in hospitals. Hospitals thus have high investment in governance platforms that document the AI lifecycle management from development to clinical deployment.
The pharmaceutical and biotechnology companies segment is expected to gain the highest market share between 2026 and 2035, as AI increasingly supports advanced drug discovery and biomedical research programs. AI-based governance platforms are used to enable pharmaceutical companies to monitor model training data and provide scientific reproducibility in research pipelines.
The World Economic Forum believes that AI technologies are changing the process of drug discovery by increasing the possibility of computational analysis of biological data. Furthermore, biotechnology firms also raise the adoption rate since AI-based studies result in complex biological data that require constant follow-up and verification.
Regional Insights
What Positioned North America as the Dominant Region in the Clinical AI Model Governance Market?
North America held a major revenue share in the market in 2025, due to a strongly developed healthcare regulatory structure and digital healthcare ecosystems to ensure the deployment of AI in a responsible way. Such measures are projected to increase the pace of governance systems that oversee algorithm verification and clinical safety.
Effective regulatory control has prompted healthcare institutions to implement AI governance platforms. The regional market growth is further supported by standards and ethical systems that direct the implementation of clinical AI, which are actively advanced by professional bodies like the American Medical Informatics Association.
U.S. Driving Clinical AI Governance Adoption in North America
U.S. leads the market, driven by the presence of an advanced healthcare ecosystem. AI tools are being implemented to a greater extent in hospitals, academic research institutions, and health technology companies to aid in workflow optimization, predictive analytics, and diagnostics.
Based on the latest studies on healthcare adoption, 86% of U.S. healthcare organizations used AI technologies by 2024, which indicates a high pace of AI implementation in medical systems. These research, regulatory, and technological projects enhance the leadership of the U.S. in the development of AI governance infrastructure.
How is Asia-Pacific Growing in the Clinical AI Model Governance Market?
Asia-Pacific is expected to experience the fastest growth during the predicted timeframe, as healthcare systems of the countries in the region are quickly developing digital health and AI infrastructure. Governments are engaging in the active development of AI technology to overcome the shortage of workforce and the increase in disease burden in healthcare.
China, Japan, South Korea, and India are among the countries that are making national investments in AI-enabled healthcare technologies and medical data platforms. According to the World Health Organization, multiple Asia-Pacific countries strengthened national digital health strategies during 2024 and 2025.
China Emerging as a Major Growth Engine for Clinical AI Governance in Asia-Pacific
China is leading the charge in the Asia-Pacific market, as large hospitals are increasingly utilizing AI platforms to aid medical imaging , pathology diagnostics, and instruments of clinical decision support. The extreme pace of AI use in hospitals has thus prompted the need for governance systems to oversee the performance of the algorithms and their ethical adherence. The results of the research indicate that AI systems have grown rapidly in tertiary hospitals of China since 2025 and have enhanced the diagnostic process and clinical decision support system.
Clinical AI Model Governance Market Supply Chain Analysis
- Healthcare Data Acquisition & Curation
The value chain begins with the collection and preparation of high-quality healthcare datasets such as electronic health records, medical imaging, and clinical trial data used for AI model development.
Key Players: Epic Systems, Oracle Health, Illumina, Thermo Fisher Scientific.
- AI Model Development & Training
Technology companies and research institutions use curated healthcare datasets to develop and train clinical AI algorithms for diagnostics, predictive analytics, and clinical decision support systems.
Key Players: Google Health, Microsoft, NVIDIA, IBM.
- AI Model Governance & Validation Platforms
Specialized governance platforms monitor algorithm performance, detect bias, ensure transparency, and manage the lifecycle compliance of AI models deployed in healthcare environments.
Key Players: SAS Institute, Databricks, Palantir Technologies, IBM.
- Clinical Deployment & Integration in Healthcare Systems
Validated AI models and governance platforms are integrated into hospital IT infrastructure and diagnostic platforms to support safe, compliant, and real-time clinical decision-making.
Key Players: Siemens Healthineers, GE HealthCare, Philips Healthcare, Medtronic.
Clinical AI Model Governance Market Companies
Recent Developments
- In March 2026, Alation introduced Curation Automation, a new capability designed to automate metadata governance using agentic AI. The feature reduces manual data management work by automatically enforcing metadata standards across enterprise data assets. Alongside this launch, the company unveiled an outcome-based governance framework that integrates Curation Automation with its CDE Manager and Data Quality tools.
- In November 2025, the Ministry of Electronics and Information Technology introduced the India AI Governance Guidelines under the IndiaAI Mission. The framework aims to promote safe, inclusive, and responsible deployment of AI technologies across multiple industries. Government officials described the guidelines as a major policy milestone supporting India's broader strategy for ethical AI adoption.
Segments Covered in the Report
By Component
- Software Platforms
- Services (Consulting, Implementation, Monitoring & Support)
By Deployment Mode
- Cloud-Based
- On-Premises
- Hybrid
By Application
- Model Validation and Testing
- Risk & Compliance Management
- Model Monitoring and Performance Management
- Data Governance and Lifecycle Management
By End User
- Hospitals and Health Systems
- Pharmaceutical and Biotechnology Companies
- Clinical Research Organizations (CROs)
- Health Insurance Providers
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
