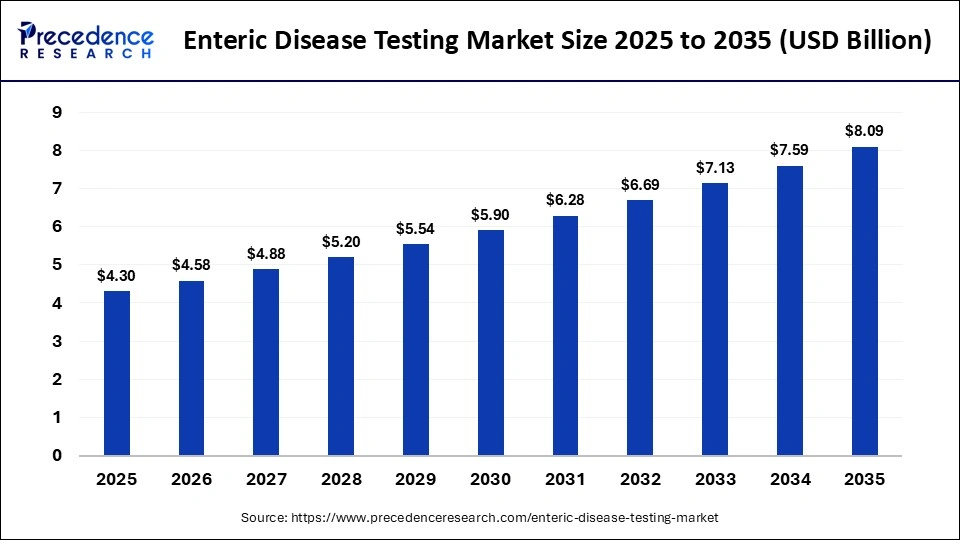
What is the Enteric Disease Testing Market Size in 2026?
The global enteric disease testing market size accounted for USD 4.30 billion in 2025 and is predicted to increase from USD 4.58 billion in 2026 to approximately USD 8.09 billion by 2035, expanding at a CAGR of 6.52% from 2026 to 2035. The increasing cases of cryptosporidiosis in the African region, coupled with the rapid investment in expanding accessibility to healthcare services, have boosted the market expansion.
Key Takeaways
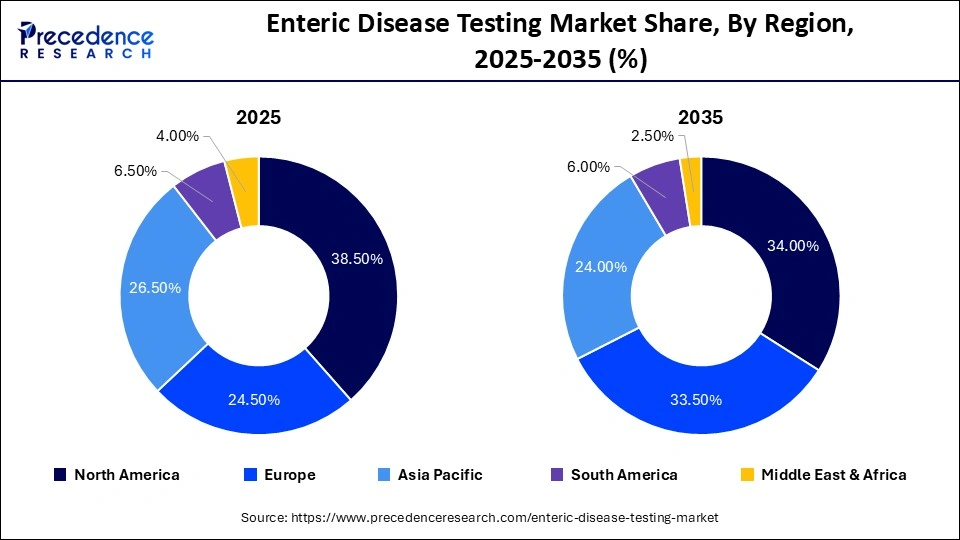
- North America led the market with a share of 38.5% in 2025.
- Asia Pacific is expected to grow at the fastest rate during the forecast period.
- By disease type, the bacterial enteric disease segment held the largest market share of 55.1% in 2025.
- By disease type, the viral enteric disease segment is the second-largest shareholder and is expected to grow at the fastest rate between 2026 and 2035.
- By technology, the molecular diagnostics segment dominated the market with a share of 45% in 2025.
- By product and service, the reagents & consumables segment dominated the market in 2025.
- By product and service, the software & services segment is the second-largest shareholder and is expected to grow with a considerable CAGR during the forecast period.
- By end-user, the hospital laboratories segment led the market in 2025.
- By end-user, the pharmaceutical & biotechnology companies segment is the second-largest shareholder in the market and is expected to grow with a robust CAGR during the forecast period.
Market Overview
The enteric disease testing market deals with companies that are involved in the production and distribution of advanced solutions for diagnosing various types of enteric diseases. Enteric disease testing helps in the identification of numerous types of microorganisms, such as bacteria, viruses, and parasites that cause gastrointestinal infections. It involves several processes, ranging from traditional cultures to advanced PCR techniques, that are essential to diagnose causes of diarrhea and vomiting, along with enhancing the treatment methodology. This market is expected to attain immense growth with the development of the healthcare sector globally.
How is AI Impacting the Enteric Disease Testing Market?
AI plays a significant role in shaping the enteric disease testing market. AI helps in analyzing stool samples for parasitic infections by scanning slides, which enhances testing speed and accuracy as compared to manual microscopic examination. In addition, AI algorithms simplify complex data from surface-enhanced Raman spectroscopy (SERS), fluorescence, and electrochemical biosensors. Moreover, AI models can process large datasets from hospital records, epidemiological sources, and geographic information systems (GIS) to track pathogen spread and predict infection outbreaks.
- In September 2025, Olympus launched OLYSENSE CAD/AI. It is an AI-based platform used to enhance clinical outcomes, enable earlier detection, and ultimately improve patient care.
Enteric Disease Testing Market Trends
- Collaborations: Several diagnostics chains are collaborating with equipment manufacturers to deploy advanced equipment for enhancing diagnostics capabilities.
- Establishment of New Laboratories: Private laboratories are expanding their branches in different regions to deliver superior testing facilities.
- Government Investments: Governments of various nations, such as India, China, Canada, and the UK, are investing heavily in opening laboratories in underserved areas to enhance accessibility.
- Product Launches: Market players are constantly launching a wide range of diagnostic products for testing enteric diseases.
- Surging Prevalence of Cholera: The cases of cholera have grown significantly in several regions, which increases the application of immunoassays for enhancing detection methodologies.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 4.30 Billion |
| Market Size in 2026 | USD 4.58 Billion |
| Market Size by 2035 | USD 8.09 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 6.52% |
| Dominating Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Disease Type, Technology, Product & Service, End-User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Increasing Prevalence of Gastrointestinal Infections
One key driver of the market is the rising prevalence of gastrointestinal infections worldwide. Increasing cases of bacterial, viral, and parasitic infections, particularly in developing regions with limited sanitation, are driving demand for rapid and accurate diagnostic tests. Early detection through advanced testing helps in timely treatment, reducing complications and the spread of infections, which further fuels market growth.
Restraint
High Cost of Molecular Testing and Regulatory Hurdles
The enteric disease testing industry encounters several challenges. Firstly, the cost of molecular testing is increasing rapidly, which deters people from adopting these testing processes. Secondly, various regulatory issues related to enteric disease testing in several regions have negatively impacted the industry.
Opportunity
Advancements in CRISPR-Based Diagnostics
Biotech companies are investing rapidly in advancing several CRISPR-based technologies, such as SHERLOCK (Cas13) and DETECTR (Cas12). These technologies help in delivering attomolar sensitivity and provide results within an hour. Thus, rapid technological advancements in CRISPR-based diagnostics solutions are expected to create ample growth opportunities for the market players in the future.
- In September 2025, VedaBio partnered with Mammoth Biosciences Technology. This partnership aims to expand the CRISPR-based molecular detection platform globally.
Segment Insights
Disease Insights
Enteric Disease Testing Market Share, By Disease Type, 2025-2035 (%)
| Disease Type | 2025 | 2035 |
| Bacterial | 55.10% | 58.50% |
| Viral | 28.40% | 26.00% |
| Parasitic | 16.50% | 15.50% |
The Bacterial Enteric Disease Segment Held a Market Share of 55.1 % in 2025
The bacterial enteric disease segment dominated the enteric disease testing market with the largest share of 55.1% in 2025. This is due to the surging transmission rate of Salmonella, E. coli, and Shigella. These diseases cause major foodborne and waterborne diseases in developing nations due to inadequate sanitation infrastructure. The growing nature of bacterial infections leads to several gastrointestinal disorders, which increases the demand for accurate diagnostic testing.
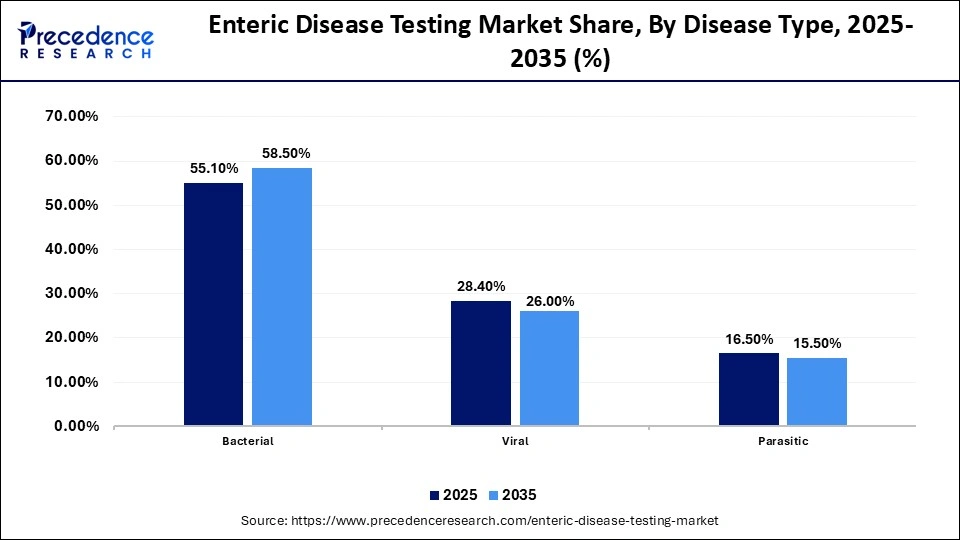
The viral enteric disease segment held a 28.4% share of the market in 2025 and is expected to grow at the fastest rate in the coming years. This is mainly due to the growing transmission of several viral pathogens, including rotavirus, norovirus, and adenovirus. These viral infections lead to acute gastroenteritis in children, which increases the need for molecular diagnostics technologies. Nowadays, hospitals are prioritizing viral testing solutions for timely intervention and accurate diagnostic capabilities.
The parasitic enteric disease segment held a share of 16.5% in the market in 2025. This is due to the increasing burden of parasitic diseases in tropical regions, which have limited access to clean water. Several types of diseases are caused by parasites, including Giardia, Entamoeba histolytica, and Cryptosporidium. These diseases increase the application of specialized diagnostic technologies such as antigen-based assays and microscopy in modern laboratories.
Technology Insights
Enteric Disease Testing Market Share, By Technology, 2025-2035 (%)
| Technology | 2025 | 2035 |
| Molecular | 45.00% | 52.00% |
| Immunoassays | 30.00% | 28.50% |
| Traditional | 25.00% | 19.50% |
The Molecular Diagnostics Segment Held a Share of 45.0% in 2025
The molecular diagnostics segment dominated the enteric disease testing market with a share of 45% in 2025. This is due to its enhanced accuracy, high sensitivity, and low turnaround time as compared to conventional diagnostic solutions. In addition, the demand for PCR panels has grown significantly as they are used to identify multiple pathogens in one stool sample. The segment's dominance is also reinforced by the increased use of Polymerase Chain Reaction (PCR) by hospital laboratories to identify bacterial and viral pathogens.
The immunoassays segment held a share of 30% in the market in 2025. This is mainly due to their rapid diagnostics capabilities and cost-effectiveness, which make them suitable for smaller clinics and PoC settings. Immunoassay-based tests, including ELISA and lateral flow assays, are increasingly used for detecting antigens related to bacterial pathogens.
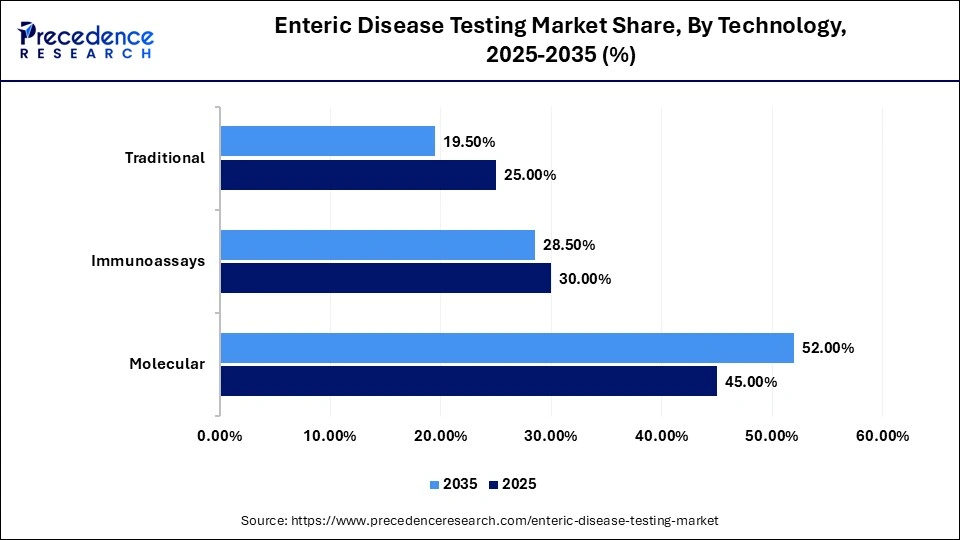
The traditional culture methods segment held a share of 25.0% in the market in 2025. This is due to their rising application in antimicrobial susceptibility testing and pathogen identification. Traditional culture methods are used in the determination of causative organisms and deliver appropriate antibiotic treatments. These testing methods are widely adopted by private laboratories due to their regulatory approval and lower cost.
The chromatography & spectrometry segment held a notable share of the market in 2025. This is due to its superior sensitivity, high precision, and the ability to deliver detailed analysis of the pathogens and toxins. Advancements in gas chromatography and liquid chromatography enable accurate identification of biomarkers related to enteric infections.
Product & Service Insights
The Reagents & Consumables Segment Held a Dominant Share of the Market in 2025
The reagents & consumables segment dominated the enteric disease testing market with the largest share in 2025. This is due to their essential role in diagnostic centers for testing a wide range of viral diseases. Molecular diagnostics tests require several types of reagents, including culture media, assay kits, and buffers. Market players are engaged in the development of advanced, highly sensitive consumables for improving testing efficiency in laboratories. The use of reagents and consumables in diagnostic centers has grown significantly to detect, measure, and analyze several types of samples, including blood, urine, and tissue, for disease monitoring, diagnosis, and treatment planning.
- In November 2025, Synthego launched a new range of molecular reagents for diagnostics & research. This new range of reagents is engineered to deliver the performance, precision, and reliability required to advance molecular biology and enhance innovation in the field of clinical and molecular diagnostics.
The instruments/equipment segment held a considerable share of the market in 2025. This is due to the surging use of advanced equipment for enabling automated testing. Various types of instruments, such as PCR analyzers, immunoassay platforms, and chromatography systems, are used in the laboratories to enhance diagnostic capabilities.
The software & services segment held a notable share of the market in 2025. This is due to the rising need for efficient and superior data management software and advanced analytics solutions from the diagnostic laboratories. The software enables real-time data tracking, enhances decision-making capabilities, and integrates with diagnostic equipment.
End-User Insights
The Hospital Laboratories Segment Held the Dominant Share of the Market in 2025
The hospital laboratories segment dominated the enteric disease testing market with a major share in 2025. This is because of the increased preference of patients to visit hospitals for diagnosing infectious diseases. Hospital laboratories are equipped with advanced diagnostic capabilities, which enable rapid pathogen detection. Rapid investment by hospitals in developing sophisticated laboratories to ensure timely diagnosis has driven segmental expansion.
The pharmaceutical & biotechnology companies segment held a significant share of the market in 2025 and is expected to grow at a significant rate in the upcoming period. This is because these organizations are increasingly investing in enteric disease research, drug development, and clinical trials. Their focus on developing targeted therapies and vaccines drives demand for advanced diagnostic testing to support pipeline development and regulatory approvals.
The diagnostic centers segment held a notable share of the market in 2025. This is due to the rapid expansion of the diagnostic centers in urban areas across several countries, such as New Zealand, China, and Canada. Diagnostic centers are collaborating with equipment manufacturers to deploy high-quality instruments for enhancing diagnostic capabilities, ensuring the long-term growth of the segment.
The academic & research institutes segment held a considerable share of the market in 2025. This is mainly due to a strong emphasis of these institutes on developing advanced treatment methods for treating infectious diseases. Additionally, these institutions rely heavily on advanced diagnostic testing for experimental studies, clinical trials, and validation of new treatments.
Regional Insights
North America Enteric Disease Testing Market Size and Growth 2026 to 2035
The North America enteric disease testing market size is estimated at USD 1.66 billion in 2025 and is projected to reach approximately USD 3.16 billion by 2035, with a 6.65% CAGR from 2026 to 2035.
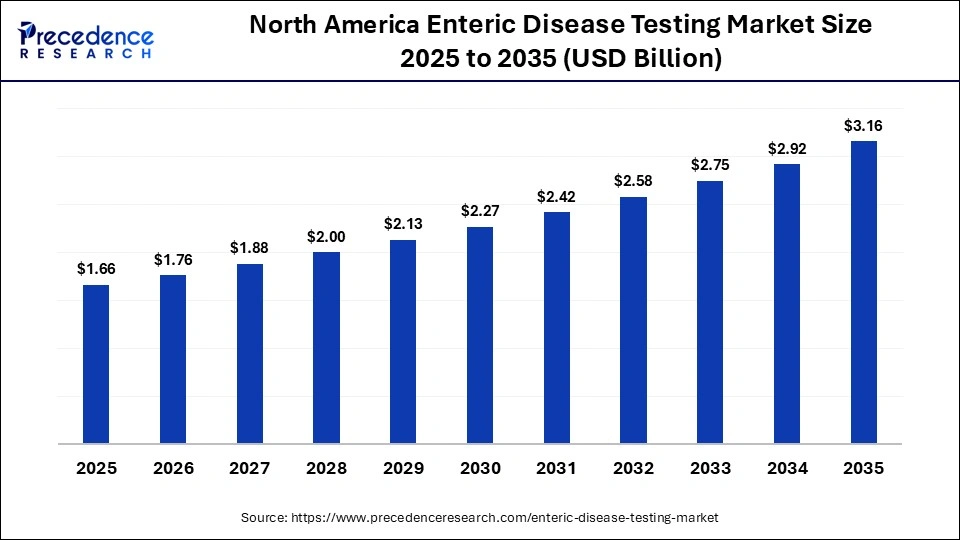
North America Held a 38.5% Share of the Enteric Disease Testing Market in 2025
North America dominated the enteric disease testing market while holding a major share of 38.5% in 2025. This is mainly due to the strong emphasis of the government on strengthening the biotechnology sector, coupled with the surging adoption of stringent CDC/FDA food safety regulations. The availability of advanced diagnostic infrastructure, as well as the rapid investment in infectious disease tracking, has contributed to the development of the market. The region is home to several prominent players, such as Hologic, Inc, Neogen Corporation, Thermo Fisher Scientific Inc., and Bio-Rad Laboratories, Inc, who are engaged in launching a wide range of solutions to enhance the testing of enteric diseases, which propels the market growth.
- In January 2025, Neogen launched MDA2QSAL96. MDA2QSAL96 is a molecular detection assay designed for testing Salmonella-based diseases in the U.S.
U.S. Enteric Disease Testing Market Size and Growth 2026 to 2035
The U.S. enteric disease testing market size is calculated at USD 1.24 billion in 2025 and is expected to reach nearly USD 2.38 billion in 2035, accelerating at a strong CAGR of 6.74% between 2026 and 2035.
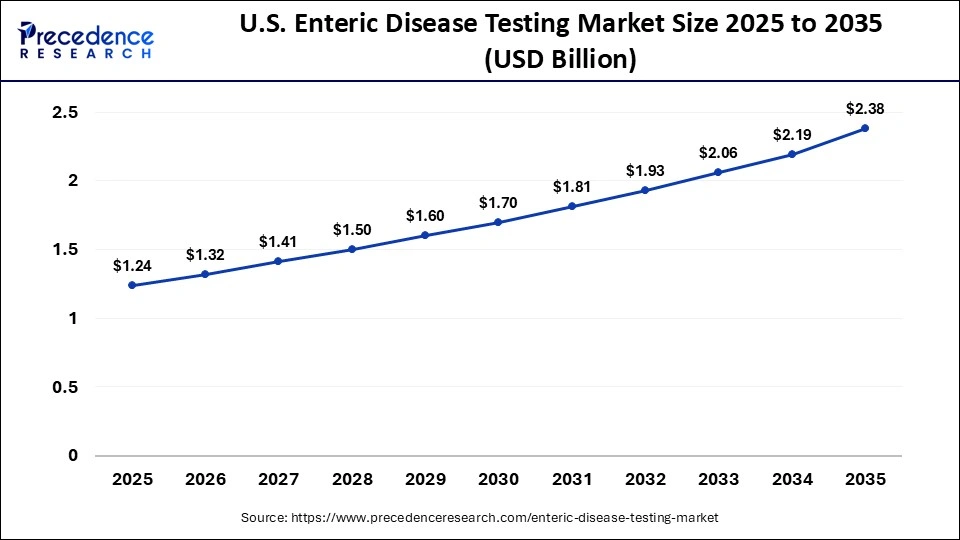
U.S. Enteric Disease Testing Market Analysis
U.S. leads the North American market, driven by the rapid advancements in the biotechnology sector, coupled with the surging adoption of molecular diagnostics solutions in diagnostic centers. The growing pool of patients suffering from cryptosporidiosis, as well as the presence of several research and academic institutions, has driven the market expansion.
How is the Opportunistic Rise of Asia Pacific in the Enteric Disease Testing Market?
Asia Pacific held a 24.5% share of the market in 2025 and is expected to grow at the fastest rate during the forecast period. This is mainly due to the growing prevalence of infectious diseases in China and India, along with the strong emphasis on public health surveillance. Companies, such as InfeXn Laboratories, Mindray Medical, and Autobio Diagnostics, are engaged in delivering high-quality molecular, immunoassay, and automation solutions to cater to the end-users. Numerous government policies aimed at establishing new laboratories, coupled with the surging investments in the healthcare sector by private players, are anticipated to propel market growth.
- In August 2025, Mindray launched CL-8000i. CL-8000i is a chemiluminescence immunoassay analyzer designed to improve the working efficiency of modern laboratories in the APAC region.
China Enteric Disease Testing Market Analysis
China is a major contributor to the Asia Pacific enteric disease testing market due to the surging prevalence of viral diseases, coupled with the rise in the number of private laboratories. The integration of advanced technologies in the biotech sector, as well as the presence of various medical healthcare equipment manufacturers, is driving the market growth.
Supply Chain Analysis of Enteric Disease Testing Solutions
- Raw Materials Procurement
Raw materials used in the manufacturing of enteric disease testing equipment comprise biologic components, substrates, polystyrene, and UV-curable adhesives.
Key Companies: Alpek, Sabic, and Synthos.
- Quality Control
Quality control (QC) of enteric disease testing equipment is a multi-layered process designed to enhance accuracy, sensitivity, and specificity in the detection of diseases.
Key Companies: Pro QC International, Element Materials Technology, and TUV SUD.
- Distribution Channel
The distribution channels for enteric disease testing equipment are operated through direct-to-customer methods and indirect networks designed to reach hospitals, clinical laboratories, and food safety testing centers.
Key Companies: Abbott, Becton Dickinson (BD), and bioMErieux.
Enteric Disease Testing Market Companies
- Abbott Laboratories
- Becton, Dickinson and Company (BD)
- bioMérieux SA
- Danaher Corporation (Cepheid)
- Thermo Fisher Scientific Inc.
- Bio-Rad Laboratories, Inc.
- Roche Holding AG
- Hologic, Inc.
- QuidelOrtho Corporation
- PerkinElmer, Inc.
- Neogen Corporation
- SGS SA
- QIAGEN N.V.
- DiaSorin S.p.A
- Meridian Bioscience, Inc
Recent Developments
- In March 2026, Roche launched the Cobas MPX-E assay. Cobas MPX-E assay is an advanced testing solution designed to detect pathogens and allows diagnostic centers to implement testing of Hepatitis E (HEV) screening efficiently.(Source: https://www.globenewswire.com)
- In October 2025, Hologic, Inc. made an announcement stating that the U.S. Food and Drug Administration (FDA) had approved Panther Fusion gastrointestinal (GI) bacterial and expanded bacterial assays. This testing methodology is designed to detect bacterial pathogens that cause infectious gastroenteritis.(Source: https://www.hologic.com)
- In August 2025, Bio-Rad Laboratories, Inc. launched EZ-Check Salmonella spp. Kit. This new range of testing kits is designed for the detection of salmonellosis.(Source: https://www.bio-rad.com)
Segments Covered in the Report
By Disease Type
- Bacterial Enteric Disease (C. Difficile, Salmonella, E. Coli, Campylobacter, H. Pylori, Cholera, Shigella)
- Viral Enteric Disease (Norovirus, Rotavirus, Adenovirus)
- Parasitic Enteric Disease (Amebiasis, Cryptosporidiosis, Giardiasis)
By Technology
- Molecular Diagnostics (PCR, NAAT, NGS)
- Immunoassays (ELISA, Rapid Antigen Tests)
- Traditional Culture Methods
- Chromatography & Spectrometry
By Product & Service
- Reagents & Consumables
- Instruments/Equipment
- Software & Services
By End-User
- Hospital Laboratories
- Diagnostic Centers
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutes
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
