What is the Fallopian Tube Cancer Therapeutics Market Size in 2026?
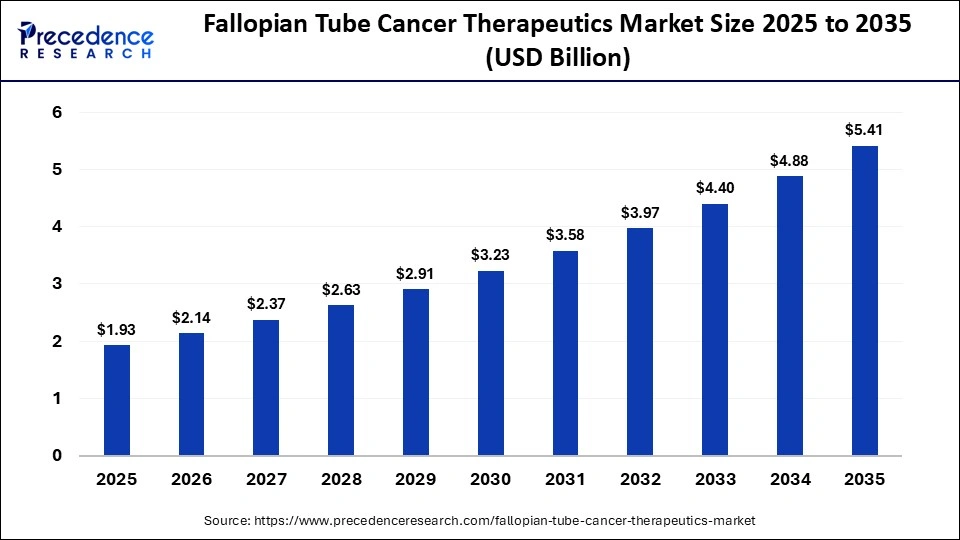
The global fallopian tube cancer therapeutics market size was calculated at USD 1.93 billion in 2025 and is predicted to increase from USD 2.14 billion in 2026 to approximately USD 5.41 billion by 2035, expanding at a CAGR of 10.86% from 2026 to 2035. The market is witnessing substantial growth due to a rapid shift toward targeted, personalized, and minimally invasive treatments that improve patient survival and quality of life.
Key Takeaways
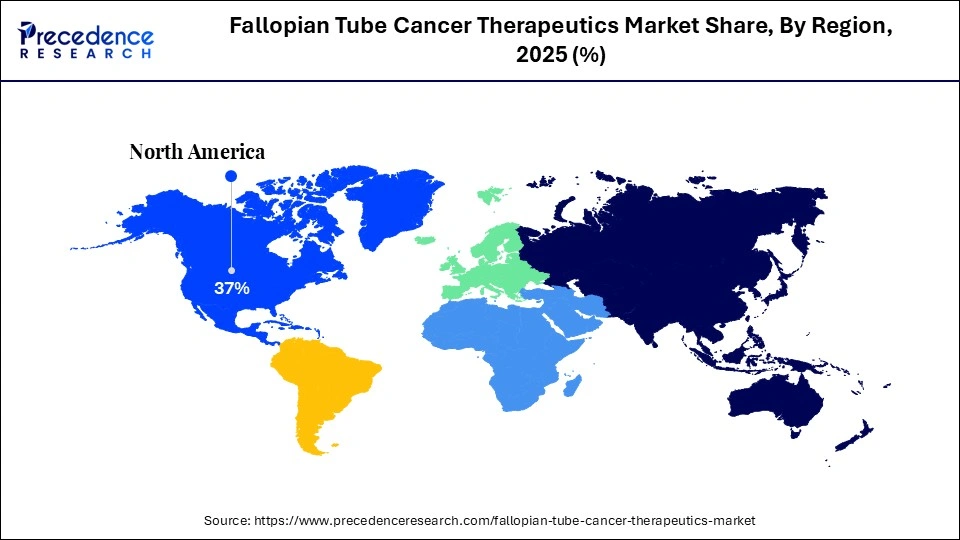
- North America dominated the market with a major market share in 2025.
- Asia Pacific is expected to grow at the fastest CAGR between 2026 and 2035.
- By treatment type, the chemotherapy segment contributed the highest market share in 2025.
- By treatment type, the targeted therapy segment is expected to grow at a strong CAGR between 2026 and 2035.
- By stage, the stage I segment held a major market share in 2025.
- By stage, the stage II segment is expected to expand at a notable CAGR from 2026 to 2035.
- By end-user, the hospitals segment captured the highest market share in 2025.
- By end-user, the specialty clinics segment is poised to grow at a healthy CAGR between 2026 and 2035.
Market Overview
The global fallopian tube cancer therapeutics market includes drugs, therapies, and diagnostic technologies aimed at managing and treating cancers that originate in the fallopian tubes. This market focuses on diagnosing and treating primary malignant neoplasms of the fallopian tubes through targeted therapies such as PARP inhibitors, chemotherapy, and emerging immunotherapies. The growth of this market is driven by increasing awareness, a rising incidence of the disease, and advancements in personalized medicine .
How is AI Transforming the Fallopian Tube Cancer Therapeutics Market?
Artificial intelligence (AI) is transforming the fallopian tube cancer therapeutics market. It accelerates drug discovery, enables personalized medicine through biomarker identification, and optimizes clinical trial designs. AI analyzes complex datasets to identify novel therapeutic targets, reducing discovery timelines. It also examines genetic and molecular profiles to identify biomarkers that predict patient responses to therapies, enhancing personalized treatment options. Furthermore, AI-based high-resolution imaging improves tumor detection, segmentation, and classification, allowing for better staging and monitoring of therapies
Major Trends in the Fallopian Tube Cancer Therapeutics Market
- Rise of Targeted Therapies: The adoption of PARP inhibitors is transforming treatment, especially for patients with BRCA mutations. These therapies provide maintenance treatments that lead to improved survival rates with fewer side effects compared to traditional chemotherapy.
- Expansion of Immunotherapy and Antibody-Drug Conjugates: Immunotherapy, including checkpoint inhibitors, is gaining popularity for advanced or resistant cases of fallopian tube cancer.
- Advanced Diagnostic Technologies and Early Detection: There is a strong emphasis on utilizing liquid biopsies and next-generation sequencing to detect tumors earlier, which improves patient stratification and guides treatment decisions.
- Shift to Personalized and Multidisciplinary Care: Treatment is increasingly personalized based on molecular profiling. This approach involves multidisciplinary teams, including surgeons, oncologists, and geneticists, providing comprehensive, tailored care that is becoming standard practice.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.93 Billion |
| Market Size in 2026 | USD 2.14 Billion |
| Market Size by 2035 | USD 5.41 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 10.86% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Treatment Type, Stage, End-User, and region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Treatment Type Insights
What Made Chemotherapy the Dominant Segment in the Fallopian Tube Cancer Therapeutics Market?
The chemotherapy segment dominated the market in 2025, primarily due to its role as the standard first-line treatment for gynecological cancers. This approach has been the cornerstone of care in this field for decades, owing to proven clinical outcomes and its widespread use, especially in combination with surgical procedures. The integration of traditional chemotherapy with newer targeted therapies has improved treatment efficacy. Chemotherapy remains the most accessible, cost-effective, and standardized treatment worldwide, used both before surgery to shrink tumors and afterward to eliminate residual cancer cells, thereby reducing recurrence rates.
The targeted therapy segment is expected to experience the fastest growth during the forecast period. The growth of the segment is driven by the high efficacy of PARP inhibitors in treating specific genetic mutations. Targeted therapies are designed to specifically inhibit the function of cancer-causing genes or proteins, offering higher success rates. The increasing use of next-generation sequencing and liquid biopsies allows clinicians to identify actionable mutations earlier. A surge in clinical trials focusing on targeted combinations and antibody-drug conjugates leads to the new, more effective treatment options.
Stage Insights
How Did the Stage I Segment Lead the Fallopian Tube Cancer Therapeutics Market?
The stage I segment led the market in 2025, primarily due to improved early detection of tumors before they spread, facilitated by advanced imaging, proactive genetic testing, and surgical screening. Early diagnosis allows for higher-margin, localized treatments. Increased awareness and advanced diagnostic imaging have led to a higher identification rate of localized, early-stage tumors. Additionally, the rise in prophylactic salpingectomies for high-risk, BRCA-positive patients often uncovers early-stage cancer, driving therapeutic intervention at this stage. This early treatment offers better survival rates and greater patient demand for curative, specialized therapies.
The stage II segment is expected to grow at a significant rate during the forecast period, driven by the increasing number of patients diagnosed at a moderately advanced stage, which requires a combination of surgery and systemic therapies. The adoption of advanced diagnostics, including molecular diagnostics and imaging, enables better identification of cancers in intermediate stages like stage II, ensuring improved management and higher utilization of combined treatment modalities for these patients. Additionally, rising awareness and early detection efforts are enabling timely intervention, further supporting the growth of Stage II treatments.
End-User Insights
Why Did the Hospitals Segment Dominate the Fallopian Tube Cancer Therapeutics Market?
The hospitals segment dominated the market in 2025, mainly due to high patient influx, specialized care, and advanced treatment infrastructure. Hospitals serve as the primary, often exclusive, setting for comprehensive cancer management, including complex surgical procedures and intensive multidisciplinary inpatient care. They also integrate advanced diagnostic imaging and molecular diagnostic tools to identify BRCA mutations, facilitating personalized and targeted treatments. Rapid investment in healthcare infrastructure and increased cancer awareness in emerging economies are contributing to higher patient turnover.
The specialty clinics segment is projected to grow at the fastest rate during the forecast period, primarily due to the increasing adoption of personalized medicine, targeted therapies, and the demand for specialized care. Specialty clinics are enhancing their capabilities with sophisticated diagnostic tools, such as molecular diagnostics, liquid biopsies , and next-generation sequencing. The growing focus on molecular diagnostics and tailored therapies necessitates specialized expertise, which is often found in specialized centers. Additionally, the trend toward outpatient and supportive care promotes faster recovery and improved quality of life, further driving demand.
Regional Insights
How Did North America Dominate the Fallopian Tube Cancer Therapeutics Market?
North America led the market while holding the largest share in 2025, primarily due to a high prevalence of disease, advanced healthcare infrastructure, significant research and development investments, and strong adoption of personalized medicine. Major government agencies like the National Cancer Institute, along with private investments, have accelerated the development and approval of new therapeutics. A large patient population and heightened awareness of gynecological cancers in the U.S. and Canada are boosting demand. The widespread availability of cutting-edge diagnostic tools enables earlier, more accurate detection, allowing patients access to high-cost, specialized, and targeted therapies.
U.S. Fallopian Tube Cancer Therapeutics Market Trends
The U.S. plays a dominant role within the region, mainly due to advanced healthcare infrastructure, substantial research investment, and early adoption of novel therapies. Major pharmaceutical companies are concentrating on targeted therapies like PARP inhibitors, immunotherapy, and precision medicine . The FDA establishes global standards for drug approval, while institutions drive clinical trials focused on advanced diagnostics, emphasizing patient-centric care and digital health integration .
Why is Asia Pacific Considered the Fastest-Growing Region in the Fallopian Tube Cancer Therapeutics Market?
Asia Pacific is expected to experience the fastest growth during the forecast period. This growth is primarily driven by a large, increasing patient population, greater adoption of PARP inhibitors and targeted therapies, and enhanced diagnostic infrastructure. Significant investments in oncology are shifting from generics to innovative biopharmaceutical development , particularly in China and India. High population density in this region allows for quicker recruitment for clinical trials compared to Western countries. Additionally, there is a growing focus on women's health, heightened cancer awareness, and improved reimbursement policies for cancer drugs.
India Fallopian Tube Cancer Therapeutics Market Trends
India is emerging as a key player in the region, supplying cost-effective generic drugs and biosimilars that enhance access to advanced treatments. There has been a notable increase in clinical trials and research partnerships targeting therapies and PARP inhibitors, aligning with global standards. Additionally, investments in oncology infrastructure, including minimally invasive surgical techniques, are rising. Government initiatives aim to improve awareness and provide subsidized screenings for cancer.
How is the Opportunistic Rise of Europe in the Fallopian Tube Cancer Therapeutics Market?
Europe is a notably growing region in the market, mainly driven by high disease prevalence, significant investment in PARP inhibitors and immunotherapy, and favorable regulatory frameworks for novel treatments. Strong, centralized healthcare systems and specialized oncology centers in countries like Germany and the UK facilitate rapid adoption of new treatments. The European Medicines Agency uses specialized pathways like PRIME to speed up approvals for unmet needs. Fallopian tube cancer is recognized as a primary source of many high-grade serous ovarian cancers, leading to higher diagnosis rates.
Fallopian Tube Cancer Therapeutics Market Value Chain Analysis

Fallopian Tube Cancer Therapeutics Market companies
- AstraZeneca
- GSK
- F. Hoffmann-La Roche Ltd.
- Merck and Co., Inc.
- AbbVie
- Pfizer Inc.
- Bristol-Myers Squibb
- Janssen Pharmaceuticals
- Novartis AG
- Eli Lilly and Company
- ImmunoGen, Inc.
- Genmab A/S
- Boehringer Ingelheim GmbH
- Alkermes plc
- Clovis Oncology, Inc.
Recent Developments
- In February 2026, Merck announced FDA approval of KEYTRUDA (pembrolizumab) and KEYTRUDA QLEX™ (pembrolizumab and berahyaluronidase alfa-pmph) plus paclitaxel, with or without bevacizumab, for adults with PD-L1+ platinum-resistant ovarian, fallopian tube, or primary peritoneal carcinoma after one or two prior treatments. These approvals were based on the Phase 3 KEYNOTE-B96 trial, which showed a 28% reduction in disease progression for patients facing limited treatment choices after platinum resistance.(Source: https://www.merck.com )
- In May 2025, Verastem Oncology received FDA approval for AVMAPKI™ FAKZYNJA™ CO-PACK as the first treatment for KRAS-mutated recurrent low-grade serous ovarian cancer following systemic therapy. This accelerated approval was based on tumor response rates and offers hope for patients in need of new options.(Source: https://investor.verastem.com )
- In September 2025, the FDA granted breakthrough therapy designation to raludotatug deruxtecan (R-DXd), a potential first-in-class antibody-drug conjugate for adult patients with CDH6-expressing platinum-resistant ovarian cancer who have previously been treated with bevacizumab. The designation aims to provide new treatment avenues for patients facing limited options. (Source: https://www.onclive.com )
Segments Covered in the Report
By Treatment Type
- Chemotherapy
- Targeted Therapy
- Hormonal Therapy
- Immunotherapy
- Others
By Stage
- Stage I
- Stage II
- Stage III
- Stage IV
By End-User
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
- Others
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
