What is the Gene-Replacement Therapy Market Size?
The global gene-replacement therapy market is evolving rapidly due to improvements in viral vector delivery systems and precision medicine adoption. The market is witnessing substantial growth due to the increasing number of genetic diseases, a growing pipeline of therapies in clinical development, and advancements in vector delivery technologies. Furthermore, there is a transition from traditional treatments to more precise, one-time genetic cures and an increasing focus on therapies for rare and inherited conditions.
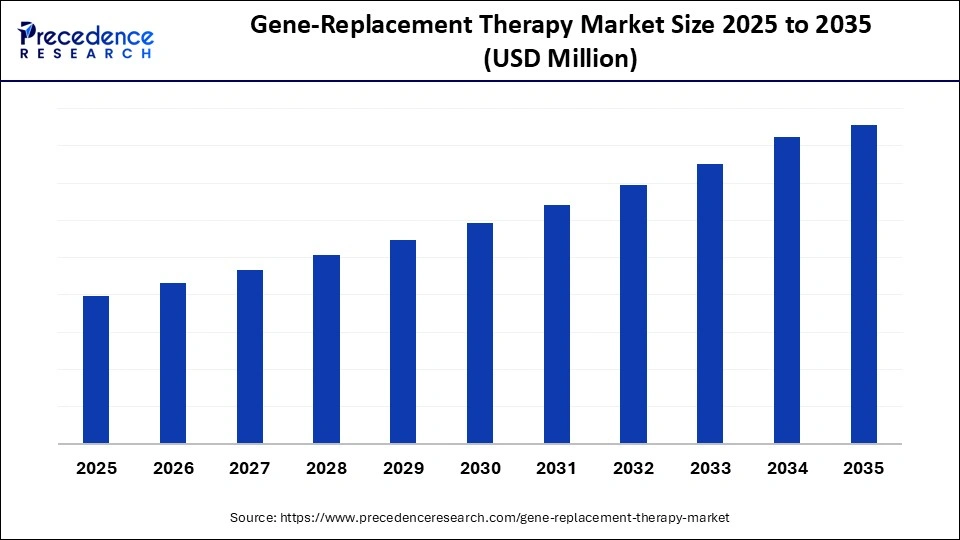
Market Highlights
- North America accounted for the largest market share in 2025.
- Asia Pacific region is expected to witness the fastest CAGR from 2026 to 2035.
- By therapy type, the viral vector-based gene replacement segment held the largest market share in 2025.
- By therapy type, the non-viral gene replacement segment is expected to grow at the fastest CAGR from 2026 to 2035.
- By disease type, the genetic disorders segment dominated in 2025.
- By disease type, the ophthalmic disorders segment is anticipated to grow at a significant CAGR from 2026 to 2035.
- By end user, the hospitals and specialty clinics segment led in 2025.
- By end user, the gene therapy centers segment is expected to witness the fastest CAGR from 2026 to 2035.
What Is the Gene-Replacement Therapy?
Gene-replacement therapy is a therapeutic approach that involves a healthy, functional copy of a defective gene into a patient's cells to restore normal cellular function, particularly for monogenic diseases caused by a single gene defect. This differs slightly from broader gene therapy, inactivating a problematic gene by introducing new genetic material to help the body fight a disease. The gene-replacement market has immense promise for providing life-saving and potentially curative treatments for previously untreatable conditions.
How Does AI Influence the Gene-Replacement Therapy Market?
Artificial intelligence (AI) is transforming the gene-replacement therapy market by accelerating discovery and development by improving gene variant analysis, enhancing the precision of gene editing tools like CRISPR, and personalizing treatment plans. AI can rapidly analyze vast genomic datasets to identify gene variants linked to diseases, predict their function, and validate the potential of therapeutic targets. AI can also streamline clinical trials by optimizing patient recruitment, improving trial protocols, and reducing development time and costs, ensuring consistent quality and making production more effective.
Market Trends in the Gene-Replacement Therapy Market
- Growing Clinical Validation of Single-Dose, Durable Therapies: Gene-replacement therapies are increasingly demonstrating long-term therapeutic effects from a single administration, shifting treatment paradigms away from chronic disease management toward one-time or infrequent interventions for monogenic disorders.
- Expanded Use of AAV and Non-Viral Delivery Platforms: Continued optimization of adeno-associated viral vectors and emerging non-viral delivery systems is improving tissue targeting, transgene expression stability, and safety profiles across neurology, ophthalmology, and rare disease indications.
- Rising Regulatory Engagement and Accelerated Approval Pathways: Health authorities are increasing use of expedited pathways such as orphan drug designation, breakthrough therapy status, and accelerated approvals to support faster patient access to gene-replacement therapies for high-unmet-need conditions.
- Scaling of Manufacturing and Process Standardization: Significant investment is being directed toward scalable vector manufacturing, quality control analytics, and process standardization to address supply constraints and reduce batch variability in commercial gene therapy production.
- Integration of Long-Term Follow-Up and Real-World Evidence Programs: Post-approval monitoring requirements are driving adoption of structured long-term safety and efficacy tracking systems, reinforcing the importance of real-world data in lifecycle management of gene-replacement therapies.
- Increasing Focus on Value-Based Pricing and Reimbursement Models: High upfront therapy costs are accelerating development of outcomes-based reimbursement frameworks, installment payment models, and risk-sharing agreements between manufacturers, payers, and healthcare systems.
Market Scope
| Report Coverage | Details |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Therapy Type, Disease Type, End User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Therapy Type Insights
How Will the Viral Vector-Based Gene Replacement Segment Lead the Gene-Replacement Therapy Market?
The viral vector-based gene replacement segment was the market leader in 2025, particularly the AAV-based therapies subsegment. This dominance is mainly due to their advantageous characteristics, including high transduction efficiency, the ability to transduce non-dividing cells, and relative safety, as they typically remain episomal. AAVs can transduce both dividing and non-dividing cells, which is crucial for treating a wide array of genetic diseases. Several recent FDA-approved gene therapies have utilized AAVs, demonstrating a clear pathway for product development and market entry, establishing AAV as a leading platform for delivering gene replacement therapies.
The non-viral gene replacement segment is expected to be the fastest-growing in this market. This growth is attributed to its superior safety, scalability, and the ability to be re-administered. Non-viral vectors can be easily modified with flexible chemical compositions, allowing them to adapt to various therapeutic needs. Innovations in non-viral vectors, such as lipid nanoparticles, have significantly improved their efficiency. Their ease of mass production and reduced immunogenicity make them an attractive alternative to viral vectors for delivering mRNA and other therapeutic payloads.
Disease Type Insights
What Made the Genetic Disorders Segment Lead the Gene-Replacement Therapy Market in 2025?
The genetic disorders segment led the market in 2025 due to the severity and progression of these disorders, which often lack effective conventional treatments that only focus on symptom management rather than addressing the root cause. Gene therapies offer the potential for a one-time, long-term curative solution to this significant medical need. Continuous advancements in gene delivery methods, particularly the use of highly efficient viral vectors like Adeno-Associated Viruses, have enhanced the safety and precision of delivering genetic material to target cells in the body.
The ophthalmic disorders segment is projected to experience the fastest growth in the global market, primarily because of the biological advantages of the eye for treatment delivery and a robust pipeline of new therapies that build on existing clinical successes. Many inherited retinal diseases are monogenic, caused by defects in a single gene. The effects and safety of ocular gene therapies can be monitored non-invasively using reliable imaging techniques, which provide clear and consistent data on treatment efficacy and progression.
End User Insights
How Did the Hospitals and Specialty Clinics Segment Dominate the Gene-Replacement Therapy Market in 2025?
The hospitals and specialty clinics segment dominated the market in 2025, due to their provision of essential infrastructure for administering complex treatments, managing patient care, and conducting monitoring. These institutions often serve as hubs for clinical trials, equipped with specialized equipment and trained medical professionals who can handle the intricate logistics involved in storing and preparing gene therapies. Specialty clinics, such as cancer centers and wound care facilities, are particularly dominant due to their expertise in specific diseases and patient populations, maintaining ethical oversight and rigorous monitoring.
The gene therapy centers segment is experiencing the fastest growth globally. This increase is driven by greater patient access due to hospital expansions, advancements in delivery methods, and supportive regulatory environments. Hospitals are expanding their facilities to provide these complex treatments, while specialized cancer centers are witnessing a rise in patient volumes due to their proven expertise. Supportive regulations and a greater focus on personalized medicine and the treatment of rare diseases are also fueling the demand for gene therapy centers, encouraging more research and facilitating faster market entry.
Regional Insights
How Did North America Dominate the Gene-Replacement Therapy Market in 2025?
North America dominated the gene replacement therapy market in 2025, due to its robust research and development infrastructure, significant investment, a favorable and proactive regulatory environment, and the presence of key biopharmaceutical companies. Major gene therapy pioneers and market leaders, including Biogen Inc., Sarepta Therapeutics, Inc., Bluebird Bio, Inc., and Spark Therapeutics, have contributed to market expansion in this region. North America benefits from high healthcare expenditure and established, albeit complex, reimbursement models, including innovative outcome-based agreements by the Centers for Medicare & Medicaid Services, which support the development of potentially curative gene therapies.
U.S. Gene-Replacement Therapy Market Trends
The U.S. leads the global gene therapy market with strong R&D, significant investment, and numerous clinical trials. Major biotech companies like Sarepta Therapeutics and Novartis drive innovation in viral vectors and CRISPR technologies. The FDA's expedited pathways allow quicker approvals for new therapies, especially for rare diseases.
Why Is Asia Pacific Considered to Be the Fastest-Growing Region in the Gene-Replacement Therapy Market in 2025?
The Asia-Pacific gene replacement therapy market is the fastest-growing worldwide, driven by a large and diverse patient population, supportive government initiatives and funding, and an increasing number of positive clinical trial results. Countries such as Japan, China, South Korea, and Australia have implemented streamlined and expedited regulatory approval pathways for regenerative medicine and gene therapies. Additionally, both governments and private companies are significantly increasing funding and investments in gene therapy research and development, fostering a thriving biotech ecosystem in the region.
India Gene-Replacement Therapy Market Trends
India is emerging as a key contributor, focusing on affordable therapies for its vast patient population, particularly for diseases like sickle cell anemia and thalassemia. Innovations, such as the affordable CAR-T cell therapy NexCAR19, exemplify the country's efforts. Government initiatives like the Make in India campaign support local manufacturing and research.
What are the Advancements in the Gene-Replacement Therapy Market in Europe?
Europe is expected to have significant market growth all throughout the forecast years, driven by advancements in regulatory frameworks, such as the European Medicines Agency's (EMA) guidelines for gene therapies. These regulations help to streamline the approval process, thus encouraging innovation and boosting investments in the sector. Countries such as Germany, France, and the UK are leading players.
Germany Market Trends
The country's market landscape is evolving rapidly, with increasing collaborations between pharmaceutical companies and research institutions. The presence of a well-established healthcare infrastructure further supports the growth of gene therapy, making it a key player in the global gene replacement therapy industry.
What are the Key Trends in the Gene-Replacement Therapy Market in Latin America?
Latin America is expected to witness significant market growth, driven by increasing investments in biotechnology, a growing patient population, and rising healthcare expenditures. Countries like Brazil and Mexico are leading players, with supportive government policies that are aimed at fostering innovation in gene therapies. The region's advanced healthcare system and constant regulatory support also contribute to growth and development.
Brazil Market Trends
The country's growth is characterized by both local and international players, all of whom are actively pursuing collaborations and partnerships in order to enhance their market presence. This also helps in fostering innovation and competition in the region.
How is the Middle East and Africa Region Growing in the Gene-Replacement Therapy Market?
The Middle East and Africa region is expected to witness significant market growth during the forecast period, driven by various factors such as a growing interest in gene therapies, increasing awareness, and investments being made from both public and private sectors. Countries like South Africa and the UAE are leading players in the region, with initiatives aimed at improving healthcare access and innovation.
Saudi Arabia Trends
The country's market landscape still appears to be in its developing stages, with a few local players and international firms exploring opportunities in the region. As the healthcare infrastructure improves, the region is poised for gradual growth in the gene replacement therapy industry.
Emerging Clinical Trials in the Gene-Replacement Therapy Market
| Study Title | Sponsor | Phases | Start Date | Completion Date |
| A Study to Investigate the Safety and Effectiveness of a Coagulation Factor IX Gene Insertion Therapy (REGV131-LNP1265) in Pediatric, Adolescent, and Adult Participants With Hemophilia B | Regeneron Pharmaceuticals | PHASE1|PHASE2 | 9/11/2024 | 12/17/2032 |
| Safety Study of rAAV2/8-hCYP4V2 in Patients With Bietti's Crystalline Dystrophy (BCD) | Beijing Tongren Hospital | EARLY_PHASE1 | 4/21/2021 | 4/29/2024 |
| The Safety and Efficacy of ZVS106e in the Treatment of IRDs Caused by Biallelic Mutations in ABCA4 | Zhongshan Ophthalmic Center, Sun Yat-sen University | EARLY_PHASE1 | 12/26/2025 | 12/30/2028 |
| Safety and Efficacy of GS-100 Gene Therapy in Patients With NGLY1 Deficiency | Grace Science, LLC | PHASE1|PHASE2 | 2/13/2024 | 1/31/2028 |
Gene-Replacement Therapy Market Value Chain Analysis

Who are the Major Players in the Global Gene-Replacement Therapy Market?
The major players in the gene-replacement therapy market include Novartis, bluebird bio, Spark Therapeutics, Gilead Sciences, Pfizer, Sarepta Therapeutics, uniQure, Bristol Myers Squibb, Orchard Therapeutics, BioMarin Pharmaceutical, CRISPR Therapeutics, Regenxbio
Recent Developments
- In November 2025, Novartis got approval from USFDA for the Itvisma (onasemnogene abeparvovec-brve) for the treatment of children two years and older, teens and adults living with spinal muscular atrophy (SMA) with a confirmed mutation in the survival motor neuron 1 (SMN1) gene, making it the first and only gene replacement therapy available for this broad population, offering the potential to reduce the need for chronically administered treatment.(Source: https://www.novartis.com)
- In November 2025, Bharat Biotech International Ltd. announced the formal launch of Nucleon Therapeutics Pvt. Ltd. to support global life science innovators by offering comprehensive process development and manufacturing solutions for advanced therapies addressing complex conditions such as cancer, autoimmune, and rare genetic disorders.(Source: https://newsmeter.in)
Segments Covered in the Report
By Therapy Type
- Viral Vector-Based Gene Replacement
- AAV (Adeno-associated virus)
- Lentiviral vectors
- Adenoviral vectors
- Non-Viral Gene Replacement
- Plasmid DNA
- Lipid nanoparticles
- Polymer-based carriers
By Disease Type
- Genetic Disorders
- Spinal Muscular Atrophy (SMA)
- Hemophilia A/B
- β-Thalassemia
- Duchenne Muscular Dystrophy (DMD)
- Metabolic Disorders
- Neuromuscular Disorders
- Ophthalmic Diseases (e.g., inherited retinal diseases)
- Hematologic Disorders
- Immunodeficiency Disorders
- Others (rare monogenic diseases)
By End User
- Hospitals & Specialty Clinics
- Gene Therapy Centers
- Academic & Research Institutes
- Biotechnology & Pharmaceutical Companies
By Region
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East and Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content

.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
