Sterile filtration is a key process used to remove microorganisms from heat-sensitive liquids and gases without damaging them. It plays a crucial role in ensuring product safety and sterility across industries like pharmaceuticals, biotechnology, food, and healthcare.
What is Sterile Filtration?
Sterile filtration is a process that helps remove microorganisms and bacteria from liquids and gases using membranes with varying pore sizes, without the need for heat or chemicals. The procedure helps maintain product sterility by passing materials through a sterilizing-grade filter that captures even the smallest bacteria or contaminants with ease. The whole procedure involves the use of different types of membranes, such as PVDF, to trap microbes of different sizes. The procedure is essential for heat-sensitive liquids, such as pharmaceutical proteins, vaccines, serums, and vitamins, as well as various forms of gases and air.
Such filters are highly essential in domains such as pharmaceuticals, biotechnology, food and beverages, and healthcare. It helps industries get rid of bacterial contaminants and maintain the sterility of a product with ease. The procedure is helpful in scenarios where solutions can’t be sterilized by heat, irradiation, or chemicals. It helps ensure a product is free from microorganisms, bacteria, and various contaminants.
Industrial Applications of Sterile Filtration
Pharmaceuticals
Sterile filtration is a crucial procedure in the pharmaceutical industry, helpful to remove harmful and deadly microorganisms, bacteria, and other contaminants from heat-sensitive liquids and gases. The procedure involves the use of non-destructive methods to ensure sterility in vaccines and drugs by following the methods of physical removal rather than killing them. It helps ensure product sterility, along with following regulatory frameworks.
Sterilizing grade filters available in minimum sizes help in the removal of minute bacteria and microorganisms. They are highly preferable for low protein binding and chemical compatibility. The procedure is essential in the industry for products that cannot withstand sterilization, such as protein-based drugs, serum, and heat-sensitive API solutions. It is also essential for the filters to pass integrity tests to ensure that they are not damaged and are doing their job the required way.
Biotechnology
Sterile filtration helps the biotech industry to remove contaminants and bacteria from heat-sensitive liquids and gases, along with sterilizing products, protecting bioreactors with the help of materials like PES, PVDF, and PTFE. The procedure is also essential for the industry for sterile drug manufacturing, upstream bioprocessing, and downstream purification. The biotechnology industry involves the use of two filters in series to ensure product quality. The industry also involves the use of nano-filtration in the case of virus removal or the separation of smaller proteins.
Food and Beverage
Sterile filtration in the food and beverage industry involves a non-destructive, cold-stabilization process, with the help of membrane filters to remove microorganisms and bacteria. The procedure is essential for the purification of water, beer, wine, soft drinks, dairy, and gas venting for storage tanks. Membrane cartridges, such as PES or PTFE, are used to ensure the complete removal of microorganisms and bacteria to clean the liquid and gaseous substances.
The procedure helps in the removal of thermal stress, preserves natural aroma, and essential vitamins that heat-sensitive liquids might lose if the procedure is not performed properly. The procedure also helps manufacturers meet stringent regulations for product safety by removing pathogens and other forms of bacteria. Sterile filtration is also essential for the dairy domain to remove harmful microbes and spores from milk and whey, along with maintaining proteins and fats. The procedure also involves the sterilization of reusable filters by using steam in place to ensure the elimination of cross-contamination.
Medical Devices
Sterile filtration helps the medical devices industry ensure sterility of drug products, IV solutions, and delicate equipment by acting as a barrier against contaminants to maintain product integrity. It helps fill injectable, vaccines, pharmaceutical water systems, and sterile gas venting. It also involves the use of membrane filters or depth filters to capture bacteria and fungi. The industry also follows stringent regulatory compliance laid by the FDA/EMA, requiring rigorous validation with different types of tests, such as bubble-point, diffusion, and pressure-hold tests, maintaining product sterility.
Cosmetics
In the cosmetic industry, sterile filtration ensures product safety, maintains stability, and longevity of different types of cosmetic products, such as creams, liquids, and lotions, by using specialized filters. The procedure also helps to prevent microbial contamination in sensitive formulations such as water-based, preservative-free, or natural products. Sterile filtration helps to remove particulate matter, large contaminants, and microorganisms.
What is the Sterile Filtration Market Size in 2026?
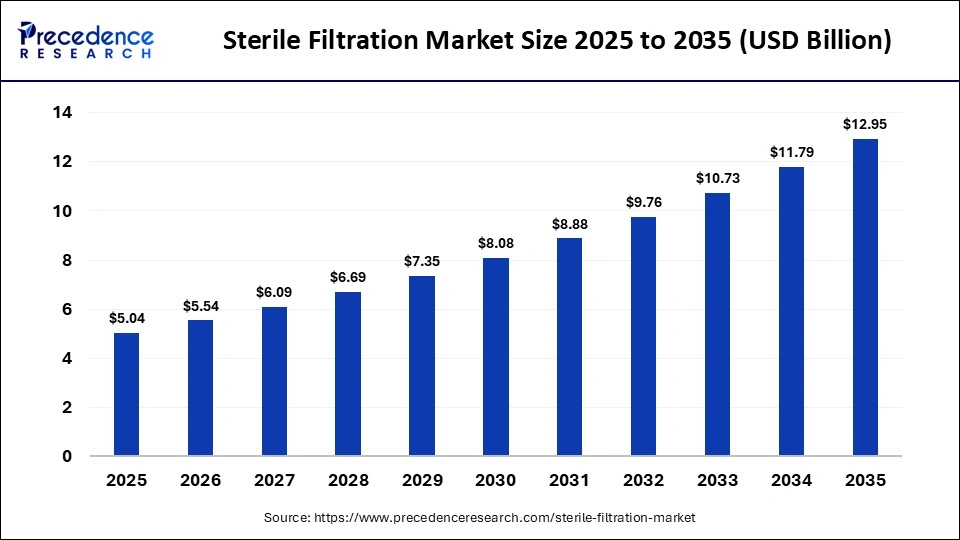
The global sterile filtration market was valued at USD 5.04 billion in 2025 and is projected to grow from USD 5.54 billion in 2026 to approximately USD 12.95 billion by 2035, registering a CAGR of 9.90% during the forecast period from 2026 to 2035. The market is expected to witness strong growth due to the expansion of the global pharmaceutical industry, along with increasing demand for vaccines, monoclonal antibodies, and gene therapies, as well as rising investments in pharmaceutical and biotechnology manufacturing.
Role of Technology in Sterile Filtration
- Improved Membrane Technology: The segment focuses on a major shift of the industry towards the use of materials such as PES and PVDF, providing superior chemical resistance, high flow rates, and strong membrane integrity, which helps process complex biopharmaceuticals. Technological advancements in the form of engineered pore structures provide three to five-fold higher throughput for filtering viscous and high-concentration biologics. Nanomaterials and surface modifications help reduce fouling and increase membrane lifespan to address the high cost of single-use filters.
- Single-Use Systems: The single-use capsules and disposable, pre-sterilized filters help to eliminate the need for costly filters for cleaning, sterilization, and validation between batches. Such systems are also easy and faster to set up, improve operational efficiency, in modular and multi-product facilities. The procedure also highlights the process of using fresh and disposable filters for every process to minimize the risk of cross-contamination for high-value biologics.
- Automation and Digitalization: The segment highlights the importance of the integration of IoT-enabled sensors and automation for continuous monitoring of filtration processes. It helps lower manual intervention and improve batch-to-batch consistency. The segment also involves integration of AI and ML, which is helpful for the prediction of filter fouling and optimal operational parameters, enhancing efficiency, and reducing unexpected downtime.
- New Modalities: Modern cell structures producing higher titers, the filtration technology has helped the industry to handle denser feeds, employ pre-filtration, and dual-layer membrane technology to protect membrane filters. Advanced filtration solutions help support continuous bioprocessing to eliminate the need for recirculation tanks and sheer stress. Specialized filtration also helps process new modalities like AAVs, to filter the minimum-sized bacteria, and ensure product safety.
Different Types of Membranes for Sterile Filtration
- Polyethersulfone (PES): The membrane is known for naturally being hydrophilic with high flux and extremely low protein binding. Hence, it is the top choice for sterile filtration in industries such as food and beverages, pharmaceuticals, and biologics. Such hydrophilic membranes are excellent in removing microorganisms with minimum product loss, making them ideal for drug formulation, buffer preparation, and media sterilization. Such membranes also offer higher hydrophilic properties with high porosity for fast flow rates and high chemical resistance.
- Polyvinylidene Fluoride (PVDF): The membrane is designed for removing microorganisms, bacteria, and mold from liquids or gases. The membrane is also highly demanded due to its high mechanical strength, chemical resistance, and low protein binding, which is ideal for sterilizing pharmaceuticals, biological solutions, and aggressive solvents.
- Nylon: The nylon membrane filters are naturally hydrophilic and durable, ideal for sterile filtration of aqueous solutions and organic solvents. Such membranes are also highly resistant to alkaline solutions, alcohols, and organic solvents.
Role of CDMO and CMO in Sterile Filtration
- CDMOs: They are involved from early-stage development through commercial manufacturing, focusing on robustness, regulatory compliance, and scalability. Such organizations also determine appropriate filtration parameters, including pressure, temperature, and flow rate for certain formulations. They also help to design and execute studies to ensure that the sterilization process removes microorganisms, along with ensuring product safety. They also help to manage the transfer of the sterile filtration process from the clinical state to commercial scale. CDMOs use single-use technologies to eliminate cross-contamination and speed up changeover between batches.
- CMOs: The organization's focus on executing pre-existing, validated processes makes them ideal for commercial-scale production. They also operate high-volume manufacturing lines, including specialized sterile filtration with immediate aseptic filling into vials, ampoules, or pre-filled syringes. They also operate under strict, audited GMP guidelines, along with handling routine monitoring of the filtration process to maintain product sterility. CMOs also provide the necessary capacity to produce large commercial batches or specialized, smaller batches for novel molecules.
Conclusion
Sterile filtration is a crucial procedure in various industries such as food and beverages, medical devices, and biosimilars, to remove microorganisms, bacteria, and different forms of contaminants. The procedure is crucial for liquids and gases that are heat-sensitive, and maintain the product's durability and sterility. Sterile filtration also involves the use of different types of membranes for the detailed removal of contaminants and bacteria for a healthier product.
Expert Advise
According to Precedence Research, sterile filtration is an essential step in various industries involved in manufacturing high-quality products that have a direct impact on human health. The increasing development of biologics, stringent regulations, and the growing demand for single-use technologies contribute to the demand for sterile filtration. Stakeholders focus on developing innovative sterile filtration methods with the integration of AI/ML, enabling digital monitoring. Furthermore, sustainability trends also promote the adoption of sterile filtration, driving enhanced contamination control and high efficiency.
About the Authors

Aditi Shivarkar
Aditi, Vice President at Precedence Research, brings over 15 years of expertise at the intersection of technology, innovation, and strategic market intelligence. A visionary leader, she excels in transforming complex data into actionable insights that empower businesses to thrive in dynamic markets. Her leadership combines analytical precision with forward-thinking strategy, driving measurable growth, competitive advantage, and lasting impact across industries.

Aman Singh
Aman Singh with over 13 years of progressive expertise at the intersection of technology, innovation, and strategic market intelligence, Aman Singh stands as a leading authority in global research and consulting. Renowned for his ability to decode complex technological transformations, he provides forward-looking insights that drive strategic decision-making. At Precedence Research, Aman leads a global team of analysts, fostering a culture of research excellence, analytical precision, and visionary thinking.

Piyush Pawar
Piyush Pawar brings over a decade of experience as Senior Manager, Sales & Business Growth, acting as the essential liaison between clients and our research authors. He translates sophisticated insights into practical strategies, ensuring client objectives are met with precision. Piyush’s expertise in market dynamics, relationship management, and strategic execution enables organizations to leverage intelligence effectively, achieving operational excellence, innovation, and sustained growth.





 Request Consultation
Request Consultation

