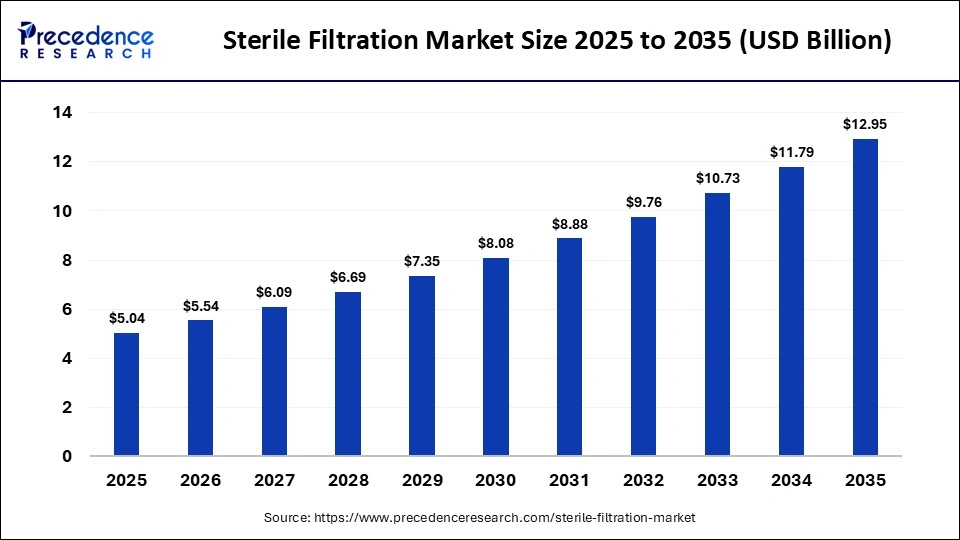
What is the Sterile Filtration Market Size in 2026?
The global sterile filtration market size was calculated at USD 5.04 billion in 2025 and is predicted to increase from USD 5.54 billion in 2026 to approximately USD 12.95 billion by 2035, expanding at a CAGR of 9.90% from 2026 to 2035. The sterile filtration market is expected to grow rapidly due to the growing pharmaceutical industry globally. The market is also expanding with the growing demand for vaccines, monoclonal antibodies, and gene therapies, as well as increasing investments in pharmaceutical and biotech manufacturing.
Key Takeaways
- North America led the global sterile filtration market with approximately 38.40% share in 2025.
- Asia-Pacific is expected to be the fastest-growing region in the foreseeable period.
- By product type, the membrane filters segment held a dominant position over the global market with a share of approximately 32.70% in 2025.
- By product type, the capsule filters segment is expected to grow with the highest CAGR of approximately 10.70% in the coming years.
- By application, the biopharmaceutical manufacturing segment held the largest market share of approximately 34.60% in 2025.
- By application, the vaccine production segment is expected to grow at the fastest CAGR of 11.60% in the market between 2026 and 2035.
- By end user, the pharmaceutical companies segment registered its dominance over the global market with a share of approximately 38.70% in 2025.
- By end user, the contract manufacturing organizations segment is expected to expand rapidly in the market with a CAGR of 11.80% in the coming years.
What is the Sterile Filtration Market?
The sterile filtration industry includes filtration products and systems used to remove microorganisms and particulates from liquids and gases during pharmaceutical, biotechnology, and laboratory processes to maintain sterility and product integrity. It includes membrane filters, cartridges, capsules, syringe filters, and filtration assemblies used across upstream processing, downstream purification, and final sterile filling operations. The market generates revenue from consumables, filtration systems, and supporting services supplied to pharmaceutical manufacturers, biotechnology companies, contract manufacturers, laboratories, and research institutions.
Role of Technology in the Growth of Sterile Filtration Market
- Advanced Membrane Materials: The major shift of the market towards higher usage of performance polyethersulfone (PES) and polyvinylidene fluoride (PVDF) membranes is a major market driver. Such membranes aid higher flow, lower protein binding, and enhanced durability. Such materials help build viscous, high-titer biologics and cell/ gene therapies .
- Disposable Technologies: The growing usage of disposable technology in the form of pre-sterilized, disposable filtration systems is helpful to eliminate the need for cleaning validation, steam-in-place sterilization, and lower the risks of cross-contamination. All such processes also help to drive the efficiency of contract manufacturing organizations (CMOs).
- Industry 4.0: Higher usage of smart technologies in the market, such as smart monitoring, sensors , and AI-powered processes for real-time tracking of the whole procedure, such as pressure and flow rates. Such systems are also useful to predict failure in advance to lower the chances of downtime.
Sterile Filtration Market Trends
- Higher Usage of Single-Use Systems: Growing adoption of single-use systems, such as disposable filters, helps reduce contamination risks and improve flexibility. The burgeoning pharmaceutical industry, along with growing vaccine production, also propels the market's growth.
- Technological Innovations: Use of technological advancements in the form of Industry 4.0 inclusion, AI-driven technology, and smart filtration technology is also a major market driver.
- Stringent Regulatory Framework: Regulatory frameworks to improve sterility today are compelling manufacturers to adopt high-level sterilization techniques.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 5.04 Billion |
| Market Size in 2026 | USD 5.54 Billion |
| Market Size by 2035 | USD 12.95 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 9.90% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product Type, Application, End User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Increasing Production of Biologics
The growing biopharmaceutical production, along with enhanced manufacturing of vaccines , monoclonal antibodies , and biologics, leading to higher demand for rigorous, contamination-free, and scalable filtration solutions for product sterility, is considered to be one of the major market drivers.
Restraint
Membrane Failure
Membrane fouling and failure of integrity test are major restraints of the market, as they lead to operational downtime, product loss, and high total cost of ownership. Aggregation is the most likely cause of clogging of sterile filters, resulting in failed test results.
Opportunity
Integration with AI
The integration of smart technologies for improved automation and the incorporation of IoT and AI-driven technologies for enhanced efficiency drive the future of sterile filtration. AI can turn cleanrooms into intelligent ecosystems by predicting contamination, thereby enhancing sterility assurance and improving HVAC control. AI-based predictive analytics enables manufacturers to detect contamination and make effective decisions.
Segmental Insights
Product Type Insights
Which Product Type Segment Dominated the Sterile Filtration Market?
The membrane filters segment dominated the global market with a share of approximately 32.70% in 2025, driven by the critical role of membrane filters in filtering harmful microorganisms, particles, and contaminants from high-value biopharmaceutical products, vaccines, and cell therapies. The shifting trend towards PES membranes due to their low protein binding characteristics, high flow rates, and strong membrane integrity makes them ideal for sensitive biologics. Single-membrane filters allow for pre-sterilized disposable filters to reduce contamination risks and cross-contamination in multi-product facilities. The exceptional chemical resistance and high thermal stability are also some of the major segment drivers.
Sterile Filtration Market Share, By Product Type, 2025-2035 (%)
| Product Type | 2025 | 2035 |
| Membrane Filters | 32.70% | 30.40% |
| Syringe Filters | 16.80% | 15.70% |
| Capsule Filters | 13.60% | 17.40% |
| Cartridge Filters | 18.70% | 20.80% |
| Filter Discs | 7.40% | 6.10% |
| Bottle-Top Vacuum Filters | 6.30% | 6.00% |
| Filtration Assemblies And Holders | 4.50% | 3.60% |
The capsule filters segment is expected to show the fastest growth with a CAGR of 10.70% over the forecast period, due to the growing demand for single-use technology, small-batch gene and cell therapy production, and stringent contamination regulations. Such membranes are fully enclosed, pre-assembled units that help to eliminate the need for cleaning, and hence they help to mitigate the risk of cross-contamination in multi-product facilities (MPFs). Disposable and pre-sterilized capsule filters provide a reliable solution to meet rigorous standards, fueling the segment's growth.
Application Insights
Why Did the Biopharmaceutical Manufacturing Segment Dominate the Sterile Filtration Market?
The biopharmaceutical manufacturing segment registered its dominance over the global market with a share of approximately 34.60% in 2025, due to the growing demand for sterilizing-grade filters in vaccine, monoclonal antibodies, and cell/gene therapy production. The major shift towards the usage of single-use technology in sterile processing for lowering cross-contamination and strict regulations laid by the EMA/FDA for sterile testing and validation to maintain product purity contribute to the segment's growth. CMOs also have a major contribution to the market's growth due to the utilization of advanced filters for large-scale production.
Sterile Filtration Market Share, By Application, 2025-2035 (%)
| Application | 2025 | 2035 |
| Biopharmaceutical Manufacturing | 34.60% | 32.10% |
| Sterile Drug Formulation and Filling | 22.40% | 20.60% |
| Vaccine Production | 14.30% | 18.80% |
| Cell Culture Media Filtration | 13.70% | 14.10% |
| Laboratory Sample Preparation | 9.80% | 8.60% |
| Others | 5.20% | 5.80% |
The vaccine production segment is expected to gain the highest market share with a CAGR of 11.60% between 2026 and 2035, driven by growing demand for high-performance filtration consumables, membranes, cartridges, and capsule filters. Growing demand for biologics, mRNA vaccines, and viral vector development, and single-use filtration systems to lower the chances of cross-contamination in multi-product facilities, also fosters the segment's growth. Vaccine manufacturing ensures terminal filtration before container closure to ensure sterility.
End User Insights
Sterile Filtration Market Share, By End User, 2025-2035 (%)
| End User | 2025 | 2035 |
| Pharmaceutical Companies | 38.70% | 34.20% |
| Biotechnology Companies | 26.80% | 28.10% |
| Contract Manufacturing Organizations | 16.30% | 22.70% |
| Academic And Research Institutes | 10.40% | 8.30% |
| Clinical Laboratories | 7.80% | 6.70% |
Which End User Segment Led the Sterile Filtration Market?
The pharmaceutical companies segment led the market with a share of approximately 38.70% in 2025, due to the growing demand for sterile, high-production of biologics, vaccines, and advanced therapies. Companies require large-scale sterile filtration for monoclonal antibodies and vaccines. Growing investments in smart, single-use, and integrated systems to lower the contamination risks and the propelling demand for cell and gene therapy manufacturing augment the segment's growth.
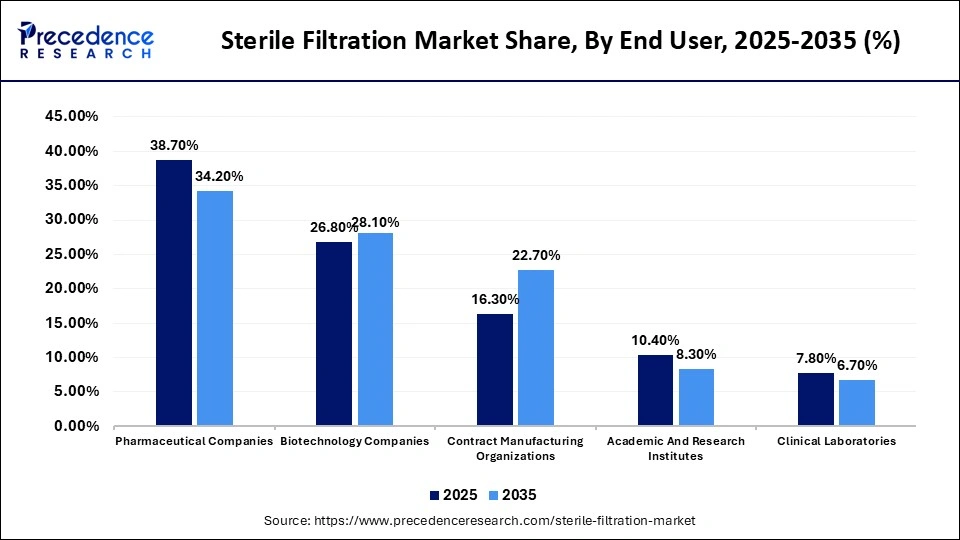
The contract manufacturing organizations segment is expected to witness the fastest growth in the market with a CAGR of 11.80% over the forecast period. Major pharma companies outsource their production part to CMOs. It helps reduce manufacturing costs substantially and allows companies to focus on their core competencies. Such organizations focus on cell and gene therapies, along with driving demand for advanced technologies like capsule filters and single-use systems to offer flexibility, ease of implementation, and cost-effectiveness. The segment's growth is also driven by the use of advanced, automated filtration systems to meet strict sterility and regulatory requirements.
Regional Insights
North America Sterile Filtration Market Size and Growth 2026 to 2035
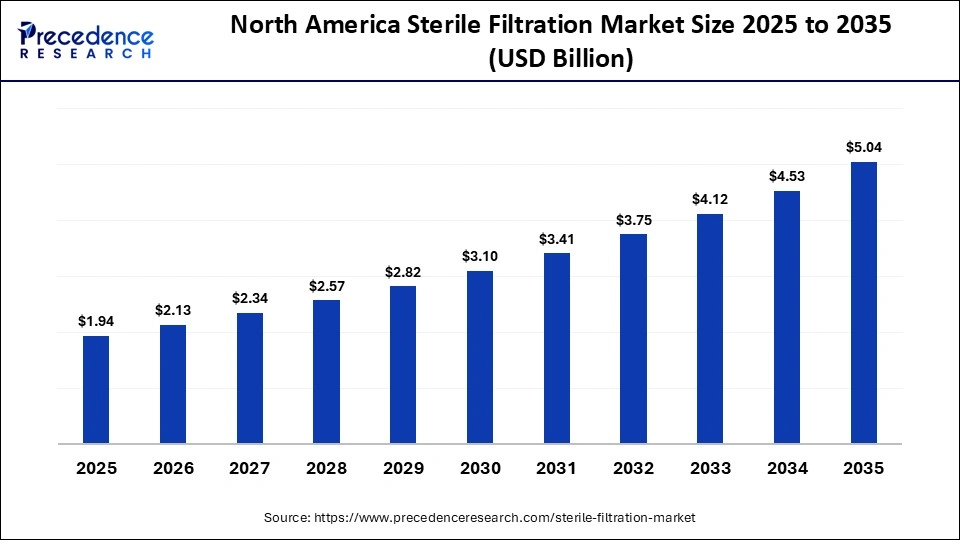
The North America sterile filtration size is estimated at USD 1.94 billion in 2025 and is projected to reach approximately USD 5.04 billion by 2035, with a 10.02% CAGR from 2026 to 2035.
Why North America Dominated the Sterile Filtration Market?
North America held a major market share of approximately 38.40% share in 2025. Factors such as high demand for higher filtration in biotech and pharmaceutical manufacturing, along with the following mandatory regulations led by the FDA, led to the need for higher filtration. Growing investments in monoclonal antibodies, vaccines, and biosimilars for advanced and sterile filtration are also major market drivers. Major shift of the industry in the region towards disposables/single-use technology to maintain sterility and lower the risks of cross-contamination also propels the market's growth in the region. Growing demand for CDMOs to enhance cost-effectiveness and allow companies to focus on core competencies is also a major market driver.
U.S. Sterile Filtration Market Size and Growth 2026 to 2035
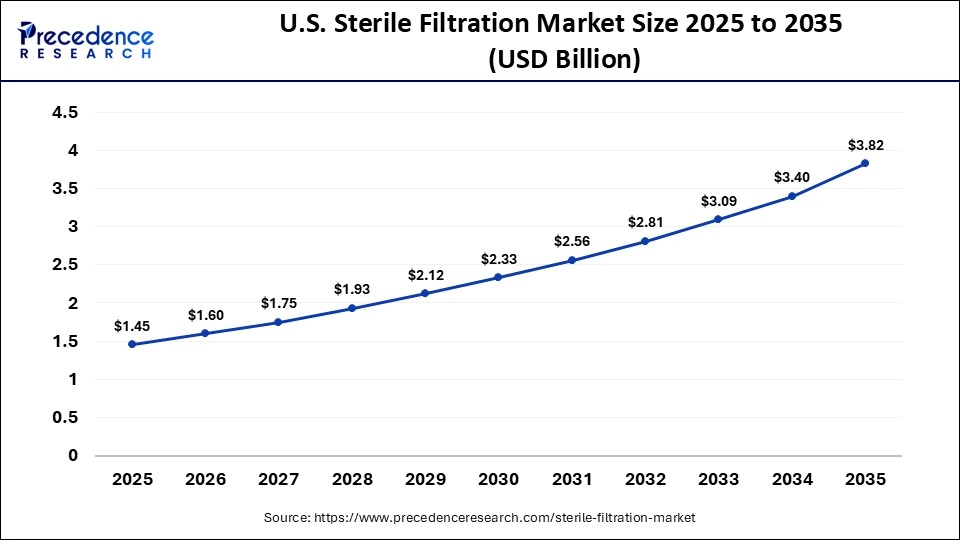
The U.S. sterile filtration market size is calculated at USD 1.45 billion in 2025 and is expected to reach nearly USD 3.82 billion in 2035, accelerating at a strong CAGR of 10.17% between 2026 and 2035.
U.S. Market Trends
The U.S. has a major contribution to the growth of the market due to improved innovation and demand in the region. The region is also a global hub for the development and manufacturing of biologics, monoclonal antibodies, and vaccines, requiring intense filtration to maintain complete safety and efficacy. The growing culture of CMOs/CDMOs in the region by pharma companies for expertise in manufacturing further fuels the market's growth.
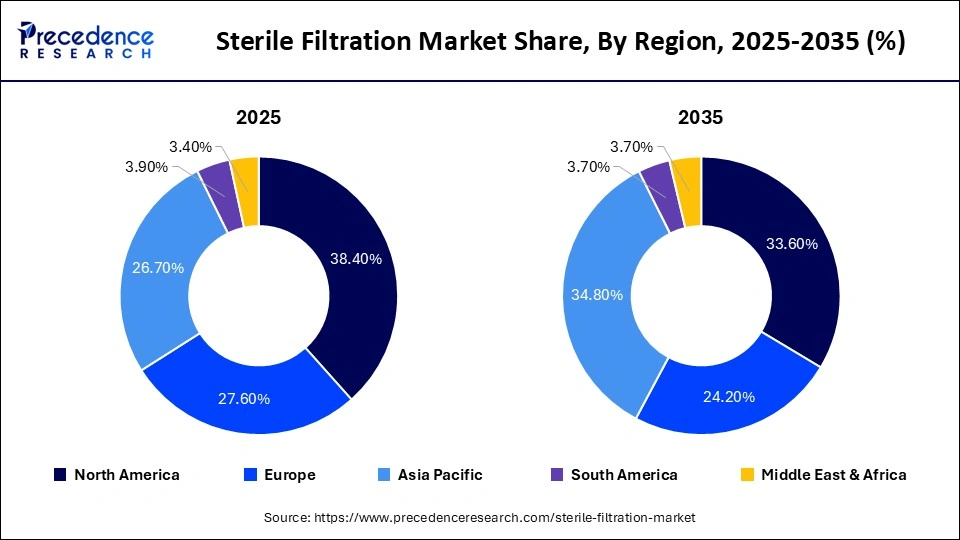
How is Asia-Pacific Growing in the Sterile Filtration Market?
Asia-Pacific is expected to experience the fastest growth during the predicted timeframe, due to booming vaccine production in multiple Asian countries. The market is also observed to grow due to growing investments in domains such as biomanufacturing, stringent sterility regulations, and growing demand for biosimilars .
Growing demand for PES and PVDF membrane filters due to the accelerating production of monoclonal antibodies and vaccines also helps propel market growth. The region is also known as a hub for CDMOs and CMOs to outsource major activities to such organizations and focus on the core areas of a company, which is helpful for market growth. Increased healthcare expenditure and growing government support are other major factors propelling the market.
China Market Trends
China leads the market in Asia-Pacific as it has massive manufacturing capacity expansion, growing demand for biologics, and stricter regulatory standards. The demand is also propelling due to the booming vaccine manufacturing industry in the country. Growing usage of single-use sterile filters to lower the chances of downtime is also a major market driver.
Sterile Filtration Market Companies
- Merck KGaA
- Sartorius AG
- Danaher Corporation
- Thermo Fisher Scientific
- Parker Hannifin
- 3M Company
- Pall Corporation
- GE HealthCare Life Sciences
- Eaton Corporation
- Amazon Filters Ltd
- Cobetter Filtration Equipment
- Meissner Filtration Products
- Porvair Filtration Group
- Sterlitech Corporation
- Graver Technologies
Recent Developments
- In June 2024, Asahi Kasei announced the launch of a membrane system to produce WFI (water for injection), a type of sterile water that is used for the preparation of injections. The membrane system was developed for the production of WFI by leveraging the system design and development capabilities of Microza hollow-fiber membrane.(Source- https://www.asahi-kasei.com )
- In July 2024, Parker Bioscience Filtration introduced a new range of sterilizing-grade gas filters designed to increase microbial security and reduce operational costs for the food and beverage industry. The Asept-X2 filers feature a unique hydrophobic and hydrophilic construction that enables them to withstand elevated temperatures.(Source- https://www.foodbev.com )
Segments Covered in the Report
By Product Type
- Membrane Filters
- Syringe Filters
- Capsule Filters
- Cartridge Filters
- Filter Discs
- Bottle-Top Vacuum Filters
- Filtration Assemblies And Holders
By Application
- Biopharmaceutical Manufacturing
- Sterile Drug Formulation and Filling
- Vaccine Production
- Cell Culture Media Filtration
- Laboratory Sample Preparation
- Others
By End User
- Pharmaceutical Companies
- Biotechnology Companies
- Contract Manufacturing Organizations
- Academic and Research Institutes
- Clinical Laboratories
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
