This blog explores how generative AI is transforming clinical research by automating workflows improving trial accuracy and enabling predictive insights. It covers global adoption industry contributions and future trends shaping modern drug development processes.
Clinical trials have always been one of the most complex and resource-intensive stages of drug development. The process involves extensive data collection, patient recruitment, protocol design, monitoring, and regulatory documentation. In recent years, generative artificial intelligence (GenAI) has begun to reshape how these activities are designed and executed.
GenAI refers to systems capable of creating new content such as text, synthetic data, images, or structured outputs based on learned patterns. In clinical trials, this capability is being applied to streamline documentation, support protocol design, simulate patient populations, and enhance decision-making across trial phases.
Redefining Clinical Trial Design
One of the most impactful applications of GenAI lies in clinical trial design. Traditionally, protocol development requires iterative collaboration among clinicians, statisticians, and regulatory experts. GenAI tools can now assist in drafting protocol frameworks by analyzing historical trial data, therapeutic areas, and regulatory guidelines.
These systems can generate structured protocol suggestions that include inclusion and exclusion criteria, endpoints, and study arms. This reduces the time required for initial drafting and helps teams explore multiple design variations before finalizing a protocol. By incorporating insights from prior studies, GenAI enables more informed design decisions that align with scientific objectives and operational feasibility.
Synthetic Data Generation for Trial Simulation
GenAI has introduced the concept of synthetic data generation, which allows researchers to simulate patient populations without using sensitive personal data. These synthetic datasets are modeled on real-world patterns but do not contain identifiable information.
This capability is particularly valuable in early-stage trial planning, where simulations can help estimate recruitment feasibility, response variability, and outcome distributions. It allows researchers to test assumptions and refine trial parameters before actual execution begins. Synthetic data also supports collaboration between stakeholders by enabling data sharing without compromising privacy regulations.
What is the Generative AI in Clinical Trials Market Size in 2026?
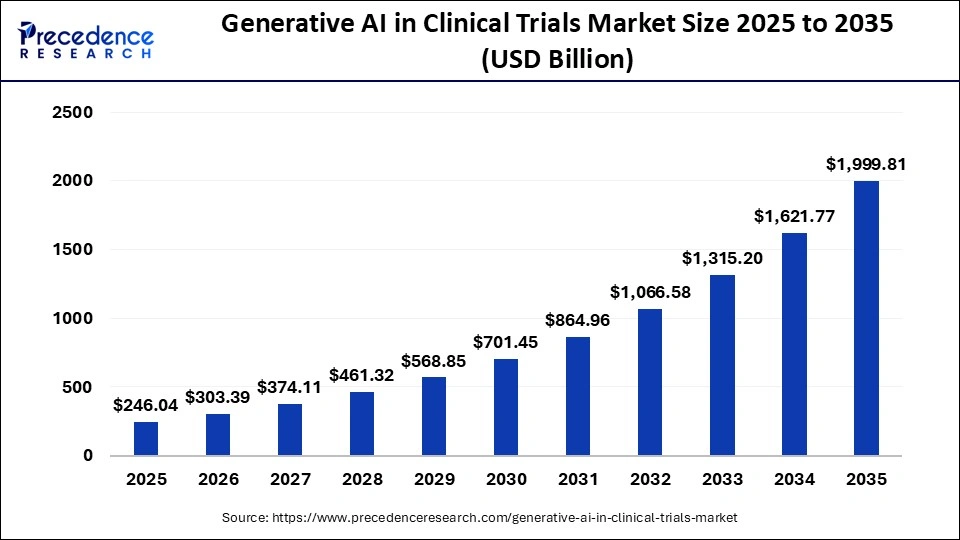
The global generative AI in clinical trials market size was calculated at USD 246.04 billion in 2025 and is predicted to increase from USD 303.39 billion in 2026 to approximately USD 1999.81 billion by 2035, expanding at a CAGR of 23.31% from 2026 to 2035. The market is growing due to increasing demand for faster drug development and decreasing trial costs. Increasing use of AI for patient data analysis and trial optimization is another factor contributing to market growth. Complexity in clinical trials is another factor contributing to market growth for advanced AI solutions.
Get a Free Sample Report with Key Market Trends: https://www.precedenceresearch.com/sample/8217
Adoption of Generative AI in Clinical Trials Across Key Countries
| Country | How Generative AI is Being Used | Key Focus Areas |
| United States | AI-assisted protocol design, patient matching, and clinical documentation | Drug development efficiency, data interoperability |
| United Kingdom | AI-supported trial design and use of national health data for research insights | Public health research, trial optimization |
| Germany | Generative AI used in structured clinical documentation and regulatory submissions | Compliance, precision medicine |
| France | AI-enabled clinical data analysis and trial optimization | Research collaboration, healthcare innovation |
| India | AI-driven patient recruitment and trial site identification | Large-scale patient access, cost efficiency |
| China | Generative AI used in trial simulation, data processing, and drug discovery support | Rapid trial execution, innovation scaling |
| Japan | AI-assisted documentation, imaging analysis, and trial monitoring | Aging population healthcare needs, precision medicine |
Private Sector Contributions and Innovation
Private companies are playing a central role in advancing GenAI applications in clinical trials. Technology firms, pharmaceutical companies, and specialized AI startups are investing heavily in developing solutions tailored to clinical research needs.
Major organizations such as Microsoft, Google, and IBM are providing cloud-based AI platforms that enable data processing, model training, and deployment at scale. These platforms support healthcare organizations in building custom AI workflows for clinical operations.
At the same time, companies like OpenAI are contributing foundational generative models that can be adapted for specialized use cases, including medical and scientific applications. Pharmaceutical companies are also forming partnerships with AI firms to integrate generative capabilities into their research pipelines.
These collaborations are helping bridge the gap between advanced technology and practical clinical application.
Government and Regulatory Support
- Governments and regulatory bodies across the world are increasingly acknowledging the role of AI in healthcare innovation. Agencies are developing frameworks to guide the responsible use of AI in clinical research.
- Organizations such as the U.S. Food and Drug Administration and the European Medicines Agency have introduced guidelines that address the validation, transparency, and ethical use of AI tools in clinical settings. These guidelines emphasize the importance of explainability, data integrity, and human oversight.
- Public sector initiatives are also supporting digital transformation in healthcare through funding programs, research collaborations, and innovation hubs. These efforts are creating an environment where generative AI can be safely integrated into clinical trial workflows while maintaining regulatory compliance.
Generative AI in Clinical Trials: How Different Companies are Contributing
Pharmaceutical and biotechnology companies are increasingly investing in GenAI to improve efficiency, reduce costs, and enhance decision-making across the clinical trial lifecycle, from protocol design to patient recruitment, trial monitoring, and regulatory submissions. Below is an overview of how major companies are advancing efforts in this space.
Large Pharmaceutical Companies Embedding GenAI in R&D Workflows
AstraZeneca
- Clinical trial design: Using AI to analyze real-world evidence and historical trial data to inform protocols.
- Statistical programming & documentation: Automating repetitive tasks such as protocol drafting and report generation.
- Biomarker discovery and patient stratification: Leveraging GenAI models to identify patterns in complex datasets.
- Operational efficiency: Automating administrative workflows, reducing manual effort, and speeding up trial setup
- AstraZeneca also uses GenAI to improve productivity in oncology trials and has reported significant reductions in document authoring time through AI-assisted tools.
Roche
- Large-scale AI infrastructure investments, including high-performance GPU systems to support AI/GenAI workloads.
- Integration of clinical, genomic, and real-world datasets to train models for trial optimization.
- “Lab-in-the-loop” approach, where AI models continuously learn from experimental and clinical data to refine hypotheses.
- Use of AI in patient safety and trial efficiency, including predictive modeling for adverse events and trial outcomes.
Pfizer
- Collaborations with AI platforms and startups to apply machine learning and generative models to drug discovery and trial optimization.
- Use of AI-driven tools for patient selection, biomarker identification, and trial simulation.
- Investments in external AI companies to enhance data-driven clinical trial decision-making.
Future Outlook of Generative AI in Clinical Trials
The future of GenAI in clinical trials is closely tied to the evolution of digital health infrastructure and data availability. As more clinical data becomes digitized and interoperable, AI systems will have greater access to high-quality information for training and inference.
We can expect deeper integration of GenAI into trial operations, including real-time monitoring, adaptive trial design, and automated reporting. These capabilities will enable more flexible and responsive trial execution models. In addition, advancements in multimodal AI, which combine text, imaging, and genomic data, will further enhance the ability to analyze complex clinical datasets and support personalized medicine approaches.
GenAI is reshaping clinical trials by introducing new levels of efficiency, precision, and adaptability across the research lifecycle. From protocol design and patient recruitment to documentation and regulatory interaction, its applications are transforming traditional workflows into more intelligent and automated processes.
With continued support from regulatory bodies, active participation from private companies, and growing integration into clinical ecosystems, GenAI is becoming an essential component of modern clinical research infrastructure. Its ability to synthesize information, generate insights, and support decision-making positions it as a powerful tool in advancing drug development and improving the overall effectiveness of clinical trials.
As adoption deepens, GenAI will continue to redefine how clinical studies are planned, executed, and evaluated, ultimately contributing to faster and more informed pathways for bringing new therapies to patients.
Expert Advise
According to our analysis, GenAI is evolving at a rapid pace to find numerous applications in the healthcare sector, including clinical trials. Advanced technologies, such as machine learning and natural language processing, perform vital functions that are difficult or time-consuming through human methods, thereby addressing bottlenecks in clinical trials. Investments are made in continuous model monitoring and re-training, expanding further applications of GenAI, and improving their capabilities. Stakeholders prioritize ROI-driven applications of GenAI rather than focusing on experimental deployments.
About the Authors

Aditi Shivarkar
Aditi, Vice President at Precedence Research, brings over 15 years of expertise at the intersection of technology, innovation, and strategic market intelligence. A visionary leader, she excels in transforming complex data into actionable insights that empower businesses to thrive in dynamic markets. Her leadership combines analytical precision with forward-thinking strategy, driving measurable growth, competitive advantage, and lasting impact across industries.

Aman Singh
Aman Singh with over 13 years of progressive expertise at the intersection of technology, innovation, and strategic market intelligence, Aman Singh stands as a leading authority in global research and consulting. Renowned for his ability to decode complex technological transformations, he provides forward-looking insights that drive strategic decision-making. At Precedence Research, Aman leads a global team of analysts, fostering a culture of research excellence, analytical precision, and visionary thinking.

Piyush Pawar
Piyush Pawar brings over a decade of experience as Senior Manager, Sales & Business Growth, acting as the essential liaison between clients and our research authors. He translates sophisticated insights into practical strategies, ensuring client objectives are met with precision. Piyush’s expertise in market dynamics, relationship management, and strategic execution enables organizations to leverage intelligence effectively, achieving operational excellence, innovation, and sustained growth.





 Request Consultation
Request Consultation

