What is Radioligand Therapy Market Size in 2026?
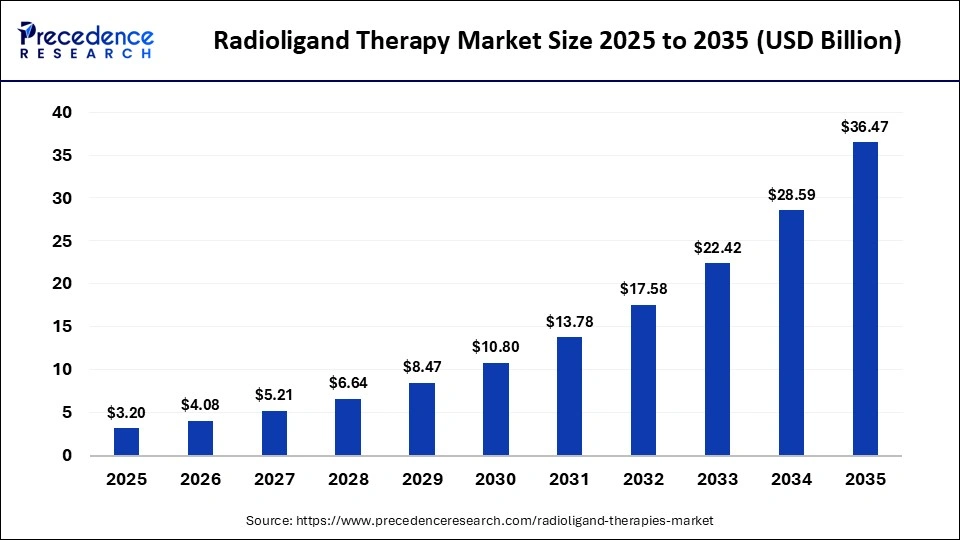
The global radioligand therapy market size was calculated at USD 3.20 billion in 2025 and is predicted to increase from USD 4.08 billion in 2026 to approximately USD 36.47 billion by 2035, expanding at a CAGR of 27.55% from 2026 to 2035. The growth of the market is driven by rising cancer awareness, continuous radioligand innovation, and a favorable regulatory framework.
Key Takeaways
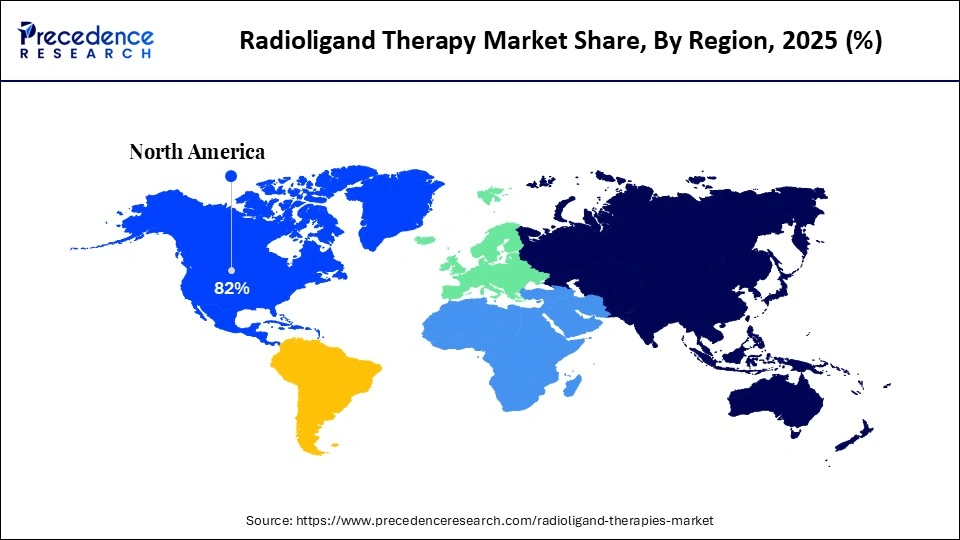
- North America led the radioligand therapy market share of 82% in 2025 with a significant share.
- Asia Pacific is expected to grow at a rapid pace in the coming years.
- By product, the Lutetium Lu 177 Vipivotide Tetraxetan segment led the market in 2025.
- By product, the Lutetium Lu 177 Dotatate segment is expected to grow at the fastest CAGR in the foreseen period.
- By indication, the prostate cancer segment led the global market in 2025.
- By indication, the neuroendocrine tumors segment is expected to grow at the fastest CAGR in the upcoming period.
- By target, the prostate-specific membrane antigen (PSMA) segment led the global market in 2025.
- By target, the somatostatin receptor segment is expected to grow at a significant rate in the foreseeable period.
- By end user, the tertiary care academic/comprehensive cancer centers segment dominated the market in 2025.
- By end user, the specialized nuclear medicine centers segment is observed to grow at the fastest rate in the coming years.
Radioligand Therapy Market Overview
Radioligand therapy is an advanced targeted treatment that delivers radiation directly to cancer cells. Instead of traditional chemotherapy , it uses a radioactive isotope linked to a specific molecule that binds to tumor cells. Alpha-emitting isotopes release high-energy radiation over a short distance, enabling precise tumor cell destruction. In contrast, beta-emitting isotopes travel slightly farther, allowing treatment of a broader tumor area. By concentrating radiation at the tumor site, this approach helps limit damage to surrounding healthy tissue. The regulatory environment is also progressing, with global health authorities increasingly supporting the clinical adoption of radioligand therapies in oncology .
Impact of Artificial Intelligence on the Radioligand Therapy Market
Artificial intelligence is transforming healthcare by improving diagnostic accuracy, enabling real-time imaging, and supporting data-driven decision-making. Using artificial intelligence enhances ligand design and development, imaging in preclinical settings, and practice sustainability. Additionally, medical image analysis and decision-making can also be supported by Generative AI. CNN U-Net architectures can deconvolve low-count SPECT and PET images.
The use of AI-driven digital twins also decreases costs, shortens the preclinical phases of radioligand development, and supports sustainability. CBCT-based systems using AI, such as the Varian Ethos, have enabled clinical online adaptive radiotherapy. Predictive analytics and machine learning help identify suitable patients, optimize treatment schedules, and anticipate adverse effects, improving both safety and efficacy. Additionally, AI-driven automation in radiopharmaceutical synthesis and quality control streamlines production, reduces human error, and accelerates the availability of RLT therapies, ultimately supporting wider adoption in oncology.
Radioligand Therapy Market Trends
- Rising cancer awareness among clinicians and patients is accelerating early diagnosis and driving adoption of radioligand therapies, supported by growing recognition of their targeted treatment benefits.
- Government initiatives to expand specialized cancer and nuclear medicine centers are strengthening radioligand therapy infrastructure and enhancing comprehensive cancer care and post treatment support.
- Rising adoption of alpha and beta emitting isotopes in radioligand therapy is enabling targeted tumor cell destruction through localized radiation, improving treatment precision and efficacy while expanding oncology applications.
- Strategic manufacturing scale up by major players is strengthening the radioligand supply network and accelerating global precision oncology market growth.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 3.20 Billion |
| Market Size in 2026 | USD 4.08 Billion |
| Market Size by 2035 | USD 36.47 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 27.55% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product,Indication,Target,End User, and region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Product Insights
How Did the Lutetium Lu 177 Vipivotide Tetraxetan Segment Dominate the Market?
The Lutetium Lu 177 Vipivotide Tetraxetan segment dominated the market with the largest share in 2025. This is mainly due to its approval for use in earlier stages of disease treatment. It is administered intravenously and acts via ionizing radiation emission. Lutetium Lu 177 Vipivotide Tetraxetan, marketed under the brand name PLUVICTO, is a radioligand therapeutic agent developed by Advanced Accelerator Applications (a subsidiary of Novartis) for the treatment of prostate-specific membrane antigen (PSMA)-expressing metastatic prostate cancer in patients who have already received hormone therapy and are considered appropriate to delay chemotherapy. However, treatment with PLUVICTO may pose risks, including radiation exposure, myelosuppression, and renal toxicity.
The Lutetium Lu 177 Dotatate segment is projected to grow at the fastest CAGR in the upcoming period due to its effectiveness in treating neuroendocrine cancers. It is marketed under the brand name Lutathera and administered intravenously. Lutetium-177 (177 Lu)-Dotatate therapy is indicated for the treatment of somatostatin receptor–positive gastroenteropancreatic neuroendocrine tumors (GEP-NETs), including tumors arising in various parts of the gastrointestinal tract. The rising need for targeted radiotherapy solutions, amid increasing global cancer incidence, is supporting segmental growth.
Indication Insights
What Made Prostate Cancer the Leading Segment in the Market?
The prostate cancer segment led the radioligand therapy market in 2025, primarily due to the increasing number of patients diagnosed with prostate cancer. Prostate cancer is the most common cancer and the second leading cause of cancer-related death among men in the U.S. The disease may be asymptomatic in its early stages and often follows an indolent course that may require only active surveillance. About 1 in 44 men die from prostate cancer, and men with a family history of the disease are at higher risk. These factors collectively support the growth of the segment.
The neuroendocrine tumors segment is expected to expand at the fastest CAGR over the forecast period, primarily due to the rising prevalence and clinical recognition of malignant neuroendocrine tumors. Neuroendocrine tumors (NETs) are rare, heterogeneous malignancies with highly variable clinical behavior. It can arise anywhere in the body, but most commonly occurs in the lungs, appendix, small intestine, rectum, and pancreas. NETs are broadly classified into functional NETs, which secrete hormones and cause characteristic symptoms, and non-functional NETs, which do not secrete hormones but can still lead to symptoms by compressing or invading surrounding organs. As most NETs are malignant and increasingly being diagnosed and treated with targeted radioligand therapies, this indication segment is experiencing robust market growth.
Target Insights
What Made Prostate-Specific Membrane Antigen (PSMA) the Dominant Segment in the Market?
The prostate-specific membrane antigen (PSMA) segment dominated the market while holding a major share in 2025 and is expected to sustain its position in the coming years. This is mainly due to the increasing adoption of PSMA PET diagnostics. PSMA-targeted therapy is a newer form of radiation therapy used to treat prostate cancer. PSMA is a highly validated, high-contrast target for both diagnostic imaging and therapeutic applications. The decline in prostate cancer mortality is likely due to earlier detection and advances in treatment, thereby reinforcing the sustained growth of the segment in the overall market.
The somatostatin receptor segment is expected to grow at a notable rate in the market because somatostatin receptors are highly expressed in neuroendocrine tumors (NETs), which makes them ideal targets for targeted radioligand therapies. Treatments like Lutetium-177 DOTATATE selectively bind to these receptors, allowing precise delivery of radioactive isotopes to tumor cells while sparing healthy tissue. The rising incidence of NETs, coupled with strong clinical evidence supporting improved patient outcomes and prolonged survival, is driving the adoption of somatostatin receptor-targeted therapies, fueling growth in this segment.
End User Insights
How Did the Tertiary Care Academic/Comprehensive Cancer Centers Segment Lead the Market?
The tertiary care academic/comprehensive cancer centers segment led the market with the highest share in 2025. This is because these centers have advanced infrastructure, specialized oncology departments, and highly trained staff capable of handling complex RLT procedures. They are also at the forefront of clinical trials and research, enabling early adoption of novel therapies like somatostatin receptor-targeted treatments. Their high patient volumes, focus on rare and complex cancers, and access to multidisciplinary teams make them the preferred end users for implementing radioligand therapies, driving the segment's market dominance.
The specialized nuclear medicine centers segment is projected to grow at the fastest rate during the forecast period because these centers focus exclusively on advanced nuclear imaging and targeted radiotherapies, allowing them to efficiently handle complex RLT procedures. They offer specialized expertise, state-of-the-art equipment, and streamlined workflows, which improve treatment accuracy and patient outcomes. Additionally, the increasing patient demand for precision oncology and the rising adoption of radioligand therapies in outpatient and specialized settings are driving the growth of this segment.
Regional Insights
North America Radioligand Therapy Market Size and Growth 2026 to 2035
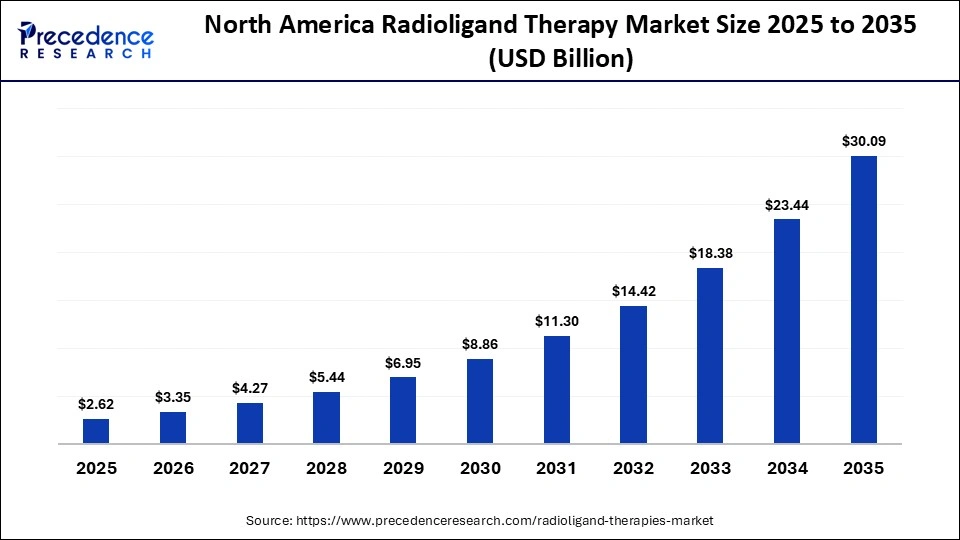
The North America radioligand therapy market size is estimated at USD 2.62 billion in 2025 and is projected to reach approximately USD 30.09 billion by 2035, with a 27.65% CAGR from 2026 to 2035.
Which Factors Contribute to North America's Leading Position in the Radioligand Therapy Market?
North America led the radioligand therapy market by holding a major share of 82% in 2025 and is projected to sustain its leadership over the forecast period. This growth is driven by a large patient population, a mature and well‑established reimbursement system, high penetration of PET diagnostics, and a high concentration of theranostic centers. In the U.S., prostate cancer remains one of the leading causes of death among men, significantly contributing to the demand for radioligand therapy (RLT). Rising incidences of target cancers, continuous innovation in radiopharmaceutical development and imaging technologies, and favorable regulatory support are enhancing treatment efficacy and safety, thereby encouraging wider adoption.
U.S. Radioligand Therapy Market Size and Growth 2026 to 2035
The U.S. radioligand therapy market size is calculated at USD 2.10 billion in 2025 and is expected to reach nearly USD 24.22 billion in 2035, accelerating at a strong CAGR of 27.70% between 2026 to 2035.
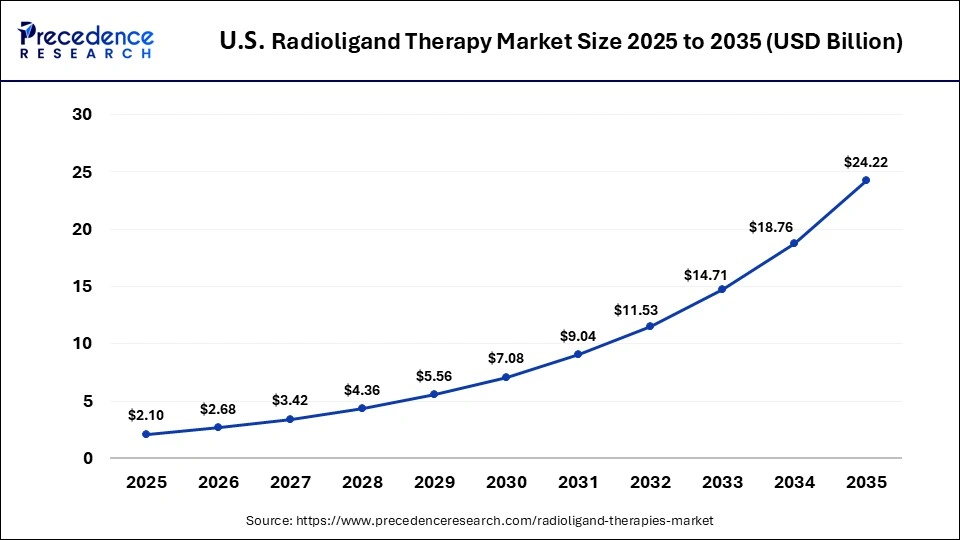
The United States is a central driver of the global Radioligand Therapy (RLT) market, underpinned by its advanced healthcare infrastructure, strong investment in nuclear medicine research, and early adoption of precision oncology treatments. The U.S. contributes a substantial share of market revenues in North America where radioligand therapies for prostate cancer and neuroendocrine tumors are widely prescribed and benefits from favorable FDA regulatory pathways and supportive reimbursement policies that facilitate faster clinical uptake of novel radiopharmaceuticals. Major pharmaceutical firms and biotech innovators are actively expanding manufacturing and clinical development within the country, with planned facilities to scale domestic production of key RLT agents and improve supply chain logistics.
Canada Radioligand Therapy Market Analysis
Canada is one of the major contributors to the North American market. This is due to the increased awareness among healthcare providers and patients regarding the benefits of RLT. Moreover, growing clinical trial activity is strengthening the country's position within the integrated RLT landscape.
Europe demonstrates strong demand for radioligand therapies, with countries such as Germany, France, and the UK leading adoption. The region benefits from a stringent yet supportive regulatory environment, growing awareness among healthcare professionals, and increasing integration of advanced imaging and radiopharmaceutical technologies into oncology care. These factors collectively support the steady growth of the radioligand therapy market across Europe.
How is the Opportunistic Rise of Asia Pacific in the Radioligand Therapy Market?
Asia Pacific is expecetd to grow at a rapid pace due to rising cancer incidences, increasing healthcare investment, and improved access to cutting-edge therapies. China and Japan are key markets in the region. Japan in particular holds a large share of the market, owing to high clinical adoption. Strong cancer care infrastructure and rapid adoption of novel therapies are likely to support regional market growth.
In India, the central government has implemented the Strengthening of Tertiary Care Cancer Facilities scheme under the National Program for Prevention and Control of Non-Communicable Diseases (NP-NCD). Demand for nuclear medicine is growing rapidly, especially in advanced hospitals offering integrated cancer care, driven by increasing diagnostic capabilities and disease prevalence. In addition, a favorable regulatory scenario is expected to support market growth across Asia Pacific.
Radioligand Therapy Market Value Chain Analysis
- Raw Material Sourcing:
This stage involves sourcing critical isotopes, radionuclides, and chemical precursors needed for RLT production.
Key players: Nordion, IBA, and Lantheus Medical Imaging
- Radioligand Manufacturing & Synthesis:
Companies develop and radiolabel peptides, antibodies, or small molecules for targeted therapy.
Key players: Novartis, Advanced Accelerator Applications (AAA), and Bayer
- Healthcare Providers / Treatment Centers:
Deliver RLT to patients, including administration, monitoring, and follow-up.
Key players: tertiary care cancer centers, specialized nuclear medicine centers, and hospitals like Mayo Clinic and Memorial Sloan Kettering Cancer Center
Radioligand Therapy Market Companies
- Novartis AG
- Bayer AG
- Eli Lilly and Company
- AstraZeneca
- Lantheus Holdings, Inc.
- Curium Pharma
- Bristol Myers Squibb
- Telix Pharmaceuticals
- ITM Isotope Technologies Munich SE
- Molecular Partners AG
- Orano Med
- Ariceum Therapeutics
- Clarity Pharmaceuticals
- Radiopharm Theranostics
- Perspective Therapeutics
Recent Developments
- On February 26, 2026, Novartis announced plans to build a new radioligand therapy site in Denton, Texas, to deliver more next generation treatments to patients. The company plans to establish a new 46,000 square foot radioligand therapy (RLT) manufacturing facility in Denton, which will be its fifth RLT site in the US and its first manufacturing facility in Texas, marking further progress in Novartis 23 billion US investment program.(Source: https://www.novartis.com )
- In 2025, the FDA approved Novartis' radioligand therapy Pluvicto (lutetium Lu 177 vipivotide tetraxetan) for allowing administration before chemotherapy in patients with prostate specific membrane antigen (PSMA) positive mCRPC who have previously received an androgen receptor pathway inhibitor (ARPI) and are considered suitable candidates to delay chemotherapy.(Source: https://www.novartis.com )
- In 2025, the FDA cleared an investigational new drug (IND) application for AB001, a prostate-specific membrane antigen (PSMA) targeted radioligand therapy, enabling the initiation of a phase 1 trial in patients with metastatic castration resistant prostate cancer (mCRPC).(Source: https://www.urologytimes.com )
Segments Covered in the Report
By Product
- Lutetium Lu 177 Vipivotide Tetraxetan
- Lutetium Lu 177 Dotatate
- Others
By Indication
- Prostate Cancer
- Neuroendocrine Tumors
- Others
By Target
- Prostate-Specific Membrane Antigen (PSMA)
- Somatostatin Receptor
- Others
By End User
- Tertiary Care Academic/Comprehensive Cancer Centers
- Specialized Nuclear Medicine Centers
- Others
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting