What is Medical Device Manufacturing Equipment Production Market Size in 2026?
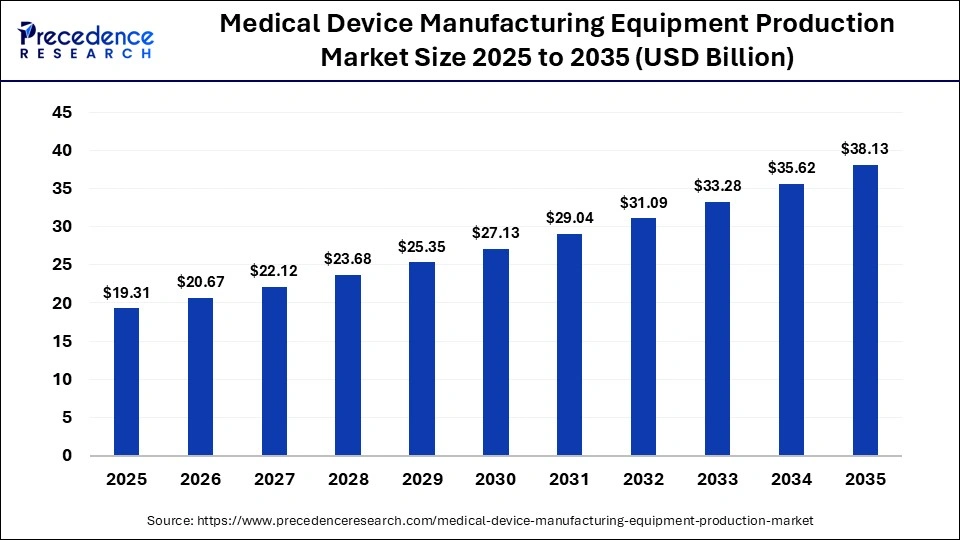
The global medical device manufacturing equipment production market size was calculated at USD 19.31 billion in 2025 and is predicted to increase from USD 20.67 billion in 2026 to approximately USD 38.13 billion by 2035, expanding at a CAGR of 7.04% from 2026 to 2035. The market is driven by growing demand for advanced medical devices and increasing automation in production processes.
Key Takeaways
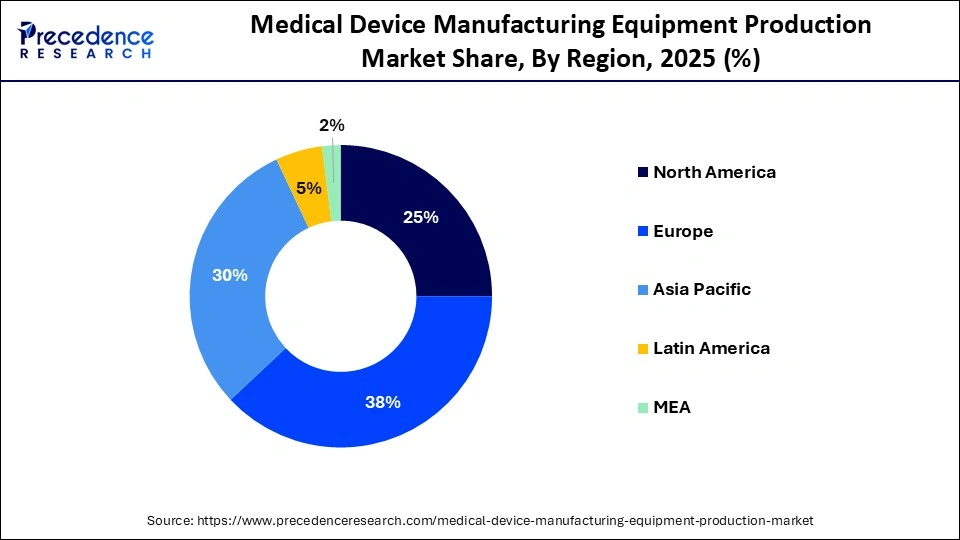
- By region, Europe dominated the market with a major share of 38% in 2025.
- By region, Asia Pacific is expected to grow at the fastest CAGR of 8.0% in the market during the forecast period.
- By equipment type, the material processing equipment segment holds a dominant position in the market with a 45% share in 2025.
- By equipment type, the inspection & quality control equipment segment is expected to grow at the fastest CAGR of 6.3% in the market between 2026 and 2035.
- By application, the consumables & disposables segment accounted for a major revenue share of 40% in the market in 2025.
- By application, the sterile devices production segment is expected grow at a notable rate in the market during the studied years.
- By end user, the OEM manufacturers segment led the global market with a share of 60% in 2025.
- By end user, the CMOs segment is expected to expand rapidly in the market with a CAGR of 6.7% in the coming years.
- By technology adoption, the automation & robotics segment registered its dominance in the global market with a share of 50% in 2025.
- By technology adoption, the additive manufacturing / 3D printing segment is expected to grow at the fastest CAGR of 6.9% over the forecast period.
Market Overview
The medical device manufacturing equipment production market includes specialized machines, production systems, and integrated technologies utilized in the production and assembly of medical equipment. The market comprises material processing equipment, precision molding equipment, extrusion equipment, CNC machining equipment, sterilization equipment, automated inspection platforms, cleanroom equipment, assembly lines, and innovative packaging equipment. They are needed to ensure that the levels of accuracy, repeatability, ability to control contamination, and compliance with high international regulatory standards. The growth of the market is driven by rising global medical device production, automation adoption, stringent safety & quality standards, and demand for minimally invasive devices, cleanroom solutions, and advanced manufacturing technologies across diagnostics, implants, surgical instruments, and disposables sectors.
How is AI Integration Impacting the Medical Device Manufacturing Equipment Production Market?
The medical device manufacturing equipment production market is undergoing a significant change due to artificial intelligence that enhances accuracy, quality, and performance. AI-based systems are increasingly integrated into manufacturing equipment to monitor production parameters in real time, including temperature, pressure, alignment, and material consistency. Machine learning algorithms analyze large volumes of production data to detect defects, predict equipment failures, and optimize manufacturing processes. Additionally, AI-powered simulation models and digital twins allow manufacturers to virtually test production scenarios, improving scalability, reducing development time, and enhancing overall operational efficiency.
Medical Device Manufacturing Equipment Production Market Trends
- Automation and robotics are being applied into medical device production lines to enhance precision, reduce human error, and increase efficiency in the large-scale production plants.
- The growing adoption of Industry 4.0 technologies, including IoT-based monitoring systems and AI-driven quality control tools, is streamlining production processes while ensuring regulatory compliance.
- There is a rising use of 3D printing and additive manufacturing to produce customized implants, prostheses, and minimally invasive medical devices with greater design flexibility and faster turnaround times.
- The expansion of manufacturing facilities in emerging markets is driven by local production incentives, cost advantages, and increasing healthcare investments, strengthening global supply chain capabilities.
- As medical devices become smaller and more sophisticated, precision machining and microfabrication technologies are gaining importance in equipment production.
- The shift toward disposable and single-use medical devices, especially in infection-control settings, is driving demand for specialized production lines.
- Cobots working alongside human operators are increasingly used to enhance flexibility and boost production efficiency without full automation.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 19.31 Billion |
| Market Size in 2026 | USD 20.67 Billion |
| Market Size by 2035 | USD 38.13 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 7.04% |
| Dominating Region | Europe |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Equipment Type,Application,Technology Adoption, End User, and region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segment Insights
Equipment Type Insights
Why Did the Material Processing Equipment Segment Hold a Major Share in the Market?
The material processing equipment segment held a 45% share in the medical device manufacturing equipment production market in 2025. This is because material processing equipment like injection molding, extrusion, laser cutting, CNC machining, and micro-manufacturing tools are essential in creating high precision in parts that would be utilized in syringes , catheters, implants, diagnostic cartridges, and surgical instruments. With more compact and technologically sophisticated medical equipment being produced, the equipment needs to be able to provide tight tolerances, biocompatible material processing, and be reliably repeatable. Furthermore, rising demand for minimally invasive and disposable devices has further elevated the need for high-speed and automated material processing lines that are associated with boosting throughput and cutting down production expenses.
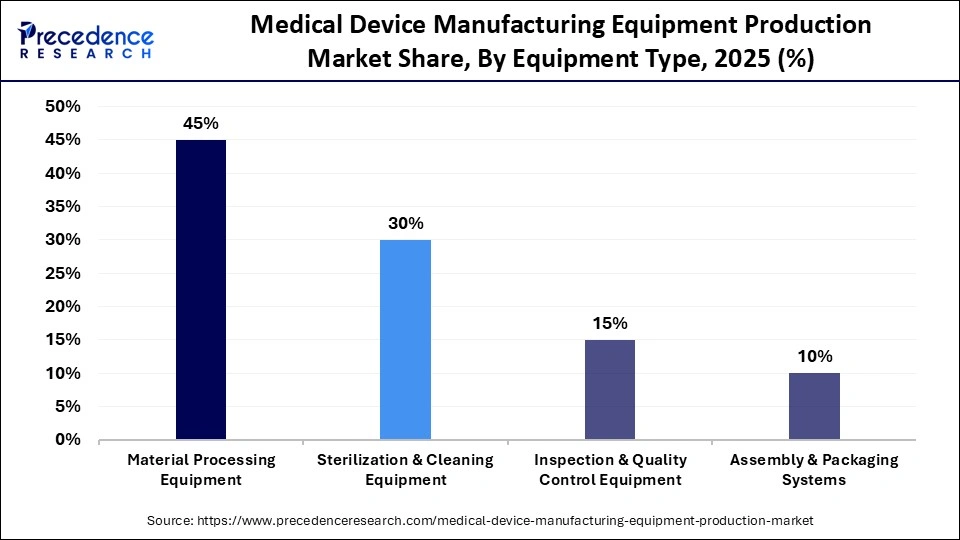
The inspection & quality control equipment segment is expected to grow at the fastest CAGR of 6.3% in the market between 2026 and 2035, driven by the increasing regulatory scrutiny, the growing number of product recalls, and the necessity to manufacture zero-defect medical devices. With the increasing standards of global compliance, manufacturers are now focusing on more sophisticated inspection technologies to have accurate traceability and patient safety. Optical inspection systems, vision analytics based on AI, non-destructive testing systems, and real-time monitoring systems are being progressively added to production lines to identify microscopic defects and performance anomalies. Also, the growth of multi-stage quality verification processes has increased due to the growth of complex and implantable devices.
Application Insights
Why Did the Consumables & Disposables Segment Lead the Market in 2025?
The consumables & disposables segment led the medical device manufacturing equipment production market with a share of 40% in 2025 and is expected to maintain its leading position in the coming years. This is mainly due to the high-volume demand for single-use products such as syringes, IV sets, catheters, surgical masks, blood collection tubes, and diagnostic kits. These items are essential in daily healthcare operations across hospitals, clinics, and diagnostic laboratories, ensuring a continuous production cycle. Rising chronic disease prevalence, increasing hospitalizations, expanded immunization programs, and growing surgical procedures worldwide have further accelerated demand for disposable medical products, supporting segment dominance.
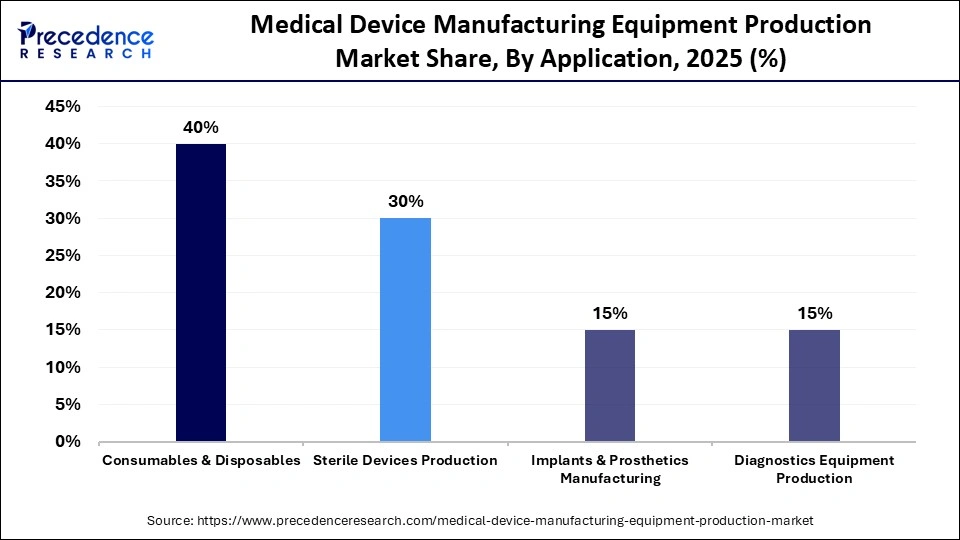
The sterile devices production segment is expected to grow at a notable rate in the upcoming period due to the increasing regulatory scrutiny and rising demand for contamination-free medical products. Devices such as implants, surgical instruments, wound-care products, and critical care equipment require validated sterilization processes and controlled manufacturing environments. With stricter global quality and safety standards, manufacturers are investing in advanced sterilization systems, cleanroom robotics, and automated monitoring technologies. Additionally, the growing adoption of minimally invasive procedures and implantable devices is further boosting demand for reliable sterile production lines.
End User Insights
Why Did OEM Manufacturers Lead the Medical Device Manufacturing Equipment Production Market?
The OEM manufacturers segment registered its dominance in the global market with a major share of 60% in 2025, supported by strong innovation capabilities, proprietary technologies, and end-to-end production control. Large OEMs are typically vertically integrated, enabling them to manage component fabrication, assembly, quality testing, and packaging within a single controlled ecosystem. This structure ensures strict compliance with global regulatory standards while protecting intellectual property and maintaining product consistency. Additionally, OEMs possess substantial financial resources, allowing continuous investment in advanced machinery, automation, digital twins, and smart factory technologies.
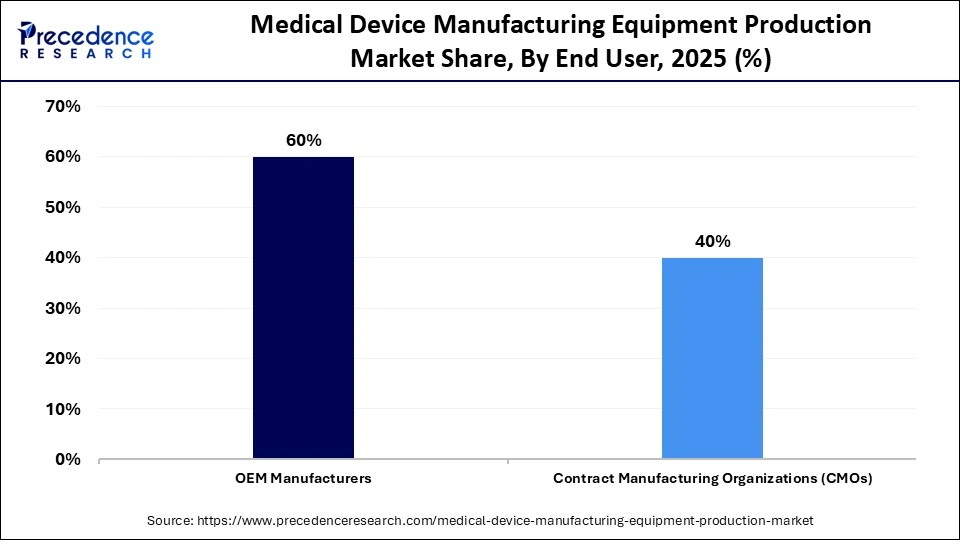
The CMOs segment is expected to grow at a CAGR of 6.7% over the forecast period, driven by the increasing outsourcing strategies of medical device companies seeking cost efficiency and operational flexibility. Small and mid-sized manufacturers, in particular, prefer partnering with CMOs to avoid heavy investments in production infrastructure and regulatory compliance systems. CMOs offer specialized expertise, scalable manufacturing capabilities, and established quality management systems, helping accelerate product commercialization and market entry.
Technology Adoption Insights
Why Did the Automation & Robotics Segment Lead the Medical Device Manufacturing Equipment Production Market?
The automation & robotics segment led the market with a share of 50% in 2025 due to the increased demand for precision, repeatability, and high-throughput production in the manufacturing of medical devices. Robotic assembly, automated material handling, and AI-based inspection platforms significantly reduce the number of human errors and ensure the highest compliance with the regulations. Automation enhances uniformity in the production process of micro-scale components, which are important in implantable devices as well as minimally invasive devices. The increasing cost of labor, as well as increasing demand to have 24/7 production efficiency, pushes the use of automation.
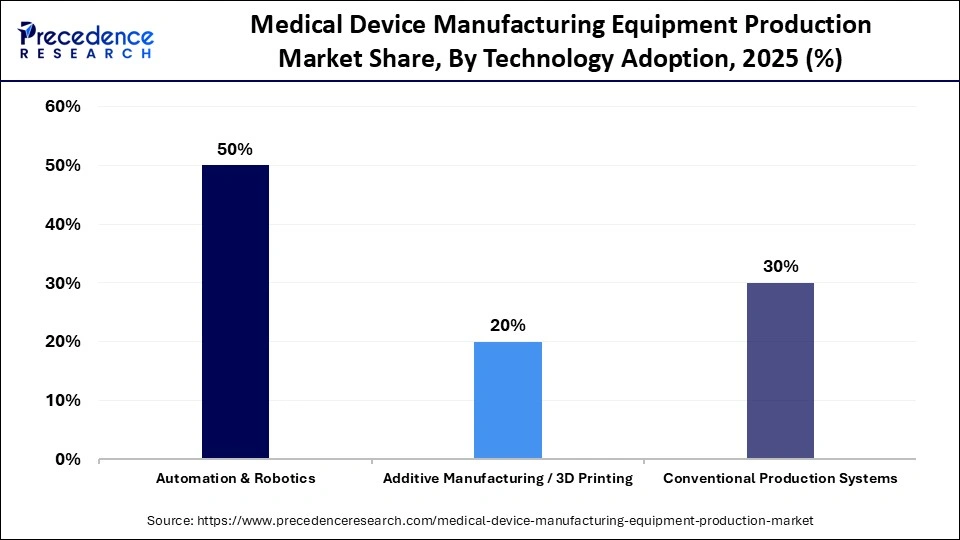
The additive manufacturing / 3D printing segment is expected to expand rapidly in the market with a CAGR of 6.9% in the coming years, driven by the increase in demand for customized and patient-specific medical devices, i.e., orthopedic implants, dental components, and surgical guides. Additive manufacturing allows the creation of complex geometries that are challenging or impossible to produce using the conventional machining process. Prototyping is also enhanced and makes technology quicker in product development, as well as decreasing the time-to-market. It also minimizes the volume of waste material and promotes lightweight design products, which promote cost effectiveness and sustainability.
Regional Insights
Europe Medical Device Manufacturing Equipment Production Market Size and Growth 2026 to 2035
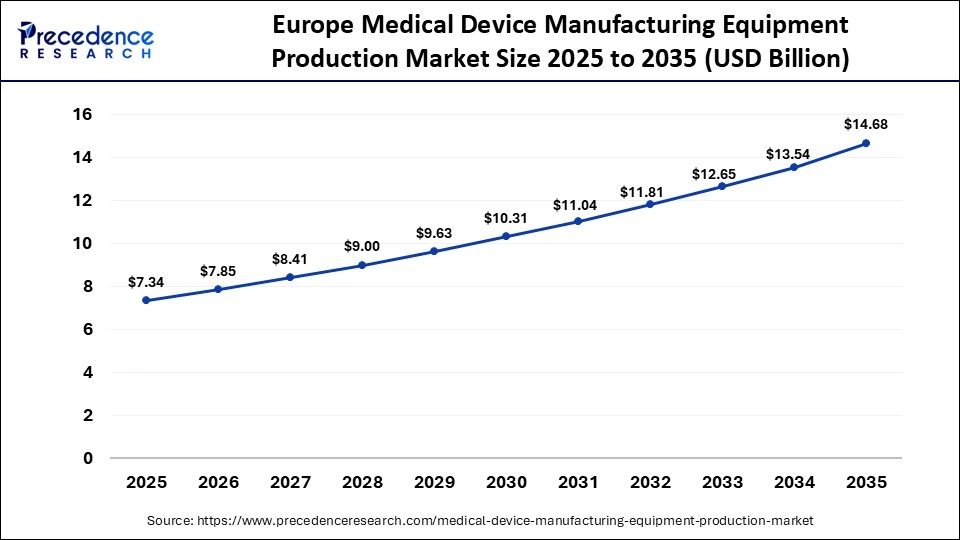
The Europe medical device manufacturing equipment production market size has grown strongly in recent years. It will grow from USD 7.34 billion in 2025 to USD 14.68 billion in 2035, expanding at a compound annual growth rate (CAGR) of 7.18% between 2026 to 2035
How Does Europe Lead the Global Medical Device Manufacturing Equipment Production Market?
Europe dominated the global medical device manufacturing equipment production market with a share of 38% in 2025. This is because of a strong base of established medical device OEMs and advanced manufacturing capabilities. The region's stringent regulatory standards drive demand for high-quality, compliant production equipment. Countries such as Germany, Switzerland, and Ireland serve as major medical technology hubs with strong technical expertise and innovation capacity. Additionally, supportive government policies promoting high-tech manufacturing and industrial automation further strengthen Europe's market leadership.
Germany Market Analysis
Germany is the major contributor to the European medical device manufacturing equipment production market due to its strong industrial base, advanced engineering capabilities, and leadership in precision manufacturing. The country hosts a large number of medical device manufacturers and equipment suppliers, supported by robust R&D infrastructure and high investment in automation and Industry 4.0 technologies.
Why is Asia Pacific Undergoing the Fastest Growth in the Market?
Asia Pacific is expected to grow at the fastest CAGR in the medical device manufacturing equipment production market during the forecast period, propelled by expanding healthcare infrastructure, increased government funding, and rising local medical device production. Countries such as China, India, and Japan are rapidly adopting advanced manufacturing technologies, including automation, robotics, and 3D printing systems. Growing domestic demand for cost-effective medical equipment is further supporting market expansion. Additionally, competitive labor costs and strengthening industrial capabilities are encouraging multinational manufacturers to establish production facilities in the region.
Medical Device Manufacturing Equipment Production Market Companies
- STERIS (U.S.)
- Nordson Corporation (U.S.)
- ENGEL (Austria)
- Zeiss Group (Germany)
- Multivac (Germany)
- Coherent Corp. (U.S.)
- KUKA AG (Germany)
- ABB (Switzerland)
- TRUMPF (Germany)
- OC Oerlikon Management AG (Switzerland)
- Arburg (Germany)
- Stratasys Ltd. (U.S.)
- Plasmatreat (Germany)
- Nikon Corporation (Japan)
- Mitutoyo Corporation (Japan)
Recent Developments
- In November 2025, Teva Pharmaceutical Industries Ltd. launched Teva Rise, an open innovation platform in the global setting to hasten partnerships in AI, Industry 4.0 smart manufacturing, digital health, and biotech. The program links startups and technology companies to their business units in order to co-create solutions in line with its Pivot to Growth strategy and propel patient-centered innovation. (Source: https://ir.tevapharm.com )
- In September 2025,the Indian Council of Medical Research licensed nine breakthrough health technologies to those industry partners who are private and who are based in India to hasten production there. The licensed technologies are the vaccines and diagnostic kits, which allow the local manufacturing and enhance the medtech ecosystem in the country. (Source: https://www.mohfw.gov.in )
- In August 2025, Vyame Holdings entered a memorandum of understanding with Embryyo Technologies to develop AI-enabled medical devices jointly. Its partnership is in interventional oncology and wearable technologies to strengthen innovation in next-generation healthcare solutions. (Source: https://www.businesswire.com )
Segments Covered in the Report
By Equipment Type
- Material Processing Equipment
- Sterilization & Cleaning Equipment
- Inspection & Quality Control Equipment
- Assembly & Packaging Systems
By Application
- Consumables & Disposables
- Sterile Devices Production
- Implants & Prosthetics Manufacturing
- Diagnostics Equipment Production
By End User
- OEM Manufacturers
- Contract Manufacturing Organizations (CMOs)
By Technology Adoption
- Automation & Robotics
- Additive Manufacturing / 3D Printing
- Conventional Production Systems
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting