What is the Medical Device Vigilance and Patient Safety Software Market Size in 2026?
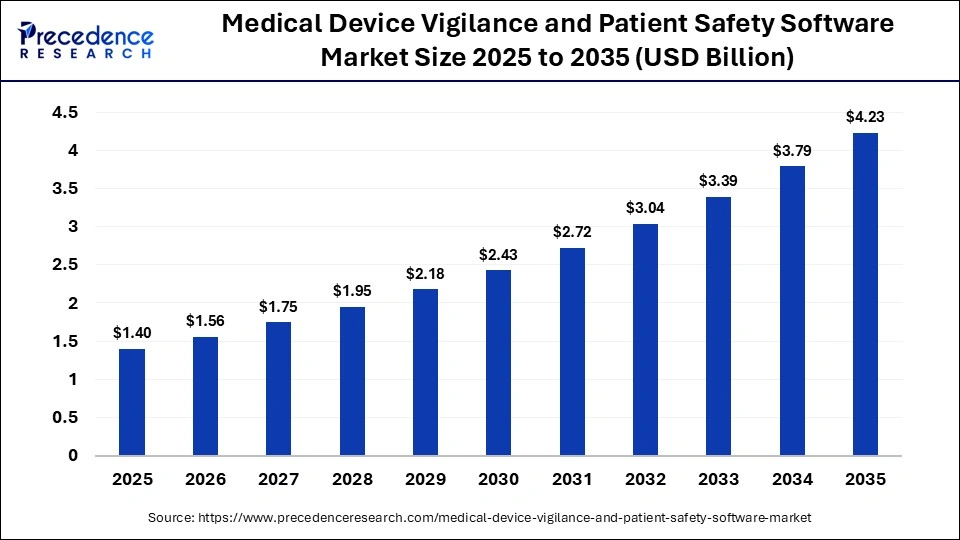
The global medical device vigilance and patient safety software market size was calculated at USD 1.40 billion in 2025 and is predicted to increase from USD 1.56 billion in 2026 to approximately USD 4.23 billion by 2035, expanding at a CAGR of 11.70% from 2026 to 2035. The growth of the medical device vigilance and patient safety software market is driven by the increasing regulatory compliance requirements, rising focus on patient safety, and the growing need for real-time monitoring and reporting of medical device-related adverse events.
Key Takeaways
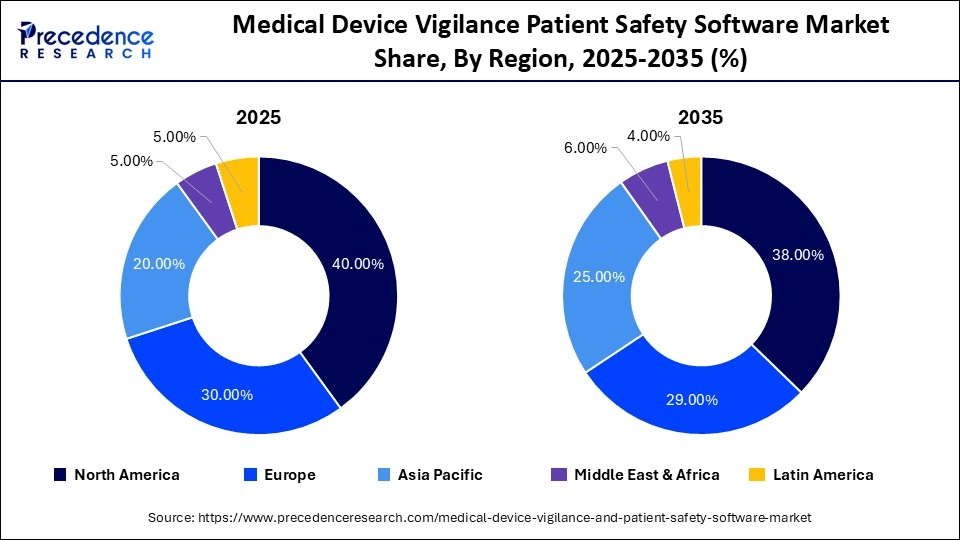
- North America led the market with w 40% share in 2025.
- Asia Pacific is expected to grow at the fastest CAGR of 16% between 2026 and 2035.
- By software type, the adverse event reporting software segment led the market with a 35% share in 2025.
- By software type, the risk management software segment is expected to grow at the fastest CAGR of 13.5% in the upcoming period.
- By deployment mode, the cloud-based software segment led the global market in 2025 with a 55% share and is observed to grow at the fastest CAGR of 14.5% between 2026 and 2035.
- By end-use, the medical device manufacturers segment led the global market in 2025 with a 45% share.
- By end-use, the contract research organizations (CROs) segment is expected to expand at the fastest CAGR of 14% during the forecast period.
Medical Device Vigilance and Patient Safety Software: Market Overview
The medical device vigilance and patient safety software market refers to solutions designed to monitor, report, and analyze adverse events and safety issues related to medical devices and patient care. These platforms help healthcare providers and manufacturers ensure regulatory compliance, improve risk management, and enhance patient outcomes through real-time data tracking and analytics. The market is growing due to increasing regulatory requirements, rising adoption of digital health systems, and a stronger focus on patient safety and quality of care.
Impact of Artificial Intelligence on the Medical Device Vigilance and Patient Safety Software Market
Artificial Intelligence (AI) significantly impacts the medical device vigilance and patient safety software market by enhancing the potential in identifying adverse events, predicting medication errors, assessing fall risks, and preventing pressure injuries. AI plays a crucial role in enhancing medical device safety, showcasing its advantages in improving patient safety, personalizing medical devices, and facilitating regulatory reporting. Additionally, AI-driven healthcare applications and software-based medical devices are currently in clinical use. The application of ML and neural technology in medical devices encompasses data classification and analysis for disease outbreaks, optimization of medical therapies, and diagnostic support, thereby enhancing software functionality and promoting market growth.
Major Market Trends
- Increasing Regulatory Reporting Requirements: Regulatory bodies are imposing increasingly stringent reporting requirements, prompting the adoption of comprehensive software solutions to enhance compliance and patient safety management.
- Strategic Collaboration Strengthening Market Position: Prominent companies are engaging in strategic collaborations to bolster their market position through partnerships between medical equipment suppliers and healthcare organizations, facilitating the broader adoption of advanced solutions.
- Need for AI-Driven Safety Analytics: Advanced AI and machine learning tools are increasingly being integrated into vigilance software to detect patterns in adverse event data and predict potential risks. This enables faster decision-making, improves accuracy in identifying safety signals, and reduces manual workload.
- Shift Toward Cloud-based Platforms: Organizations are rapidly adopting cloud-based solutions for scalability, real-time data access, and easier integration with existing healthcare IT systems. These platforms also support centralized reporting and collaboration across multiple locations, improving overall efficiency.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.40 Billion |
| Market Size in 2026 | USD 1.56 Billion |
| Market Size by 2035 | USD 4.23 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 11.70% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Software Type, Deployment Mode, End-Use, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Rising Focus on Patient Safety and Regulatory Compliance
The rising focus on patient safety is driving demand for software that can quickly detect, report, and manage adverse events related to medical devices and clinical care. At the same time, increasingly stringent regulatory requirements are compelling healthcare providers and manufacturers to adopt robust vigilance systems to ensure compliance and avoid penalties. Together, these factors are accelerating the adoption of advanced software solutions that improve transparency, accountability, and overall quality of care.
Restraint
High Setup Cost and Complex Software Management
The high initial setup costs and the complexity of software management present significant challenges. Frequent software updates and the requirement for various systems for risk management, adverse event reporting, and drug monitoring can be prohibitive. Effectively managing and maintaining medical device vigilance and patient safety software across healthcare providers can be both costly and operationally challenging. While vigilance specialists can oversee these tasks, healthcare providers need to be familiar with the fundamental aspects.
Opportunity
Adoption of New Technologies
The integration of advanced technologies and cybersecurity solutions is creating new growth opportunities in the Medical Device Vigilance and Patient Safety Software market. Increasing emphasis on cybersecurity across healthcare and home-care settings is driving the adoption of secure, real-time monitoring and reporting platforms. This trend supports safer, more efficient, and timely patient care while strengthening trust in digital health systems.
Segmental Insights
Software Type Insights
Medical Device Vigilance Patient Safety Software Market Share, By Software Type, 2025-2035 (%)
| Software Type | 2025 | 2035 | CAGR (%) |
| Adverse Event Reporting Software | 35.00% | 33.00% | 12.0% |
| Risk Management Software | 25.00% | 27.00% | 13.5% |
| Regulatory Compliance Management Software | 15.00% | 14.00% | 9.0% |
| Incident Management Investigation Software | 10.00% | 11.00% | 10.5% |
| Post-Market Surveillance Software | 10.00% | 10.50% | 11.0% |
| Others | 5.00% | 5.50% | 8.0% |
The Adverse Event Reporting Software Segment Held a 35% Market Share in 2025
The adverse event reporting software segment led the medical device vigilance and patient safety software market, accounting for approximately 35% share in 2025. This dominance stems from the critical need for effective adverse event reporting to maintain regulatory compliance, particularly given the increasing global focus on medical device safety in the post-market phase. This software empowers organizations to efficiently capture, report, and resolve adverse events while adhering to evolving regulatory standards, enabling systematic evaluation based on severity and regulatory criteria.
The risk management software segment held about 25% share of the market in 2025 and is anticipated to grow at the fastest CAGR of 13.5% in the coming years, driven by manufacturers' efforts to proactively identify and mitigate risks associated with device failures and potential patient harm. This software provides organizations with a clear understanding of risk exposure across multiple levels, making risk management an essential component of medical device vigilance.
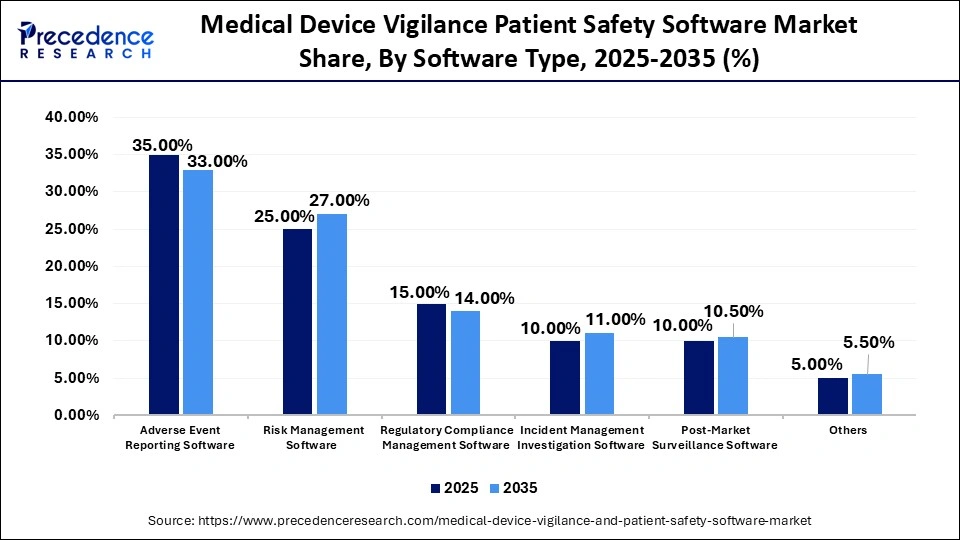
The regulatory compliance management software segment held approximately 15% market share in 2025 and is projected to grow at a CAGR of nearly 9% in the coming years, due to the necessity for manufacturers to adhere to industry standards and regulations amid heightened scrutiny from global regulatory authorities. Regulatory compliance management software serves as a digital solution that assists organizations in managing, monitoring, and maintaining compliance with relevant regulations and standards, while also facilitating the automation and standardization of compliance processes.
The incident management and investigation software segment held a 10% share of the market in 2025 and is expected to grow at a CAGR of 10.5% in the upcoming period. This growth is driven by the increasing necessity to track device-related incidents and ensure thorough investigations and corrective actions. Maintaining workplace safety and regulatory compliance is a top priority for organizations across various industries, and this software provides comprehensive tools for reporting, tracking, and resolving incidents.
Deployment Mode Insights
Medical Device Vigilance Patient Safety Software Market Share, By Deployment Mode, 2025-2035 (%)
| Deployment Mode | 2025 | 2035 | CAGR (%) |
| Cloud-based Software | 55.00% | 60.00% | 14.5% |
| On-premiseSoftware | 25.00% | 22.00% | 8.0% |
| Hybrid Software | 20.00% | 18.00% | 12.5% |
Why Did the Cloud-Based Software Segment Lead the Market in 2025?
The cloud-based software segment dominated the medical device vigilance and patient safety software market with the largest share of 55% in 2025 and is projected to continue its leadership during the forecast period. This is mainly due to the flexibility, scalability, and ease of access that cloud-based solutions offer for global operations. In addition, cloud-based pharmacovigilance systems streamline the entire adverse event reporting and risk management process. One of the most significant advantages of cloud-based solutions is their ability to scale efficiently as the volume of medical device vigilance data increases.
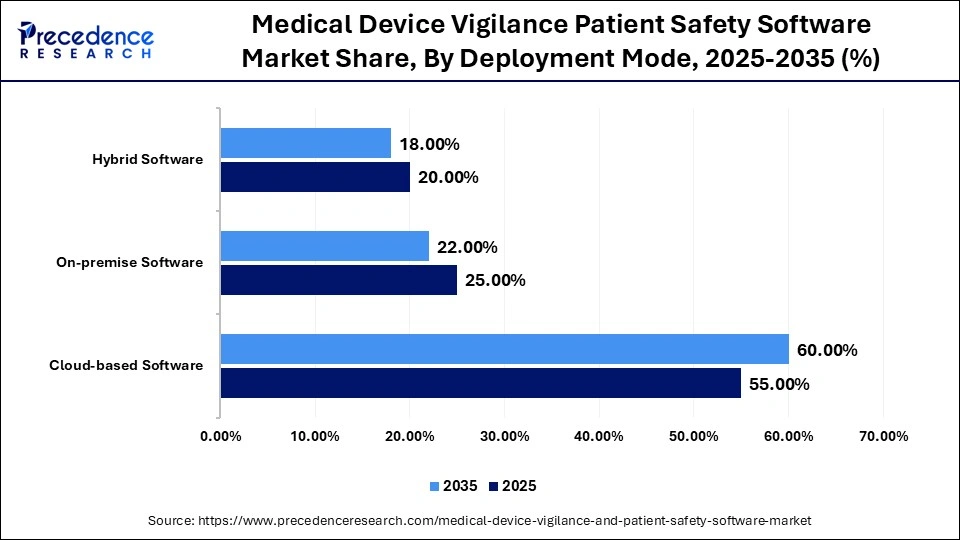
The on-premises software segment held the second-largest market share of 25% in 2025 and is expected to grow at a CAGR of 8% during the forecast period. On-premises solutions remain relevant for organizations with strict data security and privacy requirements. They offer greater control and customization but also require increased infrastructure investment. These solutions are commonly used in industries such as healthcare, where strict compliance with data protection laws such as HIPAA is mandatory.
The hybrid software segment held a 20% market share in 2025 and is expected to grow at a CAGR of 12.5% during the forecast period, driven by the increasing popularity of hybrid models, which combine the scalability of cloud solutions with the data control of on-premises systems for sensitive information. Hybrid solutions also provide greater customization and accessibility, further supporting their adoption.
End-Use Insights
Medical Device Manufacturers Dominated the Market in 2025 With a 45% Share
The medical device manufacturers segment led the medical device vigilance and patient safety software market in 2025 by holding approximately 45% revenue share. This is mainly due to the necessity of adhering to regulations and ensuring patient safety through effective vigilance and monitoring solutions. Vigilance reporting serves as a systematic approach to tracking and documenting adverse events, incidents, and safety concerns associated with medical devices post-market surveillance.
The contract research organizations (CROs) segment held around 20% share of the market in 2025 and is anticipated to grow at the fastest CAGR of 14% throughout the forecast period, driven by the rising adoption of safety software by CROs for effectively managing post-market surveillance and clinical trials involving medical devices. These software solutions facilitate vigilance reporting and documentation management, allowing CRO teams to handle various reporting requirements efficiently while maintaining connectivity with stakeholders and ethics committees through user-friendly systems.
Medical Device Vigilance Patient Safety Software Market Share, By End-Use, 2025-2035 (%)
| End-Use | 2025 | 2035 | CAGR (%) |
| Medical Device Manufacturers | 45.00% | 43.00% | 11.5% |
| Contract Research Organizations (CROs) | 20.00% | 22.00% | 14.0% |
| Regulatory Authorities | 10.00% | 11.00% | 8.5% |
| Hospitals & Healthcare Providers | 15.00% | 14.00% | 10.0% |
| Others | 10.00% | 10.00% | 8.0% |
The regulatory authorities segment held approximately 10% share of the market in 2025 and is expected to expand at a CAGR of about 8.5% between 2026 and 2035. This growth is driven by the implementation of patient safety software by regulatory bodies to oversee compliance with safety protocols and industry standards by manufacturers. Regulatory authorities must monitor protocols, reports, and documentation, and require updated software systems and ongoing maintenance to manage software updates effectively.
The hospitals and healthcare providers segment held a 15% market share in 2025 and is projected to grow at a CAGR of 10% during the forecast period. This growth is driven by the increasing adoption of software solutions by healthcare providers aimed at enhancing patient safety, particularly in monitoring and tracking adverse events associated with medical devices. Besides, this inclination is especially pronounced in hospitals engaged in clinical trials, where timely data updates and robust coordination with regulatory authorities render the use of these software solutions increasingly vital.
Regional Insights
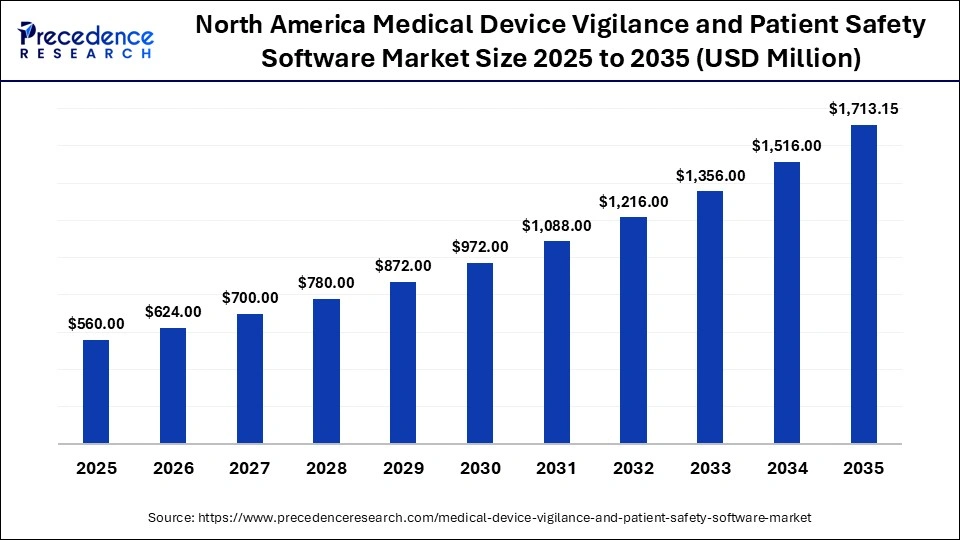
North America Medical Device Vigilance Patient and Safety Software Market Size and Growth 2026 to 2035
The North America medical device vigilance and patient safety software market size is estimated at USD 560.00 million in 2025 and is projected to reach approximately USD 1,713.15 million by 2035, with a 11.83% CAGR from 2026 to 2035.
North America Dominated the Market With a 40% Share in 2025
North America held a leading position in the medical device vigilance and patient safety software market in 2025, accounting for approximately 40% market share. This dominance is attributed to rigorous regulatory standards, widespread adoption of advanced safety technologies, and the presence of leading healthcare and device manufacturers. Additionally, a robust healthcare infrastructure, state-of-the-art medical facilities, and a high rate of medical device adoption contributed to the region's dominant position in the market.
U.S. Medical Device Vigilance Patient and Safety Software Market Size and Growth 2026 to 2035
The U.S. medical device vigilance and patient safety software market size is calculated at USD 420.00 million in 2025 and is expected to reach nearly USD 1,293.43 million in 2035, accelerating at a strong CAGR of 11.90% between 2026 and 2035.
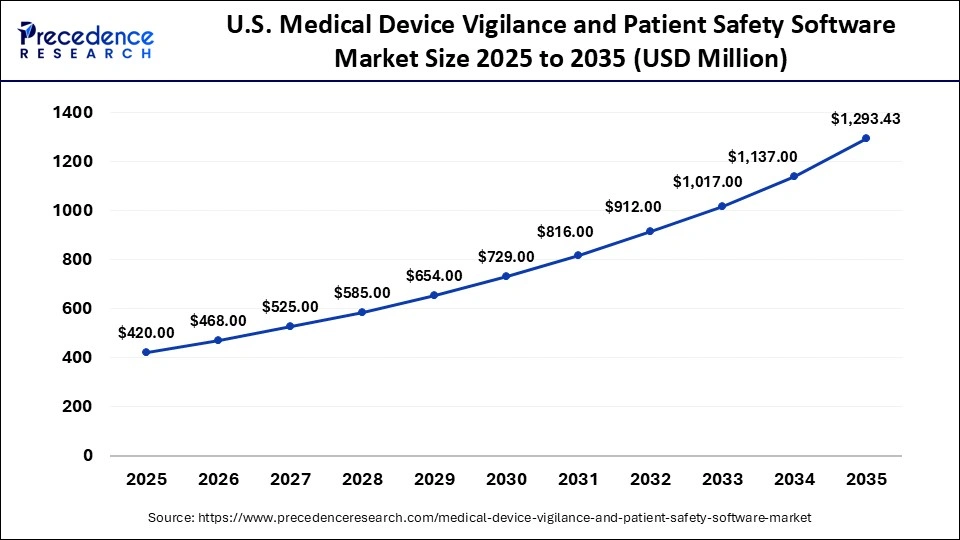
U.S. Medical Device Vigilance Patient and Safety Software Market Analysis
The U.S. is the leader in the North American market, bolstered by its strong healthcare infrastructure and the influential role of the U.S. FDA. The country benefits from a significant number of ongoing clinical studies, advancements in technology, substantial investments in research and development, and a landscape of innovation in software development, all supported by a favorable regulatory environment.
Europe: The Second-Largest Market
Europe is the second-largest market for medical device vigilance and patient safety software, holding nearly 30% share in 2025. The market in the region is projected to expand at a CAGR of 11% in the upcoming years, driven by stringent regulatory requirements, improved safety monitoring systems, and advancements in patient care infrastructure. Key regulatory bodies, including the European Medicines Agency (EMA), the Pharmacovigilance Risk Assessment Committee (PRAC), the Medical Device Regulation (MDR), and the In Vitro Diagnostic Regulation (IVDR), emphasize the necessity for robust vigilance systems.
Germany Medical Device Vigilance and Patient Safety Software Market Analysis
Germany leads the European market, characterized by a combination of established companies and innovative startups committed to enhancing patient safety through technological advancements. The market is well-positioned for growth as healthcare providers increasingly adopt software solutions to comply with regulatory standards and improve patient outcomes.
Asia Pacific: The Fastest-Growing Region
Asia Pacific captured approximately 20% market share in 2025 and is anticipated to grow at the fastest CAGR of 16% during the forecast period. This growth is supported by increased healthcare investments, heightened regulatory requirements, a surge in the adoption of medical devices in emerging economies, rising healthcare expenditures, a growing population, and an enhanced focus on patient safety. Additionally, the presence of regulatory bodies such as the MDA, NMPA, CDSCO, TGA, and HAS across the region further strengthens the market landscape.
China Medical Device Vigilance and Patient Safety Software Market Analysis
China leads the Asia Pacific market with a dynamic competitive landscape that includes both local and international players, promoting the adoption of medical devices across various medical specialties. Furthermore, a rising number of clinical trials and drug discovery initiatives are contributing positively to the market.
Medical Device Vigilance and Patient Safety Software Market Companies
- Medtronic plc
- Oracle Corporation
- Veeva Systems Inc.
- IQVIA Inc.
- Siemens Healthineers
- AbbVie Inc. (Allergan)
- GE Healthcare
- Hill-Rom Holdings, Inc.
- Becton, Dickinson and Company (BD)
- Stryker Corporation
- Bio-Rad Laboratories, Inc.
- Wolters Kluwer N.V.
- Pharmaceutical Product Development (PPD)
- Veristat, LLC
- PRA Health Sciences, Inc.
Recent Developments
- In June 2025, the Medical Devices (Post-market Surveillance Requirements) (Amendment) (Great Britain) Regulations 2024 (PMSR), which were enacted in October 2024, became effective in June 2025. This amendment updates the UK's Medical Devices Regulations 2002 (UK MDR). The primary goal of the PMSR is to enhance risk management and address safety concerns associated with medical devices and in-vitro diagnostic medical devices available in the Great Britain market.(Source: https://www.hoganlovells.com )
- In October 2025, Basil Systems announced the launch of its innovative MedTech Safety Signaling solution, a pioneering technology that revolutionizes post-market surveillance by transforming it into a proactive engine for risk detection and prevention.(Source: https://www.businesswire.com )
Segments Covered in the Report
By Software Type
- Adverse Event Reporting Software
- Risk Management Software
- Regulatory Compliance Management Software
- Incident Management and Investigation Software
- Post-Market Surveillance Software
- Others
By Deployment Mode
- Cloud-based Software
- On-premise Software
- Hybrid Software
By End-Use
- Medical Device Manufacturers
- Contract Research Organizations (CROs)
- Regulatory Authorities
- Hospitals and Healthcare Providers
- Others
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
