Gene Editing & mRNA Tool Kits Market Size and Forecast 2025 to 2034
The global gene editing & mRNA tool kits market witnessing rapid growth fueled by increased demand in healthcare, pharma, and life sciences R&D. The market is driven by rising demand for advanced research tools in precision medicine, cell therapy, and drug discovery.
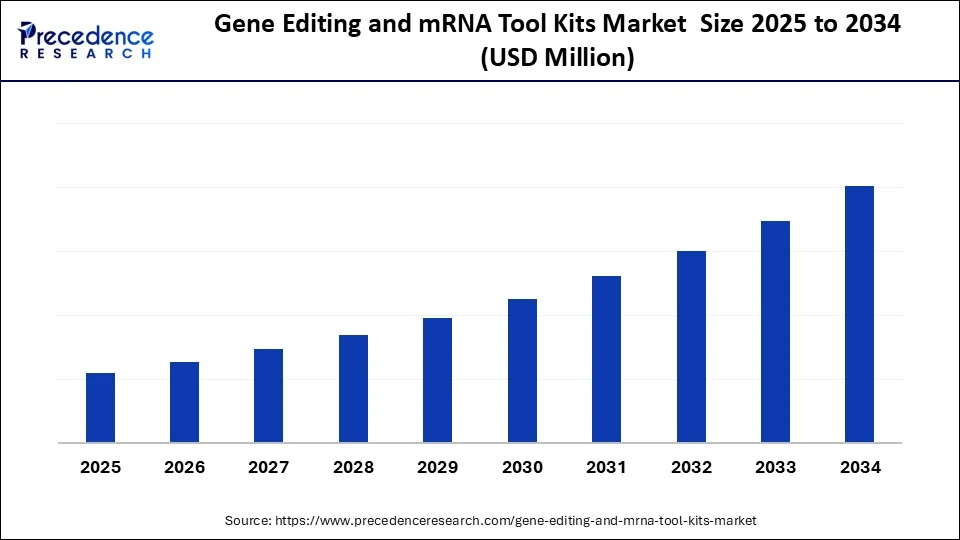
Gene Editing & mRNA Tool Kits Market Key Takeaways
- North America dominated the global gene editing & mRNA tool kits market with the largest market share of 41.5% in 2024.
- Asia Pacific is anticipated to witness the fastest growth during the forecasted years.
- By technology, the gene editing segment led the market in 2024.
- By technology, the mRNA tools segment is anticipated to show considerable growth over the forecast period.
- By application, the cell & gene therapy research segment captured the biggest market share of 38.9% in 2024.
- By application, the drug discovery & development segment is anticipated to show considerable growth over the forecast period.
- By end-user, the biotechnology & pharmaceutical companies segment held the highest market share of 46.2% in 2024.
- By end-user, the clinical laboratories segment is anticipated to show considerable growth over the forecast period.
How is AI Integration Transforming the Gene Editing & mRNA Tool Kits Market?
The coupling of artificial intelligence to gene editing, as well as mRNA tool kits, is transforming the study of genetics also precision medicine. The application can increase the accuracy and efficiency of genetic tool design by predicting off-target effects, guiding RNA patenting, and providing effective therapeutic delivery platforms. With AI algorithms, faster and cheaper development of drug discovery processes can be achieved through target identification and validation. In mRNA therapeutics, AI can assist in the development of more stable and effective molecules to use as vaccines and treatments, as well as to improve delivery. With the increasing adoption, AI will play a key role in terms of innovation in gene editing and mRNA tool kits.
Market Overview
The gene editing & mRNA tool kits market is a fast-growing sector of the biotechnology sector that is significant in research, clinical use, and industrial research. Gene editing aims to make lasting, targeted changes in the DNA of an organism. mRNA tool kits offer a temporary but convenient source of transiently altering the biological processes by instructing cells to express specific proteins. Collectively, these technologies are transforming the way scientists discover and test drugs, practice precision medicine, advance cell and gene therapy, synthetic biology, and molecular diagnostics.
The expansion of the gene editing & mRNA tool kits market is occurring as a result of a synergy of scientific innovation, growing investment in biotechnology, and the escalating need for customised treatment. The recent wave of gene and cell therapy product pipelines, particularly in cancer, rare disease, and infectious disease curation, has resulted in the heightened need for high-performance, reliable, and scalable editing and delivery technologies. An increase in investments by the governments, scientific institutions, and drug companies compounds more speed of innovation and commercialization. Besides, by using AI, bioinformatics, and the next-generation sequencing (NGS) integration, gene editing and mRNA technologies can be even more accurate and precise, further driving their adoption in academia and industry.
What Factors Are Fueling the Rapid Expansion of the Gene Editing & mRNA Tool Kits Market?
- Growing Demands of Precision Medicine: With the increasing demand for personalized drugs to treat genetic disorders, there is an increase in the demand for innovative tool kits. Researchers and clinicians are seeking to pioneer gene editing technologies and mRNA to produce patient-specific, efficacious, and safer therapeutics to fuel market growth.
- New Uses of the Imaging Agent in Drug Discovery and Development: Pharma and biotech companies are embracing gene editing and mRNA kits to accelerate their drug discovery and development pipeline. These tools can perform specific target authentication, pathway investigation, and help in medication testing, and shorten timelines and costs.
- Increases Investment in Research and Development: Genetic research is actively supported by governments, academic institutions, and independent investors to a large extent; thus, adopting advanced tool kits. Investment boosts genome editing platform innovation, mRNA innovations, and cell-related studies.
Market Scope
| Report Coverage | Details |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Technology, End User, Application, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Novel RNA Editing Capabilities Driving the Market
One of the greatest drivers in the gene editing & mRNA tool kits market is the appearance of new RNA editing methods. RNA editing does not involve permanent and generally irreversible modifications in the genome, as in usual DNA editing. This invention provides a safe alternative to researchers and clinicians, especially in acute and temporary diseases in which permanent genetic changes are not required or even desired. The significant strength of this approach is that the control of gene expression is not accompanied by a change in genetic structure; the risks of off-target effects, genomic instability, and the occurrence of unintended heritable changes are reduced. RNA editing is achieving some traction in therapeutics as an approach to cancer, infectious diseases, and neurological diseases. Safety, flexibility, and adaptability make it a growth driver of the fast-growing market.
Restraint
Lack of Standardized Guidelines in the Market
One of the factors that limits the growth of gene-modifying and mRNA tool kits market is the non-existence of default standards, guides, and regulatory certainty. Advanced applications of these technologies are used in the fields of vaccines, chronic disease therapies, regenerative medicine, and agricultural biotechnology, among others. Nevertheless, no single globally standardized framework exists to streamline their production pipeline, safety verification, or approval process. Such inconsistency is a major burden to the researchers, developers, and pharmaceutical companies that may have conducted laboratory innovations in their bid to commercialize them to clinical or commercial processes. Without standardized protocols, innovations, and patient access to breakthrough CRISPR-based and mRNA-based therapies, this enhancement is constrained.
Opportunity
Advancing Therapeutic Development through Gene Editing & mRNA Tool Kits
The major opportunity of the gene editing & mRNA tool kits market is its transformational role in the context of developing therapeutics. Delivery of mRNA that will express CRISPR systems into cells of a patient is a potentially safer and more specific approach to correcting genetic mutations than conventional gene therapy. Ongoing clinical trials investigate the use of these in such conditions as sickle cell disease (SCD), beta-thalassemia, and hereditary transthyretin amyloidosis. Moreover, CRISPR can be used outside a patient (ex vivo) to pre-modify stem cells or engineer T cells to implement cancer immunotherapies, such as CAR T-cells. In addition, new inventions like the use of CRISPR coupled with Cas13 to RNA-edit offer temporary and reversible changes in the genes, eliminating the risks involved in permanent alterations of the DNA. This is a potentially high-value approach in case of acute or reversible illness.
Technology Insights
Why Did the Gene Editing Segment Lead the Market in 2024 by Technology?
The gene editing segment dominated the market in 2024, with the CRISPR-Cas systems sub-segment holding 55.4% revenue share. The adoption of CRISPR-Cas has been accelerated by its extraordinary accuracy, affordability, and simplicity over historical methods such as TALEN and ZFN. The capacity to target and alter virtually any sequence in the genome has made it an invaluable tool in areas as diverse as functional genomics and agricultural biotechnology, and in the advancement of cutting-edge cell and gene therapy. This can be attributed to growing numbers of clinical trials that involve the application of CRISPR technology, combined with the constant improvement of the technology, to reduce cases of off-target editing, which should maintain its dominance in the particle segment.
The mRNA synthesis kits sub-segment of the mRNA tools segment dominated the market in 2024 with 42.7% of revenue Share. This market dominance is informed by the rising need to have high-quality, customizable mRNA constructs in both research and clinical applications. Synthesis kits are highly versatile because they give a researcher the materials required to create functional mRNA in a relatively short amount of time, useful in research involving the development of vaccines, protein research, and therapy development. The increasing demands of affordable and scalable platforms to perform personalized medicine, regenerative therapies, and new vaccine platforms have continued to give mRNA synthesis kits dominance in the global market.
The mRNA delivery reagents sub-segment of the mRNA tools segment is anticipated to have the fastest growth, due to the highly significant role of effective delivery systems in determining the generation of therapeutic effects. The growing number of mRNA-based drug candidates in the fields of oncology, infectious disease, and rare diseases has necessitated the need to have more advanced delivery reagents that are safer and have increased targeted delivery to a specific tissue. In addition, the strategic partnerships between the biotechnology companies and innovators in delivery technologies are facilitating the improvement of this segment at a faster rate, making the sub-segment the fastest-developing section of the mRNA tools market.
Application Insights
Why Did the Cell & Gene Therapy Research Segment Hold the Largest Share in 2024 by Application?
The cell & gene therapy research segment led the gene editing & mRNA tool kits market while holding a 38.9% share in 2024, driven by the fact that advanced molecular technologies are being deployed at a higher rate to tap into new advanced therapies for genetic disorders, cancers, and rare diseases. Scientists use CRISPR-based technologies, mRNA kit manufacturing systems, and delivery components in creating new treatment approaches, enhancing T-cell engineering, and streamlining CAR-T and TCR-T treatments. The rising number of clinical pipelines in gene and cell therapies, in addition to the growing number of investments made by the pharmaceutical companies and research centers, has greatly contributed to the dominance of the segment. Additionally, the regulatory framework in the treatment of genes by using the tool kits and breakthrough therapy designations has added more market confidence to this category, driving the demand in the segment.
The drug discovery & development segment is expected to grow at a significant CAGR over the forecast period due to the rising adoption of gene editing and mRNA technology to fast-track and simplify R&D. The tool kits enable precise disease modeling, validation of drug targets, and faster preclinical drug screen efforts by researchers, simply by reducing time and cost. Drug firms are spending on the development and application of CRISPR-Cas, mRNA, and high-throughput screening systems across their pipelines to keep up the track record of clinical trial success. In addition, the partnership between biotechnology companies and academic institutions is bringing innovation to therapeutic discovery.
End-user Insights
Why Did the Biotechnology & Pharmaceutical Companies Segment Dominate the Market in 2024 by End User?
The biotechnology & pharmaceutical companies segment held a 46.2% share in the market in 2024. This leadership position is made possible by an increasing dependence of the sector on modern molecular technologies to facilitate growth in terms of new drug development, efficient production techniques, and broadened pipelines. Such companies are actively investing in CRISPR-based technologies for editing the genomes, reagents of mRNA synthesis, and delivery reagents to expand the field of gene therapies, mRNA vaccines, protein replacement therapies, and personalized medications. Also, with significant funding and collaboration, pharma and biotechnology companies can increase their capacity in synthetic biology, regenerative medicine, and precision oncology. Biotechnology and pharmaceutical companies are expected to continue to remain the prime end-users of gene editing and mRNA tool kits due to increasing demand for novel therapeutics and competitive pressure to reduce drug development time.
The clinical laboratories segment is expected to grow substantially in the gene editing & mRNA tool kits market, owing to the rising usage of gene editing and mRNA-based techniques in diagnosis. Laboratories in clinical practice are incorporating CRISPR-Cas technologies with mRNA detection kits to create the next generation of diagnostic assays that can detect infectious diseases, cancer biomarkers, and genetic disorders that are faster and more accurately. Moreover, the increase in the need to personalize healthcare and provide early disease detection is forcing laboratories to implement new, state-of-the-art molecular tools to enhance customer outcomes. The co-operation among clinical laboratories, biotech start-ups, and healthcare providers is also increasing innovation and access.
Region Insights
Why Does North America Lead the Gene Editing & mRNA Tool Kits Market?
North America led the global gene editing & mRNA tool kits market with the highest market share of 41.5% in 2024. The market has a well-developed ecosystem of biotechnology and pharmaceutical businesses with well-equipped research facilities, sizeable investments in the sector, and an established presence of prominent industry players. Both the U.S. and Canada are centers of innovation and can be seen as the home to leading biotech clusters like Boston, San Francisco, and Toronto, among other clusters whose presence in those regions contributes to innovative advancements of CRISPR technologies, mRNA delivery systems, and gene editing toolbox systems. There is extensive use of CRISPR kits, reagents, and services in research laboratories and pharmaceutical companies across North America, which has further solidified its commanding position in the market.
North America is dominated by the biggest market share in the U.S., which is home to the most exciting companies in the field of biotechnology, proliferating clinical research, and an accommodating regulatory environment of innovation. Having invented mRNA-based vaccines, gene therapies, and personalized medicine products, the U.S. has also gained significant leverage in commercializing them with a high impact on the demand for more advanced gene editing toolkits. Moreover, strategic collaboration between U.S.-based biotech companies, pharmaceutical companies, and cloud/AI companies is also enhancing precision medicine and large-scale manufacturing potential.
Why is Asia Pacific undergoing the Fastest Growth in the Gene Editing & mRNA Tool Kits Market?
Asia Pacific is expected to realize the highest CAGR growth during the forecast period, with major drivers being growing research and development investments, government-sponsored biotechnology research, and blistering demand for precision medicine and advanced therapy treatment. Japan, China, South Korea, India, and Singapore are all coming up as important sources of innovation, with growing academic research bases, clinical trials, and associations with multinational pharmaceutical powerhouses. In addition, the conditions provided by good regulatory reforms and substantial funding by governments and private citizens are providing a favorable environment in which biotechnology innovation can take place.
China sits at the center of the Asian Pacific high-growth market and has an active national biotechnology agenda, heavy investment in research and development, and a keen interest in gene editing and mRNA. There are currently a number of innovative biotech companies and research centers in the country that are exploring uses of CRISPR technology, working on mRNA vaccines, and making contributions to agricultural biotechnology. China is rapidly becoming a world leader in the field of gene editing research, oncology, and pharmaceutical clinical trials of genetic therapies. China is also well-positioned to be an important driver of the Asia Pacific gene editing & mRNA tool kits market by increasing its domestic capabilities and expanding globally.
Value Chain Analysis
- R&D
The research area of R&D is the enhancement of methods of delivery, editing efficiency, and the safety of mRNA-based gene-editing toolkits. Ongoing innovation in CRISPR-Cas variations and base editors resolves constraints, such as off-target outcomes. Such developments are essential in developing safer and more effective clinical products in a number of disease areas.
Key Players: CRISPR Therapeutics, Intellia Therapeutics, Editas Medicine, Beam Therapeutics, Prime Medicine, Thermo Fisher Scientific, Moderna, BioNTech, CureVac, Arcturus Therapeutics,
- Clinical Trials and Regulatory Approvals
Clinical trials confirm the safety, efficacy, and therapeutic advantage in phases. Their potential is underlined by promising results in the genetic conditions of sickle cell disease and beta-thalassemia. Regulatory bodies deeply evaluate test results of effectiveness and safety to ascertain whether new treatments offer flexibility to make them available to the rest of the clinical and patient community.
Key Players: CRISPR Therapeutics, Editas Medicine, Intellia Therapeutics, Verve Therapeutics, Moderna and Pfizer/BioNTech, CureVac and Sanofi, AstraZeneca, and GSK
Regulatory Bodies: The FDA in the U.S., EMA in Europe
- Distribution to Hospitals, Pharmacies
The distribution networks depend on complex cold-chain processes, such as freezers and temperature-monitoring logistics, to ensure the stability of the products. Many of these therapies will also mandate specialty pharmacies or direct delivery to the hospital, which has advanced infrastructure to ensure the therapy is viable and can reach the patient safely and effectively, without the use of conventional medicines that have less advanced supply chain requirements.
Key Players: Specialized logistics providers (e.g., CryoPort, Marken
Healthcare Distributors: Cencora, Cardinal Health
Gene Editing & mRNA Tool Kits Market Companies

- Thermo Fisher Scientific
- Merck KGaA (Sigma-Aldrich)
- Agilent Technologies
- Illumina Inc.
- Takara Bio Inc.
- Qiagen N.V.
- Horizon Discovery (PerkinElmer)
- CRISPR Therapeutics
- Editas Medicine
- Intellia Therapeutics
- Synthego Corporation
- New England Biolabs (NEB)
- GenScript Biotech
- Bio-Rad Laboratories
- OriGene Technologies
- Integrated DNA Technologies (IDT)
- Lonza Group
- Precision Biosciences
- TriLink BioTechnologies
- Promega Corporation
Recent Developments
- In June 2025, Biomay introduced mRNA encoding of spCas9 as a new entry in its catalog product range in four forms of optimized mRNA products. These modifications are adapted to genome editing, such as TCR-T and CAR-T cell engineering. (Source: https://www.biomay.com)
- In March 2025, GenScript added a GMP-like manufacturing service to enhance its mRNA synthesis and formulation capabilities to support preclinical and IND-enabling studies. This offering brings biotechnology and wants to provide drug companies with accelerated and cost-effective access to high-quality quality regulatory-aware mRNA materials. (Source: https://www.fiercebiotech.com)
- In September 2024, Aurora Biosynthetics made its official debut as a ground-breaking RNA therapeutics manufacturing plant in the APAC region. This is supported by the NSW Government, RNA Australia, and Myeloid Therapeutics; the company is going to rapidly enter the RNA manufacturing market in the global environment. (Source: https://www.manmonthly.com.au)
Segments Covered in the Report
By Technology
- Gene Editing Tools
- CRISPR-Cas Systems
- TALENs
- Zinc Finger Nucleases (ZFNs)
- MegaTALs
- Others
- mRNA Tools
- mRNA Synthesis Kits
- mRNA Modification & Capping Kits
- mRNA Purification Kits
- mRNA Delivery Reagents
- Others
By Application
- Drug Discovery & Development
- Cell & Gene Therapy Research
- Clinical Diagnostics
- Agricultural Biotechnology
- Synthetic Biology
- Others
By End User
- Academic & Research Institutes
- Biotechnology & Pharmaceutical Companies
- Contract Research Organizations (CROs)
- Clinical Laboratories
- Others
By Region
- North America
- Europe
- Asia Pacific
- South America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content


 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
