What is the Infectious Disease Immunoassay Market Size in 2026?
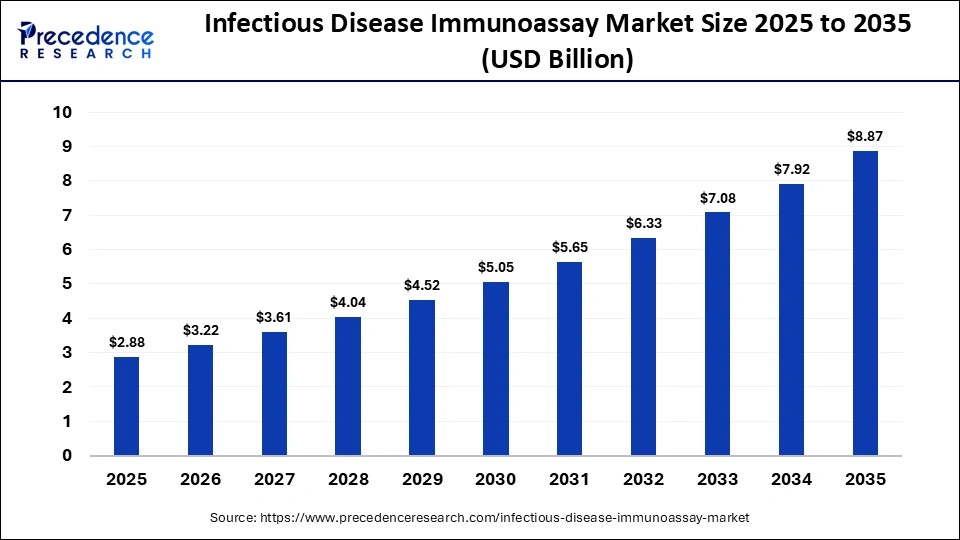
The global infectious disease immunoassay market size was calculated at USD 2.88 billion in 2025 and is predicted to increase from USD 3.22 billion in 2026 to approximately USD 8.87 billion by 2035, expanding at a CAGR of 11.90% from 2026 to 2035.The rising prevalence of infectious diseases, the urgent need for diagnosis, prevention, and treatment, and the expansion of diagnostic centers upsurge the importance of immunoassays.
Key Takeaways
- North America dominated the market in 2025, with a share of approximately 34.8%.
- Asia-Pacific is expected to grow at the fastest CAGR in the infectious disease immunoassay market during the forecast period.
- By product type, the reagents and kits segment held a dominant position in the market with a share of 56.8% in 2025.
- By product type, the software and data management solutions segment is expected to grow at the fastest CAGR of 12.3% in the market between 2026 and 2035.
- By technology, the chemiluminescent immunoassay segment led the global market with a share of 34.6% in 2025.
- By technology, the multiplex immunoassay segment is expected to grow with the highest CAGR of 13.4% in the market during the studied years.
- By test type, the antibody detection immunoassays segment registered its dominance over the global infectious disease immunoassay market with a share of 24.8% in 2025.
- By test type, the combined antigen and antibody immunoassays segment is expected to expand rapidly in the market with a CAGR of 10.8% in the coming years.
- By disease type, the hepatitis immunoassays segment dominated the global market with a share of 23.8% in 2025.
- By disease type, the respiratory infection immunoassays segment is expected to witness the fastest growth in the market with a CAGR of 10.1% over the forecast period.
- By application, the diagnostic testing segment held the largest revenue share of 38.7% in the market in 2025.
- By application, the epidemiological surveillance segment is expected to gain the highest market share with a CAGR of 12.1% between 2026 and 2035.
Infectious Disease Immunoassay: Rising at the Forefront of Modern Healthcare
The infectious disease immunoassay market comprises reagents, kits, assay consumables, calibrators, controls, analyzers, and related software used to detect antigens, antibodies, or immune markers associated with viral, bacterial, fungal, and parasitic infections. These tests are used across hospitals, diagnostic laboratories, blood banks, clinics, and public health settings for screening, diagnosis, surveillance, and treatment monitoring. The market is supported by broad immunoassay menus and established infectious disease testing portfolios across major automated diagnostic platforms.
Immunoassays are the heart of diagnostics and therapeutics, primarily performed in hospitals, blood banks, clinical laboratories, public health settings, diagnostic research centers, and personal healthcare settings. Some of the most commonly performed assays include RT-PCR, antigen rapid diagnostic tests, like fluorescence immunoassays, ELISA, CLIA, RIA, and LFIA. Among these, lateral flow immunoassays (LFIA) are widely adopted by clinical settings to detect viral diseases, including antigens, antibodies, and nucleic acids. LFIA offers rapidity, simplicity, cost-effectiveness, and portability, which make it a promising diagnostic tool to detect infectious diseases.
How is AI Transforming the Infectious Disease Immunoassay Market?
Artificial intelligence (AI) is transforming the infectious disease medicine sector through its novel features, such as pattern recognition, data processing, and intelligent decision-making. It offers accuracy, speed, efficiency, and precision in the detection of viral and animal infections in humans and other animals. AI-driven analysis of radiological and digital images and AI-enhanced point-of-care testing are significant technological advancements. AI technology enables precise interpretation of microbiological and medical data. AI contributes to high-throughput sequencing for pathogen surveillance through variant monitoring, pathogen identification, and investigation of outbreaks.
Infectious Disease Immunoassay Market Trends
- In Vitro Models for Infectious Disease Detection: Research has identified numerous emerging and re-emerging infectious diseases, owing to the growing population, environmental changes, international travel, and microbial adaptation and evolution. Traditional approaches in the medical field rely on 2D systems for detection and modelling, while modern healthcare has adopted 3D models like organoids, spheroids, and organ on a chip systems. These in vitro diagnostic models allow researchers to study host-pathogen interaction, enhance diagnosis, develop new therapeutic approaches, and strengthen control strategies for emerging outbreaks.
- Point-of-Care Infectious Disease Diagnostics: It is driven by innovations in biosensors, microfluidics, molecular amplification, and AI. Point-of-care (POC) diagnostic testing offers decentralized, rapid, and clinically actionable results. POC diagnostics are reshaping global health and the infectious disease immunoassays, while standing as the backbone of CRISPR-based platforms and molecular nucleic acid amplification tests. They achieve laboratory-grade sensitivity at POC. Accordingly, POC diagnostics have improved treatment initiation, improved early case detection, and outbreak containment for influenza, malaria, HIV, tuberculosis, and COVID-19.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 2.88 Billion |
| Market Size in 2026 | USD 3.22 Billion |
| Market Size by 2035 | USD 8.87 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 11.90% |
| Dominating Region | North America |
| Fastest Growing Region | Asia-Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product Type, Technology, Test Type, Disease Type, Application, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Improved Detection Tools
The major driving forces of the expanding infectious disease detection and diagnosis are a rapid shift towards decentralized point-of-care diagnostic testing, the huge adoption of immunoassay kits, home-based testing, and telemedicine integration. The immunoassay kits are linked to cloud platforms and smartphones, which allow patients to perform tests at home and provide real-time monitoring data to healthcare providers. These kits and reagents are adopted by urgent care clinics, pharmacies, and low-resource settings. There is a surging demand for simple-to-use and portable formats of rapid diagnostic kits like LFIA and fluorescence immunoassays that provide results in 30 minutes.
Restraint
High Cost
The financial barriers in the massively expanding infectious disease immunoassay industry include high initial investments, recurring operational costs, and inadequate reimbursement. Consumers are facing expenses for high-quality reagents, routine maintenance, specialized consumables, and calibration. Advanced automated immunoassay analyzers present affordability issues for small-to-medium-sized clinics and hospitals due to significant capital expenditure. Some regions hold low reimbursement rates for diagnostic tests that limit facilities from upgrading to newer immunoassay platforms.
Opportunity
Integration with Advanced Techniques
Emerging trends in immunoassay development are the integration of microfluidics and nanotechnology. These technologies offer different benefits, such as reduced sample volume, enhanced sensitivity, and rapid analysis. Gold nanoparticles, magnetic nanoparticles, and quantum dots are extensively employed in clinical practices to improve the detection limits of immunoassays. Microfluidic systems lead to reduced assay time and improved assay performance. These systems enable precise manipulation and control of samples and reagents.
Segmental Insights
Product Type Insights
Infectious Disease Immunoassay Market Share, By Product Type, 2025-2035 (%)
| Product Type | 2025 | 2035 |
| Reagents and Kits | 56.8% | 52.1% |
| Instruments and Analyzers | 24.7% | 22.9% |
| Calibrators | 6.1% | 5.8% |
| Controls | 7.4% | 6.8% |
| Software and Data Management Solutions | 5.0% | 12.4% |
The Reagents and Kits Segment Dominated the Market With 56.8% of Market Share in 2025
The reagents and kits segment dominated the infectious disease immunoassay market in 2025 with a revenue share of 56.8%, owing to the indispensable role of reagents, kits, and consumables for every diagnostic test. They are crucial in routine testing and outbreak monitoring. There is a recurring demand for consumables like detection substrates, antibodies, conjugates, and standards.
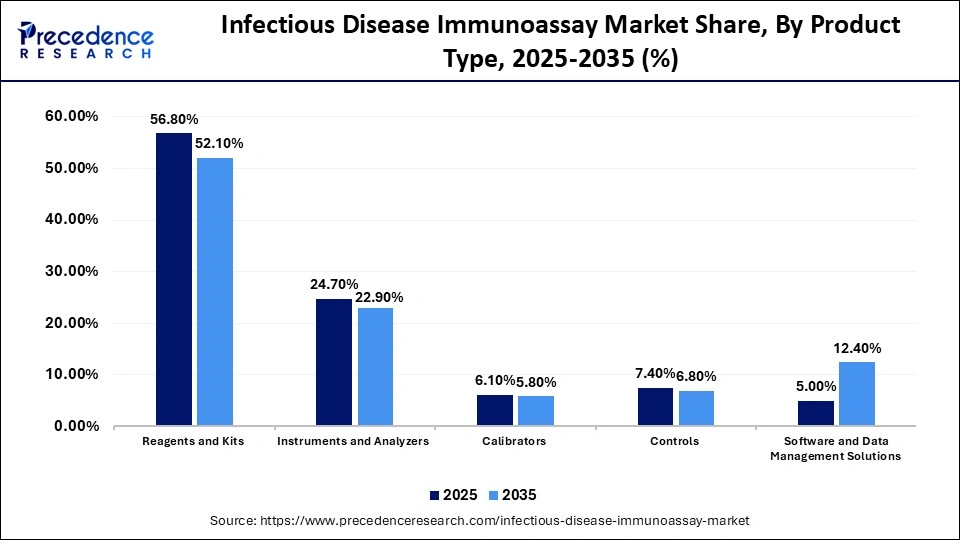
The software and data management solutions segment is expected to show the fastest growth with a CAGR of 12.3% over the forecast period, due to the integral role of clinical diagnostic laboratory management software in empowering the detection of infectious diseases. The laboratory information management system serves a crucial role in enhancing the accuracy and efficiency of clinical diagnostic laboratories. The clinical diagnostic laboratory management software offers a centralized platform to manage data related to infectious disease testing.
Technology Insights
The Chemiluminescent Immunoassay Segment Held the Largest Market Share of 34.6% in 2025
The chemiluminescent immunoassay segment held the largest revenue share of 34.6% in the infectious disease immunoassay market in 2025, owing to a rapid shift of researchers towards accurate diagnostic tests like CLIA to monitor infectious diseases. CLIA tests achieve low detection levels of proteins on diagnostic platforms. Research suggests that CLIA has been proven to be a suitable platform to monitor the progression of different stages of disease.
Infectious Disease Immunoassay Market Share, By Technology , 2025-2035 (%)
| Technology | 2025 | 2035 |
| Chemiluminescent Immunoassay | 34.6% | 36.2% |
| Enzyme Linked Immunosorbent Assay | 24.1% | 18.7% |
| Fluorescent Immunoassay | 11.4% | 10.7% |
| Radioimmunoassay | 3.2% | 1.1% |
| Rapid Lateral Flow Immunoassay | 18.7% | 17.6% |
| Multiplex Immunoassay | 8.0% | 15.7% |
The multiplex immunoassay segment is expected to gain the highest market share with a CAGR of 13.4% between 2026 and 2035, due to the importance of multiplex immunoassay platforms in the simultaneous detection of multiple analytes within a single assay. These assays provide a comprehensive diagnostic profile and include various multiplexing techniques. These techniques are microfluidic-based systems, bead-based assays, and planar arrays that enable high-throughput analysis of multiple biomarkers.
Test Type Insights
Infectious Disease Immunoassay Market Share, By Test Type , 2025-2035 (%)
| Test Type | 2025 | 2035 |
| Antigen Detection Immunoassays | 24.8% | 24.1% |
| Antibody Detection Immunoassays | 36.7% | 31.4% |
| Combined Antigen and Antibody Immunoassays | 18.4% | 24.7% |
| Confirmatory Immunoassays | 8.9% | 8.4% |
| Quantitative Immunoassays | 11.2% | 11.4% |
The Antibody Detection Immunoassays Segment Contributed the Highest Market Share of 24.8% in 2025
The antibody detection immunoassays segment accounted for the highest revenue share of 24.8% in the infectious disease immunoassay market in 2025, owing to the core functions of these assays in immunity and vaccination monitoring, and clinical diagnosis. They allow decentralized testing and assess the efficacy of vaccines. They are critical for screening chronic infections like HIV.
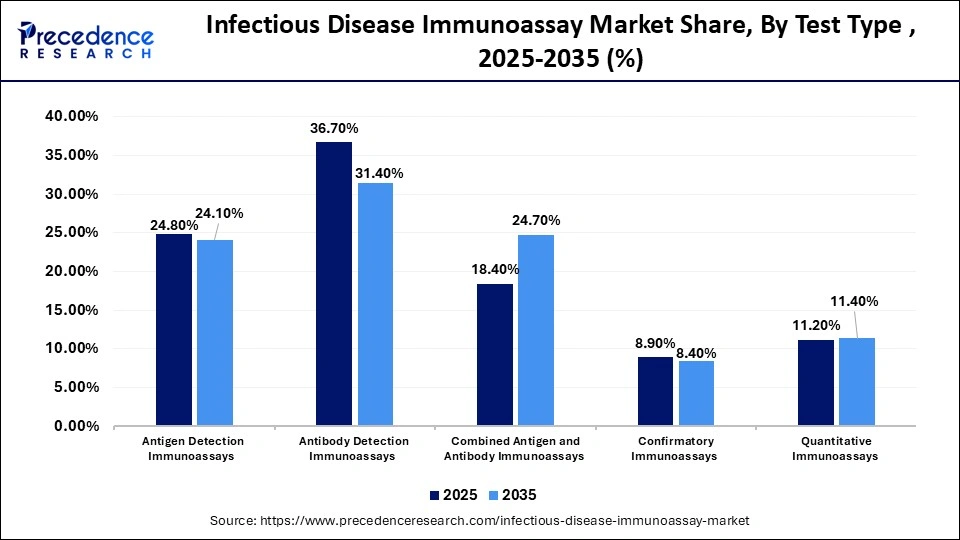
The combined antigen and antibody immunoassays segment is expected to witness the fastest growth in the market with a CAGR of 10.8% over the forecast period, due to the early detection of acute infection. Research suggests that a combined antigen and antibody testing approach improved the sensitivity of early detection of dengue virus infection. They enable intervention and treatment in a single clinical visit.
Disease Type Insights
The Hepatitis Immunoassays Segment Led the Market With 23.8% Market Share in 2025
The hepatitis immunoassays segment led the infectious disease immunoassay market with a share of 23.8% in 2025, owing to the urgent need to monitor chronic infections and detect both antigens and antibodies. Research and clinical settings have shifted towards hepatitis B testing. ELISA remains the leading technology to detect total hepatitis due to its scalability for central labs and blood banks.
Infectious Disease Immunoassay Market Share, By Disease Type, 2025-2035 (%)
| Disease Type | 2025 | 2035 |
| HIV Immunoassays | 18.6% | 17.1% |
| Hepatitis Immunoassays | 23.8% | 21.4% |
| Respiratory Infection Immunoassays | 16.7% | 19.6% |
| TORCH Infection Immunoassays | 9.4% | 8.5% |
| Sexually Transmitted Infection Immunoassays | 10.8% | 11.2% |
| Gastrointestinal Infection Immunoassays | 8.1% | 7.6% |
| Tropical and Vector Borne Disease Immunoassays | 7.4% | 8.6% |
| Healthcare Associated Infection Immunoassays | 5.2% | 6.0% |
The respiratory infection immunoassays segment is expected to expand rapidly in the market with a CAGR of 10.1% in the coming years, due to the importance of these assays to evaluate the effectiveness of vaccines by detecting antibody responses. The new assays help clinicians to differentiate between viral and bacterial infections. These POC tests allow for immediate clinical decisions in home and outpatient settings.
Application Insights
Infectious Disease Immunoassay Market Share, By Application, 2025-2035 (%)
| Application | 2025 | 2035 |
| Screening | 26.4% | 25.1% |
| Diagnostic Testing | 38.7% | 35.4% |
| Disease Monitoring | 12.6% | 12.0% |
| Blood Screening | 13.8% | 13.1% |
| Epidemiological Surveillance | 8.5% | 14.4% |
The Diagnostic Testing Segment Dominated the Market With a 38.7% Share in 2025
The diagnostic testing segment contributed the biggest revenue share of 38.7% in the infectious disease immunoassay market in 2025, owing to the wide applications of diagnostic tests for the detection of respiratory and chronic infections and evaluating the overall health. Technological advancements in diagnostics include next-generation platforms, such as CLIA, multiplexing, biosensors, and microfluidics. The rise of pharmacy-based and at-home testing is expanding access to diagnosis for respiratory viruses, HIV, etc.
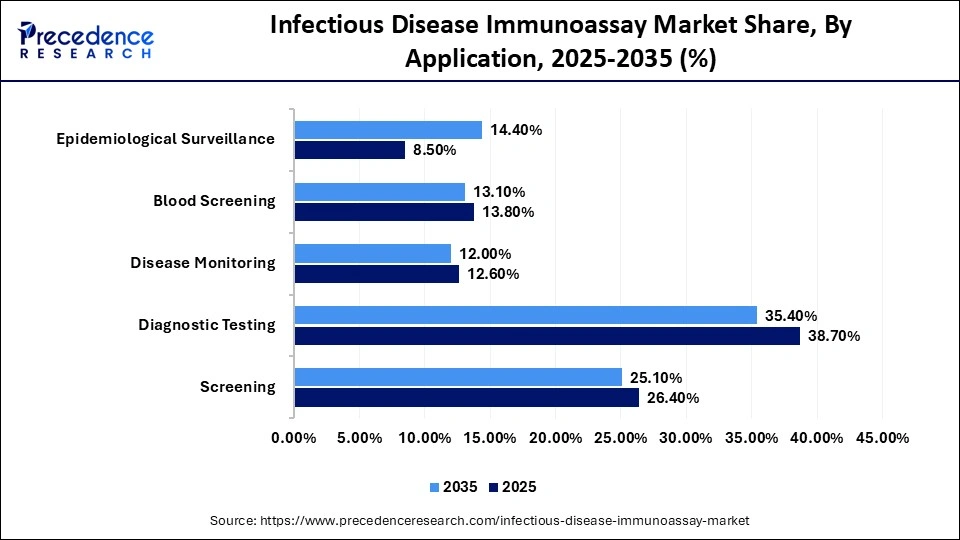
The epidemiological surveillance segment is expected to grow with the highest CAGR of 12.1% in the market during the studied years, due to the growing need to identify emerging pathogens, monitor the population community, and detect early outbreaks. The core immunoassay applications are antibody detection, antigen detection, and POC testing. Immunoassays are essential for serological surveillance to assess vaccine effectiveness and identify immunity gaps.
Regional Insights
North America Infectious Disease Immunoassay Market Size and Growth 2026 to 2035
The North America infectious disease immunoassay market size is estimated at USD 1,002.24 million in 2025 and is projected to reach approximately USD 3,131.11 million by 2035, with a 12.07% CAGR from 2026 to 2035.
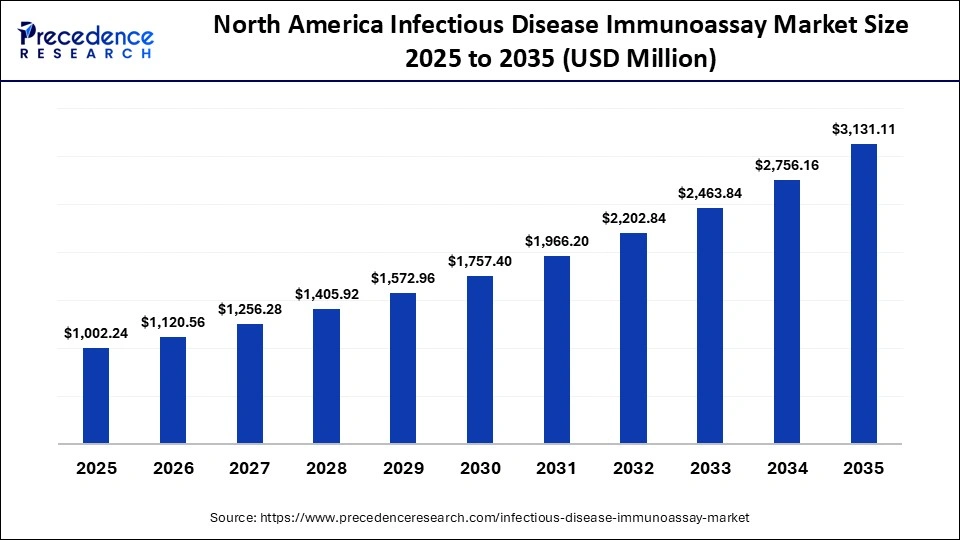
How North America Dominated the Infectious Disease Immunoassay Market?
North America held a major market share of 34.8% in 2025, owing to automation in laboratories, the rise of multiplex testing, and digital integration. The North American government programs conducted projects to advance surveillance and monitoring of food borne, climate sensitive zoonotic, and water borne diseases. These government programs aim to strengthen the monitoring and preparedness of infectious diseases and expand the infectious disease immunoassay industry. They target diagnostic platforms, respiratory multiplex testing, and climate sensitive diseases.
U.S. Infectious Disease Immunoassay Market Size and Growth 2026 to 2035
The U.S. infectious disease immunoassay market size is calculated at USD 751.68 million in 2025 and is expected to reach nearly USD 2,363.99 million in 2035, accelerating at a strong CAGR of 12.14% between 2026 and 2035.
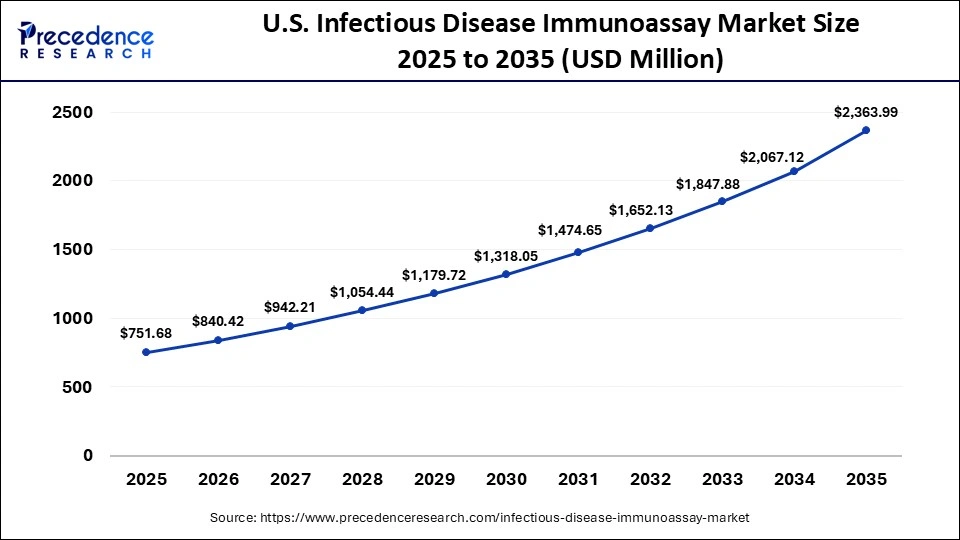
U.S. Market Analysis
The U.S. advances due to the favorable regulatory environment that facilitates the rapid approval and standardization of new diagnostic technologies. The U.S. government programs aim to build a centralized laboratory to validate, qualify, and perform standardized immunoassays to support clinical trials.
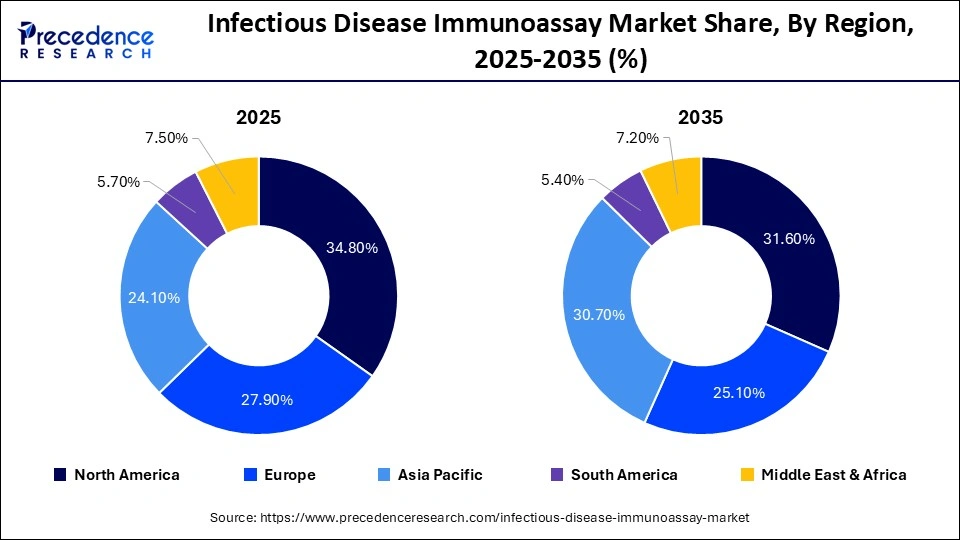
What is the Potential of the Infectious Disease Immunoassay Market in Asia-Pacific?
Asia-Pacific is expected to experience the fastest growth during the predicted timeframe, due to the rapid modernization of healthcare systems, the growing shift towards decentralized testing, and the expanding elderly population. The Asia-Pacific MedTech forum addressed regional strategies for medical technology across the expanding infectious disease immunoassay industry. It focuses on the role of advanced diagnostics like immunoassays to ensure resilient health systems and maintain global health security.
India Market Trends
The Indian industry is witnessing preventive healthcare awareness and a surge in routine health check-ups. India is building a network of Bio-Safety Level 3 laboratories. It is also strengthening its National Center for Disease Control (NCDC) to prevent, diagnose, and treat infectious diseases in the nation.
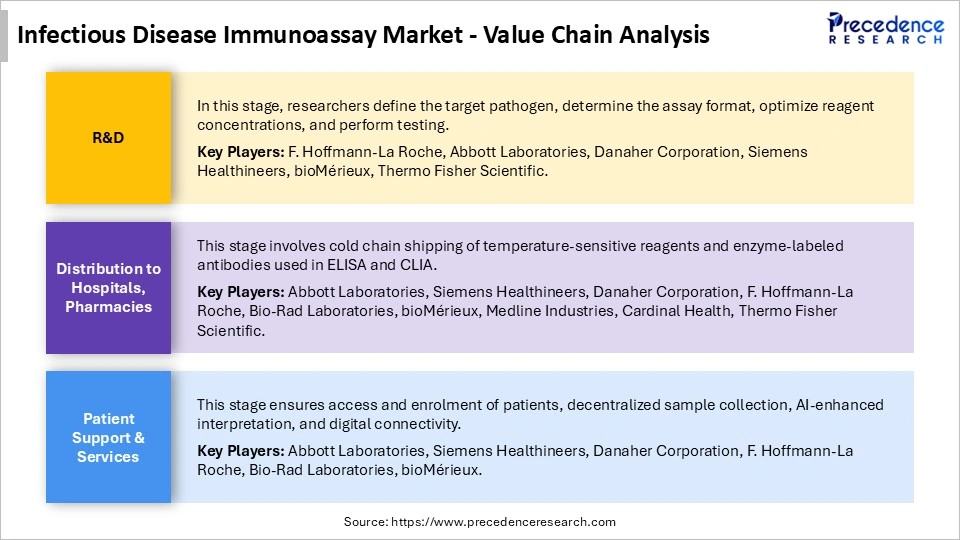
Infectious Disease Immunoassay Market Value Chain Analysis
Infectious Disease Immunoassay Market Companies
- Abbott Laboratories
- F. Hoffmann-La Roche Ltd.
- bioMérieux SA
- Siemens Healthineers AG
- Beckman Coulter, Inc.
- QuidelOrtho Corporation
- Thermo Fisher Scientific Inc.
- Danaher Corporation
- Sysmex Corporation
- Becton, Dickinson and Company
- DiaSorin S.p.A.
- Fujirebio Holdings, Inc.
- bioLytical Laboratories Inc.
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd.
- Ortho Clinical Diagnostics
Recent Developments
- In October 2025, Roche received the CE mark for the novel automated high-throughput Elecsys Dengue Ag test to diagnose dengue. This new dengue antigen test offers high clinical specificity and sensitivity for all four dengue virus serotypes.(Source: https://www.roche.com)
- In January 2025, bioMérieux SA strengthened its point of care presence with the acquisition of the immunoassay start-up SpinChip Diagnostics ASA. The startup offers a game changing immunoassay diagnostics platform for near patient testing that generates a result using a whole blood sample within 10 minutes.(Source: https://www.biomerieux.com)
Segments Covered in the Report
By Product Type
- Reagents and Kits
- Instruments and Analyzers
- Calibrators
- Controls
- Software and Data Management Solutions
By Technology
- Chemiluminescent Immunoassay
- Enzyme-Linked Immunosorbent Assay
- Fluorescent Immunoassay
- Radioimmunoassay
- Rapid Lateral Flow Immunoassay
- Multiplex Immunoassay
By Test Type
- Antigen Detection Immunoassays
- Antibody Detection Immunoassays
- Combined Antigen and Antibody Immunoassays
- Confirmatory Immunoassays
- Quantitative Immunoassays
By Disease Type
- HIV Immunoassays
- Hepatitis Immunoassays
- Respiratory Infection Immunoassays
- TORCH Infection Immunoassays
- Sexually Transmitted Infection Immunoassays
- Gastrointestinal Infection Immunoassays
- Tropical and Vector-Borne Disease Immunoassays
- Healthcare-Associated Infection Immunoassays
By Application
- Screening
- Diagnostic Testing
- Disease Monitoring
- Blood Screening
- Epidemiological Surveillance
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at [email protected]
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)


 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
