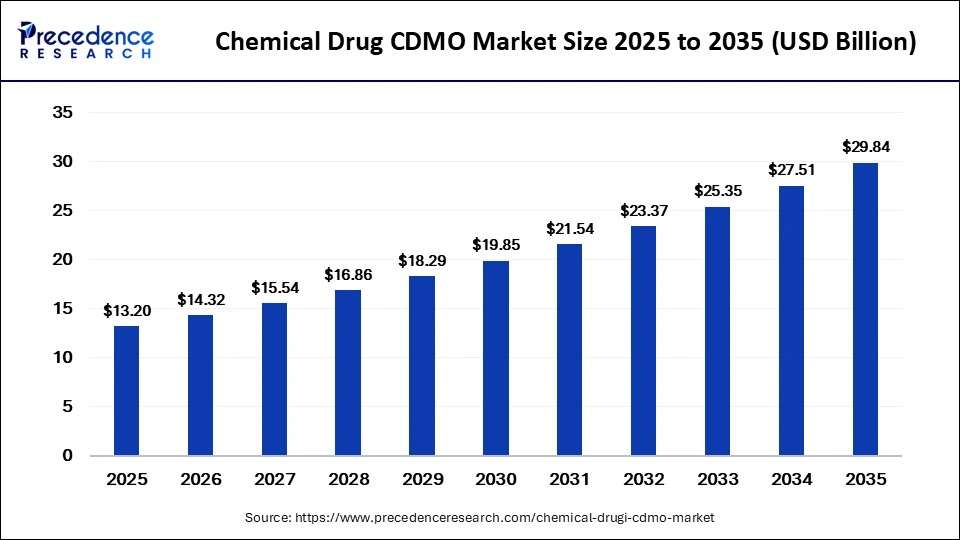
What is the Chemical Drug CDMO Market Size in 2026?
The global chemical drug CDMO market size accounted for USD 13.20 billion in 2025 and is predicted to increase from USD 14.32 billion in 2026 to approximately USD 29.84 billion by 2035, expanding at a CAGR of 8.50% from 2026 to 2035. The market growth is attributed to rising pharmaceutical outsourcing and the increasing need for cost-efficient manufacturing capacity.
Key Takeaways
- North America dominated the market with a 35% share in 2025.
- Asia Pacific is expected to grow at the fastest CAGR of 12.5% between 2026 and 2035.
- By service type, the API manufacturing segment contributed the highest market share of 50% in 2025.
- By service type, the formulation development segment held the second-largest market share of 15% in 2025 and is expected to grow at a significant CAGR of 10.5% between 2026 and 2035.
- By drug type, the branded drugs segment held a major market share of 40% in 2025.
- By drug type, the generic drugs segment held the second-largest market share of 35% in 2025 and is expected to grow at a 9.5% CAGR between 2026 and 2035.
- By API type, the small molecules APIs segment contributed the highest market share of 55% in 2025.
- By API type, the large molecule APIs (biologics, biosimilars) segment held the second-largest market share of 25% in 2025 and is expected to grow at a CAGR of 12.5% between 2026 and 2035.
- By outsourcing model, the full-service CDMOs segment contributed the highest market share of 60% in 2025.
- By the outsourcing model, the dedicated capacity CDMOs segment held a 40% share of the market in 2025 and is expected to grow at the fastest CAGR of 10% between 2026 and 2035.
- By therapeutic area, the oncology segment dominated the market in 2025.
Market Overview
Rising pharmaceutical outsourcing demand is a major factor boosting the growth of the chemical drug CDMO Market. Chemical drug CDMOs offer drug companies discovery, API manufacturing, formulation, and commercial drug production services on an outsourced basis. These services enable innovators and generic drug companies to reduce capital expenditure and efficiently enter products in the market.
The U.S Food and Drug Administration statistics reveal that about 90% of prescriptions in the U.S. utilize generic medicines, which likewise supports the high demand for outsourcing. Furthermore, the increasing pressure to reduce drug costs while scaling output remains a strong market growth drivers.
Impact of Artificial Intelligence on the Chemical Drug CDMO Market
CDMOs are increasingly leveraging artificial intelligence to identify optimal chemical synthesis routes, predict reaction outcomes, and shorten development cycles for small molecule drugs. These capabilities help reduce raw material consumption, improve production yields, and lower overall manufacturing costs. Machine learning is also being used for demand forecasting, production planning, and inventory management, enhancing supply chain efficiency and responsiveness.
Major Market Trends
- Growing Demand for High-Potency APIs: Increasing oncology and specialty therapies are driving investments in advanced containment manufacturing capabilities.
- Expansion of Global Clinical Pipelines: Rising small molecule drug candidates are boosting outsourced development and pilot-scale manufacturing demand.
- Increasing Digitalization: Increasing digitalization and AI integration is transforming CDMO operations, with applications in process optimization, predictive analytics, quality control, and supply chain management to reduce costs and development timelines.
- Demand for HPAPIs: Growing demand for high-potency APIs (HPAPIs) and complex molecules is driving CDMOs to invest in specialized containment facilities, advanced safety systems, and highly skilled technical capabilities, especially for oncology and targeted therapies.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 13.20 Billion |
| Market Size in 2026 | USD 14.32 Billion |
| Market Size by 2035 | USD 29.84 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 8.50% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Service Type, Drug Type, Therapeutic Area, API Type, Outsourcing Mode, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Increasing Patent Expirations
Increasing patent expirations across major therapeutic areas are expected to significantly boost outsourcing demand in the chemical drug CDMO industry. Global pharmaceutical companies are preparing for the launch of generic versions of cardiovascular, CNS, oncology, and metabolic drugs that are expected to lose exclusivity. These launches require rapid API development, bioequivalent formulation development, validation batches, and commercial-scale production support. CDMOs help sponsors avoid large capital investments in dedicated manufacturing facilities while meeting tight market entry timelines.
Restraint
Stringent Regulatory Compliance
Stringent regulatory compliance requirements are expected to reduce profitability in the chemical drug CDMO industry. Manufacturers must maintain certified equipment, robust documentation systems, strong data integrity controls, and continuous employee training to meet global standards. As a result, rising compliance costs are constraining margin expansion and increasing pressure on operational budgets.
Opportunity
Rising Regulatory Complexity Worldwide
Rising regulatory complexity worldwide is anticipated to create immense opportunities for the market in the coming years. Compliance with stringent quality standards, strong data integrity requirements, and validated systems mandated by regulatory bodies such as the U.S. FDA, EMA, MHRA, and PMDA has become increasingly critical. Smaller sponsors often lack the internal resources to manage multi-market regulatory compliance. In contrast, established CDMOs offer inspection-ready facilities, experienced teams, and well-documented processes that reduce compliance risks and ensure reliable execution.
Segment Insights
Service Type Insights
Chemical Drug CDMO Market Share, By Service Type, 2025-2035 (%)
| Service Type | 2025 | 2035 | CAGR (%) |
| API Manufacturing | 50.00% | 47.00% | 9.50% |
| Formulation Development | 15.00% | 16.00% | 10.50% |
| Contract R&D | 15.00% | 16.00% | 11.00% |
| Regulatory Services | 10.00% | 10.50% | 8.50% |
| Packaging Services | 10.00% | 10.50% | 7.50% |
The API Manufacturing Segment Held a 50% Market Share in 2025
The API manufacturing segment dominated the chemical drug CDMO market with a major share of 50% in 2025. This is mainly due to rising demand for active pharmaceutical ingredients across both generic and branded medicines. There is high demand for cardiovascular, oncological, CNS, and anti-infective APIs, which boost the need for large-scale, high-efficiency CDMO capabilities and robust quality systems.
The formulation development segment was the second-largest shareholder in 2025, holding a 15% share, and is expected to grow at a significant CAGR of 10.5% between 2026 and 2035. This growth is driven by increasing demand for differentiated dosage forms and accelerated product development. Pharmaceutical companies are focusing on formats such as orally disintegrating tablets, injectables, and other patient-centric formulations to improve adherence. CDMOs play a key role in excipient selection, stability studies, bioavailability enhancement, and scale-up planning.
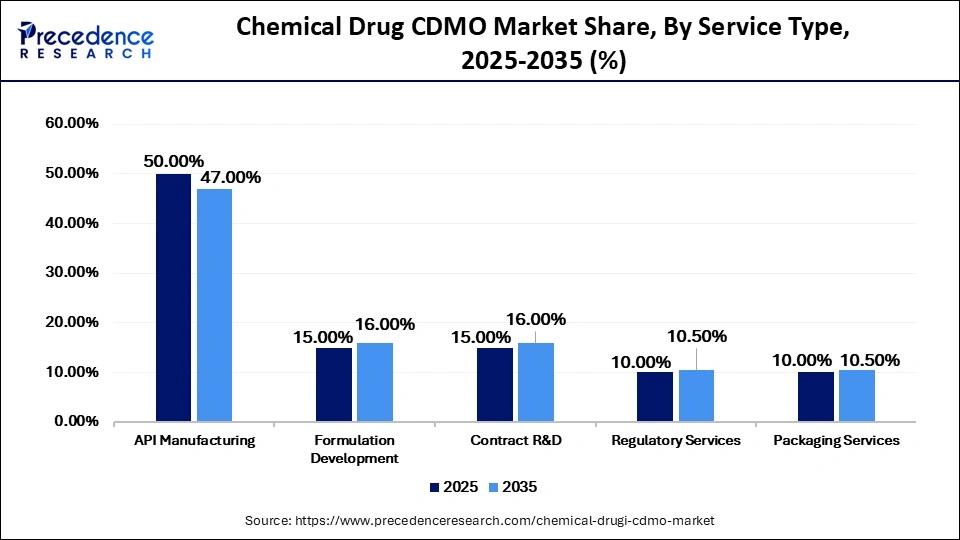
The contract R&D segment held a 15% share of the market in 2025 and is expected to grow at a 11% CAGR between 2026 and 2035. This growth is supported by increasing reliance on external scientific expertise and agile innovation to advance pharmaceutical pipelines. Small and mid-sized biotech firms, in particular, depend heavily on CDMOs for chemistry development, early-stage research, and milestone-based project execution.
The regulatory services segment contributed the 10% market share in 2025 and is estimated to grow at a strong CAGR of 8.5% over the projected period, driven by increasingly stringent global compliance requirements across regions. CDMOs offering integrated regulatory support alongside manufacturing capabilities are improving client retention and expanding project scope.
The packaging services segment held a 10% share of the market in 2025 and is expected to grow at a CAGR of 7.5% in between 2026 and 2035. This growth is supported by rising demand for secure, compliant, and market-ready packaging formats for finished pharmaceuticals. Blister packs, bottles, unit-dose packaging, tamper-evident solutions, and serialization remain critical for distribution and regulatory compliance. Increasing logistical complexity and tighter product release timelines are further driving outsourcing of packaging services.
Drug Type Insights
Chemical Drug CDMO Market Share, By Drug Type, 2025-2035 (%)
| Drug Type | 2025 | 2035 | CAGR (%) |
| Branded Drugs | 40.00% | 35.00% | 7.00% |
| Generic Drugs | 35.00% | 37.00% | 9.50% |
| Biosimilars | 15.00% | 18.00% | 14.00% |
| OTC Drugs | 10.00% | 10.00% | 6.50% |
The Branded Drugs Segment Dominated the Market With a 40% Share in 2025
The branded drugs segment dominated the chemical drug CDMO market with a major share of 40% in 2025, due to the strong commercial value of patented therapies across specialty disease areas. Innovator companies continue to outsource selected manufacturing activities to optimize launch timelines and enhance production flexibility. These high-value contracts are typically awarded to CDMOs with advanced containment capabilities and strong global regulatory compliance.
The generic drugs segment held the second-largest market share of 35% in 2025, due to the growing shift toward affordable treatment options. Pharmacy substitution policies, public tenders, and cost-driven procurement strategies continue to drive demand for generics. Expanding access to medicines in emerging markets further supports growth through budget-sensitive healthcare initiatives. According to Medicines for Europe, generic medicines account for around 70% of dispensed medicines in Europe by volume, highlighting strong demand for large-scale API sourcing.
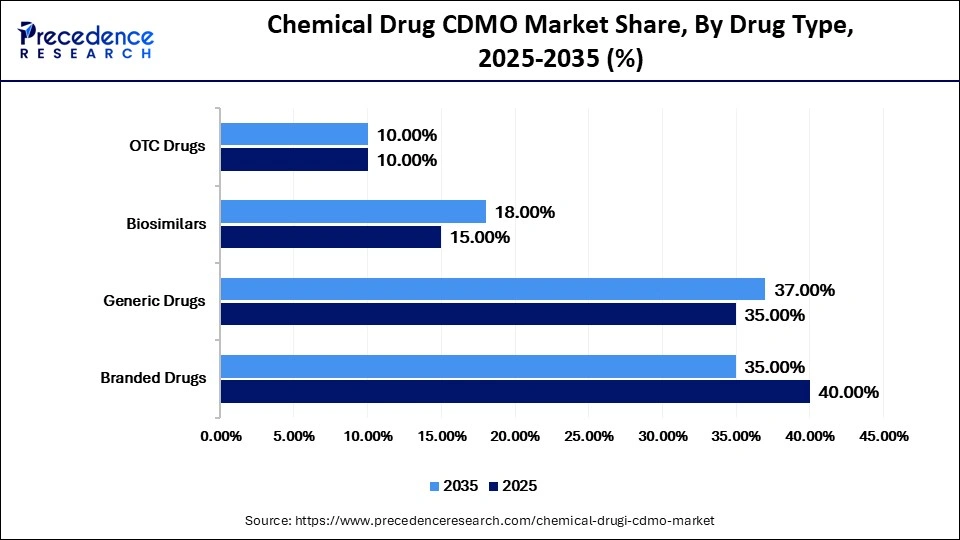
The biosimilars segment held a 15% share of the market in 2025 and is expected to grow at the highest CAGR of 14% during the forecast period, due to the increasing regulatory approvals for biosimilars and rising adoption of cost-effective biologic alternatives. Healthcare systems are actively promoting biosimilars to reduce expenditure on high-cost reference biologics. Although biosimilars are biologics, chemical drug CDMOs also support related activities such as excipient supply, packaging, fill-finish operations, and hybrid manufacturing requirements.
The OTC drugs segment held a 10% market share in 2025 and is estimated to grow at a strong CAGR of 6.5% over the projected period, owing to the increasing consumer preference for self-medication and easy access to non-prescription treatments. Brand owners increasingly outsource tablet manufacturing, liquid formulations, sachets, and packaging services to CDMOs to reduce internal operational complexity.
API Type Insights
Chemical Drug CDMO Market Share, By API Type, 2025-2035 (%)
| API Type | 2025 | 2035 | CAGR (%) |
| Small Molecule APIs | 55.00% | 52.00% | 8.00% |
| Large Molecule APIs (Biologics, Biosimilars) | 25.00% | 28.00% | 12.50% |
| Peptide-based APIs | 10.00% | 12.00% | 14.00% |
| Monoclonal Antibodies (mAbs) | 10.00% | 8.00% | 13.00% |
Why Did the Small Molecule APIs Segment Dominate the Market in 2025?
The small molecule APIs segment dominated the chemical drug CDMO market with the largest share of 55% in 2025. This is mainly due to the widespread use of chemically synthesized medicines across major therapy areas. Small molecule APIs remain essential for treating a wide range of diseases, and their well-established, scalable, and cost-efficient manufacturing processes make them highly suitable for large-scale outsourcing.
The large molecule APIs (Biologics, Biosimilars) segment held the second-largest market share of 25% in 2025 and is expected to grow at a significant CAGR of 12.5% in the upcoming period. This growth is driven by increasing adoption of recombinant proteins and biosimilars in oncology, autoimmune, inflammatory, and immunology therapies. CDMOs with advanced biologics capabilities are securing long-term, strategic partnerships with global pharmaceutical companies. As reported by the EMA, more than 100 biosimilar medicines had been approved in Europe by 2025, reflecting sustained demand for outsourced biologics development and manufacturing.
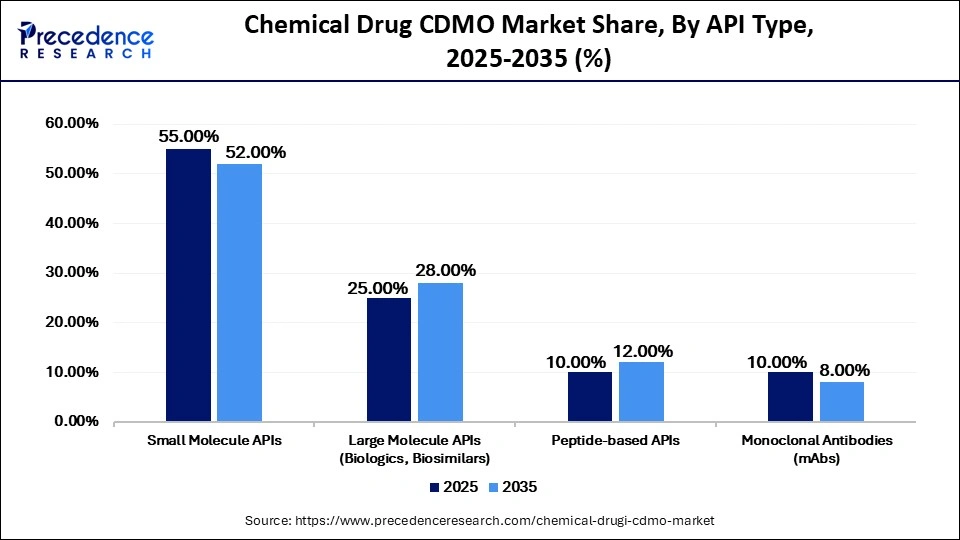
The peptide-based APIs segment held a 10% share of the market in 2025 and is expected to grow at the highest CAGR of 14% during the projection period, driven by rising investment in specialty drug pipelines and expanding peptide-based therapies. Growing research in rare diseases is further contributing to the demand for specialized CDMO capabilities in this segment.
The monoclonal antibodies (mAbs) segment held a 10% market share in 2025 and is estimated to grow at a strong CAGR of 13% over the projected period. This is mainly due to the rising need for high-capacity bioreactors and aseptic fill-finish capabilities, which remain critical strengths of CDMOs. Continued lifecycle demand for established blockbuster antibodies is further sustaining outsourcing requirements.
Outsourcing Model Insights
Chemical Drug CDMO Market Share, By Outsourcing Model, 2025-2035 (%)
| Outsourcing Model | 2025 | 2035 | CAGR (%) |
| Full-service CDMOs | 60.00% | 58.00% | 8.00% |
| Dedicated Capacity CDMOs | 40.00% | 42.00% | 10.00% |
The Full-service CDMOs Segment Dominated the Market With a 60% Share in 2025
The full-service CDMOs segment dominated the chemical drug CDMO market with a share of 60% in 2025. This is because full-service CDMOs provide end-to-end services from R&D to commercial production, meeting the diverse needs of pharmaceutical companies. The increasing trend toward single-partner outsourcing in pharmaceutical value chains also bolstered segmental dominance.
Drug sponsors are increasingly preferring providers that offer integrated services, including route development, API production, formulation, packaging, and commercial supply under a single platform. This end-to-end model reduces vendor handovers, shortens project timelines, and improves overall operational visibility.
The dedicated capacity CDMOs segment held the second-largest market share of 40% in 2025 and is expected to grow at the fastest CAGR of 10% between 2026 and 2035. The growth of this segment is driven by the increasing demand for dedicated facilities to produce high-value drugs and biologics. This model is particularly suited for sensitive molecules, large-scale product launches, and therapies requiring long-term, uninterrupted supply.
Therapeutic Area Insights
The Oncology Segment Dominated the Market in 2025
The oncology segment dominated the chemical drug CDMO market in 2025, due to the continuous expansion of global cancer treatment pipelines. Pharmaceutical companies are increasingly outsourcing the manufacturing of cytotoxic APIs, targeted small molecules, oral solid dosage forms, and sterile injectables to specialized CDMOs. Strong innovation activity in oncology continues to reinforce its position as the leading therapeutic area for outsourced chemical drug production.
The infectious diseases segment held the second-largest market share in 2025, driven by the sustained global demand for antibiotics, antivirals, antifungals, and antiparasitic medicines. The need to ensure stable and reliable supply of anti-infective therapies further highlights the critical role of CDMOs in this segment.
The cardiovascular segment is expected to grow at a significant CAGR between 2026 and 2035, driven by the rising global burden of hypertension, heart failure, and lipid disorders. High prescription volumes of statins, antihypertensives, anticoagulants, and antiplatelet drugs are supporting long-term manufacturing partnerships with CDMOs.
The neurology segment is expected to register a notable growth during the forecast period, fueled by increasing demand for treatments for epilepsy, Parkinson's disease, Alzheimer's disease, and migraine disorders. Pharmaceutical companies are increasingly relying on CDMOs for complex oral solid formulations and specialized analytical capabilities required for neurological therapies.
Regional Insights
North America Chemical Drug CDMO Market Size and Growth 2026 to 2035
The North America chemical drug CDMO market size is estimated at USD 9.90 billion in 2025 and is projected to reach approximately USD 22.53 billion by 2035, with a 8.57% CAGR from 2026 to 2035.
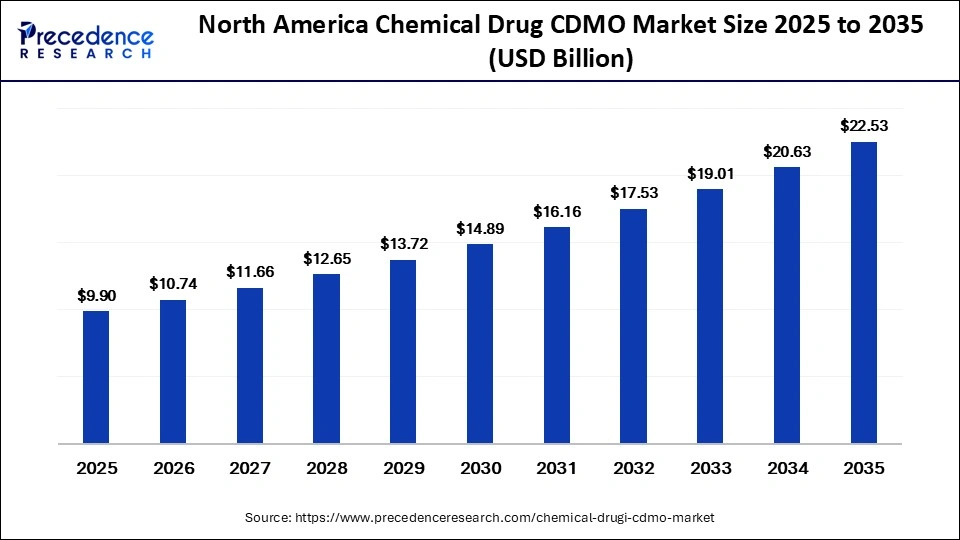
North America Held a 35% Market Share in 2025
North America led the chemical drug CDMO market, capturing the largest share of 35% in 2025. This dominance is driven by the presence of leading pharmaceutical innovators, specialty drug developers, and advanced manufacturing infrastructure. The U.S. serves as the primary growth hub in the region, supported by strong demand for outsourced API manufacturing, formulation development, and commercial packaging services.
According to the Association of Accessible Medicines, more than 3.9 billion generic prescriptions were dispensed in the U.S. in 2025, reinforcing the need for large-scale contract manufacturing. The region is home to major CDMOs such as Thermo Fisher Patheon, Catalent Pharma Solutions, Pfizer CentreOne, and Cambrex, which also contributed to market dpominance.
U.S. Chemical Drug CDMO Market Size and Growth 2026 to 2035
The U.S. chemical drug CDMO market size is calculated at USD 7.43 billion in 2025 and is expected to reach nearly USD 17.01 billion in 2035, accelerating at a strong CAGR of 8.64% between 2026 and 2035.
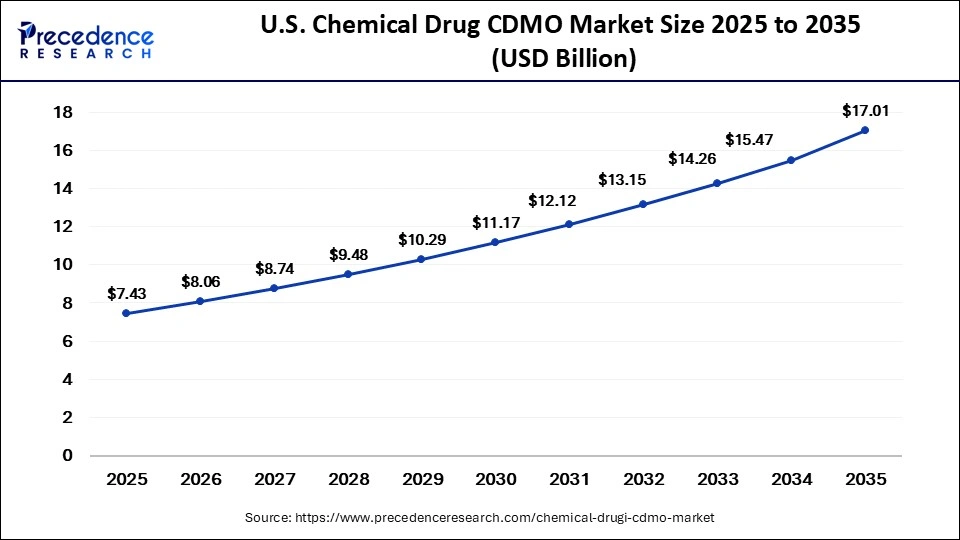
U.S. Chemical Drug CDMO Market Analysis
The U.S. leads the market within North America due to its strong pharmaceutical innovation ecosystem and highly advanced outsourcing network. Pharmaceutical companies are increasingly relying on domestic CDMOs for API production, sterile injectables, and clinical supply manufacturing, strengthening the country's leadership in the global CDMO landscape.
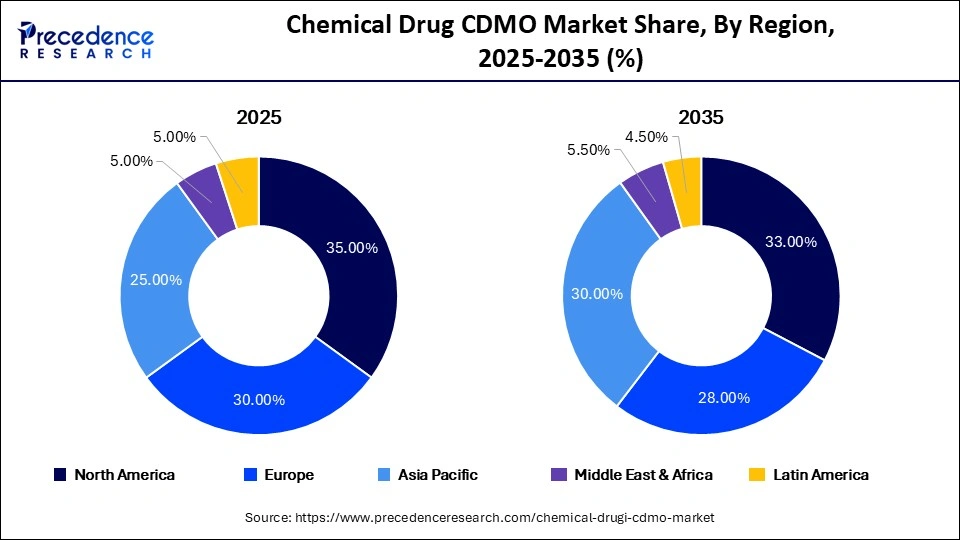
Europe: The Second-Largest Market
Europe is the second-largest market for chemical drug CDMO, holding a 30% share in 2025, and is estimated to grow at a strong CAGR of 7.5% over the projected period, driven by its mature and well-regulated pharmaceutical ecosystem. The region has strong capabilities in APIs, sterile products, and complex formulations, particularly across Germany, Switzerland, Italy, France, Ireland, and the United Kingdom. In 2025, the European Medicines Agency continued to approve biosimilars and specialty medicines, further increasing demand for outsourced manufacturing services across the region.
Germany Drug CDMO Market Analysis
Germany is a major contributor to the European market, supported by its advanced chemical industry and strong pharmaceutical manufacturing base. German CDMOs are widely preferred for high-potency APIs, precision chemistry, and regulated-market supply chains. The country's strong industrial infrastructure and manufacturing reliability continue to strengthen its position in the European outsourcing ecosystem.
Asia Pacific: The Fastest-Growing Region
Asia Pacific held a 20% share of the market in 2025 and is expected to grow at the fastest CAGR of 12.5% between 2026 and 2035. This growth is driven by expanding manufacturing capacity, rising healthcare demand, and cost-competitive production capabilities in the region. Global pharmaceutical companies are increasingly shifting sourcing to India, China, and Southeast Asia to improve supply chain resilience and optimize costs. At the same time, local pharmaceutical companies are also expanding partnerships for outsourced development and commercial manufacturing.
India Chemical Drug CDMO Market Analysis
In India, the market is driven by its cost-efficient manufacturing base and strong position in the global pharmaceutical supply chain. Indian CDMOs are increasingly providing APIs, oral solids, injectables, and development services to both innovator and generic companies. Growth is further supported by government production incentives and rising domestic healthcare demand.
Chemical Drug CDMO Market Companies
- Almac Group
- BASF SE
- Boehringer Ingelheim
- Cipla Limited
- DSM Sinochem Pharmaceuticals
- Fujifilm Diosynth Biotechnologies
- Hovione
- Lonza Group
- Mylan (Viatris Inc.)
- Patheon (Thermo Fisher Scientific Inc.)
- Recipharm AB
- Samsung Biologics
- Siegfried Holding AG
- Teva Pharmaceuticals Industries Ltd.
- WuXi AppTec
Recent Developments
- In April 2026, Mikart, LLC, a provider of formulation development, analytical testing, and finished dose manufacturing services, announced a strategic co-marketing alliance with Benuvia Operations, LLC. Benuvia contributes specialized expertise in controlled substances and complex API development, while the partnership aims to broaden integrated CDMO solutions for pharmaceutical clients.(Source: https://www.prnewswire.com )
- In April 2026, HRV Pharma entered a strategic partnership with Shodhana Laboratories to accelerate next-generation active pharmaceutical ingredient development. The collaboration combines HRV Pharma's AI-enabled virtual pharmaceutical platform with Shodhana Laboratories' expertise in precision chemistry and GMP-compliant manufacturing operations.(Source: https://www.hrvpharma.com )
Segments Covered in the Report
By Service Type
- API Manufacturing (Small Molecule APIs, Large Molecule APIs)
- Formulation Development
- Contract Research & Development (R&D)
- Regulatory Services
- Packaging Services
By Drug Type
- Branded Drugs
- Generic Drugs
- Biosimilars
- Over-the-Counter (OTC) Drugs
By Therapeutic Area
- Oncology
- Cardiovascular
- Neurology
- Infectious Diseases
- Endocrinology
- Immunology
- Others
By API Type
- Small Molecule APIs
- Large Molecule APIs (Biologics, Biosimilars)
- Peptide-based APIs
- Monoclonal Antibodies (mAbs)
By Outsourcing Mode
- Full-service CDMOs (End-to-End Services from Discovery to Commercialization)
- Dedicated Capacity CDMOs (Single-client Facility)
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
