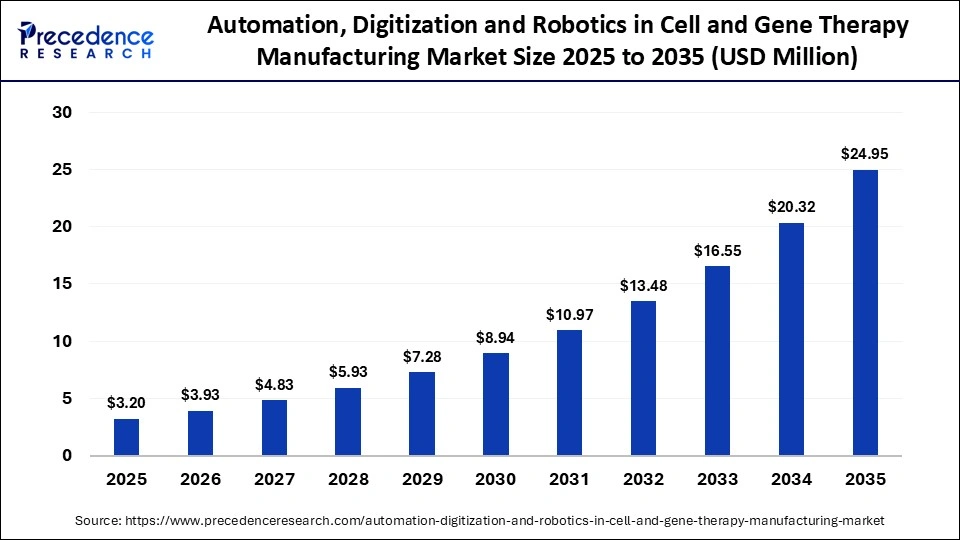
What is the Automation, Digitization and Robotics in Cell and Gene Therapy Manufacturing Market Size in 2026?
The global automation, digitization and robotics in cell and gene therapy manufacturing market size accounted for USD 950.00 million in 2025 and is predicted to increase from USD 1,127.65 million in 2026 to approximately USD 5,275.10 million by 2035, expanding at a CAGR of 18.70% from 2026 to 2035. The market is driven by the rising adoption of cutting-edge technologies by both large and small manufacturers to improve the precision of complex therapies.
Key Takeaway
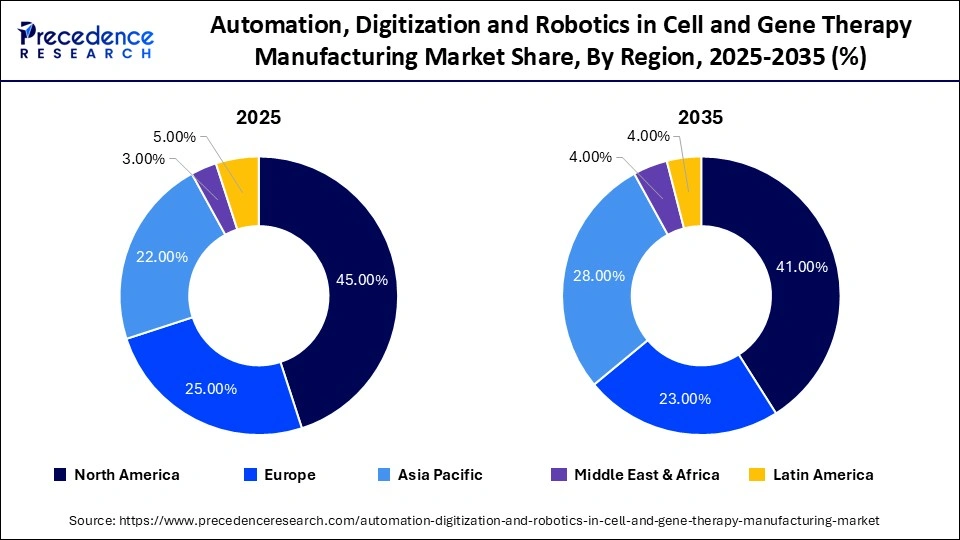
- North America led the automation, digitization and robotics in cell and gene therapy manufacturing market with a 45% share in 2025.
- The Asia Pacific is expected to grow at the fastest CAGR of 22.5% between 2026 and 2035.
- By technology type, the automated bioprocessing systems segment led the market with a 30% share in 2025.
- By technology type, the robotic cell handling & processing systems segment is observed to grow at the fastest CAGR of 20.5% in the upcoming period.
- By automation level, the semi-automated systems segment led the global market in 2025 with a 55% share.
- By automation level, the fully automated manufacturing platforms segment is expected to grow at the fastest CAGR of 22.5% during the projection period.
- By application, the cell therapy manufacturing segment led the global market in 2025 with a 35% share.
- By application, the gene therapy manufacturing segment is expected to grow at a CAGR of 20% in the coming years.
- By end-use, the biopharmaceutical companies segment led the global market in 2025 while holding a 50% share.
- By end-use, the contract development and manufacturing organizations (CDMOs) segment is expected to grow at the fastest CAGR of 20.5% between 2026 and 2035.
Market Overview
The market refers to technologies and solutions used to streamline and scale the production of cell and gene therapies by integrating automated systems, digital platforms, and robotic processes. Automation has the potential to improve process efficiency, reduce key cost drivers, and meet regulatory requirements. Digitization minimizes human error, enhances regulatory compliance, and facilitates scaling as therapies progress from clinical trials to commercial launch. Furthermore, robotic solutions assist in the scaling up and scaling out of cell therapies, which are often developed using traditional manual methods in academic laboratories and biotechnology firms. Emerging methodologies are simplifying the process for teams to interchange equipment as needed, accommodating necessary changes more seamlessly.
Impact of Artificial Intelligence on the Automation, Digitization and Robotics in Cell and Gene Therapy Manufacturing Market
The integration of artificial intelligence in cell and gene therapy manufacturing signifies a significant transformation in the field. It enhances automation, digitization, and robotics in the production of these therapies, enabling a shift from specialized, customized solutions to scalable, accessible, and highly personalized medical options. AI excels at identifying subtle patterns, predicting outcomes from complex datasets, and optimizing intricate multivariable systems, thereby unlocking the full potential of cell and gene therapies.
By incorporating AI-enabled biosensors throughout the production process, manufacturers can generate substantial volumes of real-time data that improve automation and reinforce digital control. Through strategic data collection and advanced analytics, AI is driving more efficient, consistent, and reproducible manufacturing processes across the entire cell and gene therapy value chain.
Major Market Trends
- Development of Novel Technologies: Emerging startups are at the forefront of developing innovative technologies, while established biopharmaceutical companies are enhancing their portfolios by implementing advanced automated and digital production systems.
- Rise of In Vivo Therapy Manufacturing Technology: Recent advancements in vector engineering and precise targeting are driving a significant transition toward in vivo therapy approaches. As a result, scalable, high-precision, robotics-enabled manufacturing is becoming increasingly important within the industry.
- Rising Demand for Cell and Gene Therapies: The rapid growth of advanced therapies such as CAR-T and gene editing treatments is driving the need for scalable and precise manufacturing solutions. Automation and robotics help manage the complexity of producing patient-specific therapies while maintaining consistency and quality.
- Increasing Regulatory Compliance and Quality Requirements: Cell and gene therapies require strict adherence to regulatory standards for safety, traceability, and quality control. Digital systems and automated workflows help ensure accurate documentation, real-time monitoring, and compliance with global regulatory guidelines.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 950.00 Million |
| Market Size in 2026 | USD 1,127.65 Million |
| Market Size by 2035 | USD 5,275.10 Million |
| Market Growth Rate from 2026 to 2035 | CAGR of 8.20% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Technology Type, Automation Level, Application, End-Use, Deployment Type, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Rising Adoption of Advanced Manufacturing Solutions
Manufacturers are progressively embracing automation, digitization, and robotics to optimize intricate manual processes, boost efficiency, and increase flexibility and scalability, which is a major factor driving the market. These technologies minimize human error and foster innovation, encouraging major healthcare and biopharmaceutical manufacturers to make substantial investments in advanced manufacturing solutions.
Restraint
Complexity in Process Integration
The development processes for cell and gene therapy are continually evolving. Ongoing adjustments to these processes, along with the incorporation of new software and equipment, enhance operational complexity and present challenges in both research and manufacturing settings. These challenges restrain the growth of the automation, digitization and robotics in cell and gene therapy manufacturing market.
Opportunity
Commercialization of Advanced Therapies
Advanced therapy medicinal products (ATMPs), especially genetically modified cell-based therapies, are increasingly obtaining significant regulatory approvals. The integration of automation, digitization, and robotics presents opportunities to improve processes from preclinical studies to commercial manufacturing, thereby fostering innovation and expediting the commercialization of advanced therapies.
Segment Insights
Technology Insights
Automation, Digitization and Robotics in Cell and Gene Therapy Manufacturing Market Share, By Technology Type, 2025-2035 (%)
| Technology Type | 2025 | 2035 | CAGR (%) |
| Automated Bioprocessing Systems | 30.00% | 32.00% | 19.5% |
| Robotic Cell Handling & Processing Systems | 20.00% | 21.50% | 20.5% |
| Digital Twin & Simulation Platforms | 15.00% | 14.50% | 18.0% |
| AI-driven Manufacturing Execution Systems (MES) | 20.00% | 19.00% | 19.0% |
| Automated Quality Control & Analytics Systems | 15.00% | 13.00% | 17.5% |
Automated Bioprocessing Systems Segment Dominated the Market With a 30% Share in 2025
The automated bioprocessing systems (closed & modular systems) segment dominated the automation, digitization and robotics in cell and gene therapy manufacturing market with nearly 30% share in 2025. This is due to the increasing demand for scalable, closed-system manufacturing, which mitigates contamination risks and ensures reproducibility in cell and gene therapies. Automation systems facilitate real-time monitoring and control of the bioprocessing workflow, enabling precise management of process parameters. The implementation of automation in bioprocessing diminishes variability by maintaining consistent process conditions, minimizing the impact of unit operation errors, and reducing batch-to-batch variability.
The robotic cell handling & processing systems segment accounted for approximately 20% market share in 2025 and is projected to grow at the fastest CAGR of 20.5% in the coming years. This growth is fueled by the swift adoption of robotics for the precise and sterile handling of cells, which enhances consistency and minimizes manual intervention in intricate workflows, ultimately improving manufacturing efficiency and precision. Robotic cells consist of components utilized by one or multiple robots to carry out tasks on a production or assembly line. These systems operate as standalone units or as integrated elements within a larger production line.
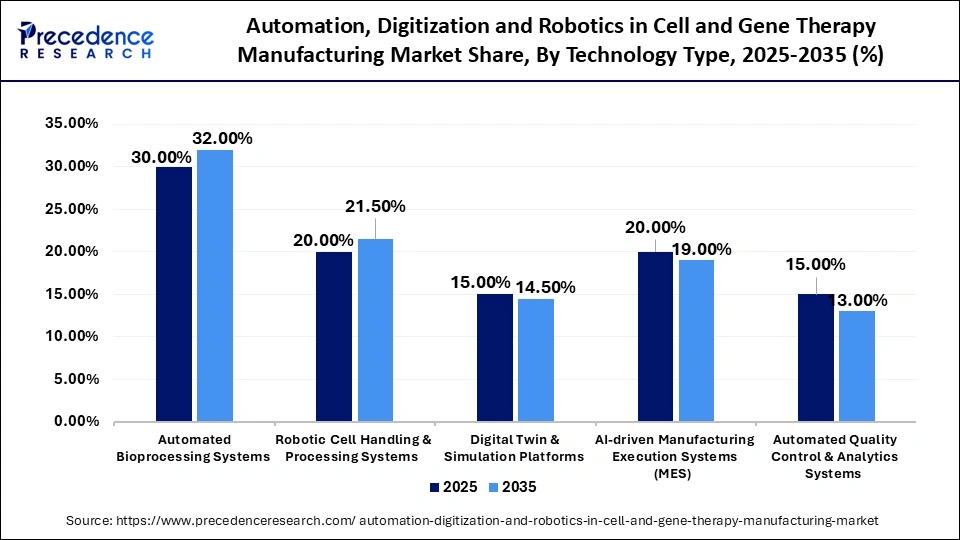
The digital twin & simulation platforms segment held approximately 15% market share in 2025 and is projected to grow at a CAGR of 18% in the coming years, due to the capability of digital twins to facilitate process optimization, predictive modelling, and accelerated scale-up, ultimately reducing the time required to bring therapies to market. These platforms enable dynamic simulations and enhance decision-making throughout the manufacturing lifecycle. Additionally, digital twins can utilize each patient's unique clinical, biological, imaging, and genomic data to create a computational representation of the patient and their specific disease. These advancements are significantly promoting the adoption of digital twin and simulation technologies in the manufacturing of cell and gene therapies.
The AI-driven manufacturing execution systems (MEA) segment held approximately 20% market share in 2025 and is anticipated to grow at a CAGR of 19% in the coming years. This growth is fueled by the rising adoption of AI-driven MES solutions that enhance workflow orchestration, data integration, and compliance tracking throughout manufacturing processes. Furthermore, AI MES and its data analytics capabilities can expand access to cell and gene therapies for more patients and broaden the range of diseases that can be addressed, thereby supporting market adoption.
Automation Level Insights
Automation, Digitization and Robotics in Cell and Gene Therapy Manufacturing Market Share, By Automation Level, 2025-2035 (%)
| Automation Level | 2025 | 2035 | CAGR (%) |
| Semi-automated Systems | 55.00% | 48.00% | 15.5% |
| Fully Automated Manufacturing Platforms | 45.00% | 52.00% | 22.5% |
Semi-Automated Systems Segment Dominated the Market in 2025 With a 55% Share
The semi-automated systems segment dominated the automation, digitization and robotics in cell and gene therapy manufacturing market by holding a 55% share in 2025. This is mainly due to their lower costs and the gradual shift from manual processes in various facilities, which allows for enhanced flexibility in manufacturing. Additionally, semi-automated systems are highly effective in reducing labor costs while maintaining a strong level of quality control, thus facilitating their market adoption.
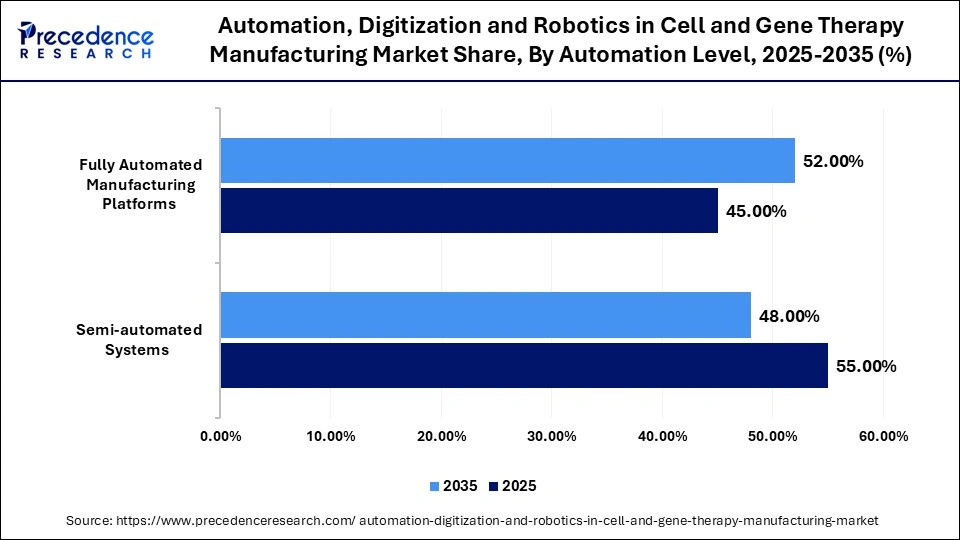
The fully automated manufacturing platforms segment held nearly 450% share of the market in 2025 and is anticipated to expand at the fastest CAGR of 22.5% during the forecast period. The growth of this segment is attributed to manufacturers' preference for fully automated systems, as they aim to scale production, enhance consistency, and comply with stringent regulatory standards. These platforms provide notable benefits in terms of speed and operational efficiency, while also minimizing variability in the products manufactured. Consequently, fully automated manufacturing platforms are drawing significant interest and investment.
Application Insights
The Cell Therapy Manufacturing Segment Led the Market With a 35% Share in 2025
The cell therapy manufacturing segment led the automation, digitization and robotics in cell and gene therapy manufacturing market with about 35% share in 2025. This is because of the increasing adoption of CAR-T and stem cell therapies, which necessitate highly controlled and automated manufacturing processes. Cell therapies play a key role in treating a variety of serious conditions, including blood cancers, autoimmune disorders, and rare diseases. The manufacturing of cell therapy involves the development, production, and delivery of viable therapeutic cells, which involves several stages, including cell selection, genetic modification, expansion, and stringent quality control. This significantly boosts the adoption of automation, digitization, and robotics tools.
The gene therapy manufacturing segment held approximately 30% market share in 2025 and is expected to expand at the fastest CAGR of around 20% throughout the forecast period, owing to the rapid advancement of gene therapy, bolstered by an expanding pipeline of viral vector-based therapies and an increasing number of regulatory approvals. Viruses have naturally developed the ability to transfer genetic material into host cells; gene therapy harnesses this capability. Nevertheless, the limitations associated with viral vectors have heightened interest in non-viral delivery systems. These include lipid nanoparticles (LNPs) and polymer- or peptide-based carriers. Collectively, these innovations are significantly improving the market penetration of gene therapy manufacturing technologies.
Automation, Digitization and Robotics in Cell and Gene Therapy Manufacturing Market Share, By Application, 2025-2035 (%)
| Application | 2025 | 2035 | CAGR (%) |
| Cell Therapy Manufacturing | 35.00% | 36.50% | 19.5% |
| Gene Therapy Manufacturing | 30.00% | 31.50% | 20.0% |
| Process Development & Scale-up | 15.00% | 14.00% | 17.0% |
| Quality Control & Release Testing | 10.00% | 9.00% | 16.5% |
| Cold Chain & Logistics Automation | 10.00% | 9.00% | 18.5% |
The process development & scale-up segment held approximately 15% market share in 2025 and is expected to expand at a CAGR of 17.0% in the coming years. This segment's growth is driven by increasing use of automation tools that enhance process optimization and facilitate the transition from laboratory settings to commercial production. These activities adhere to current Good Manufacturing Practice (cGMP) regulations and incorporate advanced bioprocessing technologies alongside high-throughput analytical methods. It ensures the creation of scalable and robust processes from the outset, allowing for smooth transitions throughout the program, from early-stage clinical development to full commercialization.
The quality control & release testing segment held nearly 10% share of the market in 2025 and is expected to grow at a CAGR of 16.5% in the upcoming period, driven by the importance of QC automation in ensuring product safety, despite its smaller share compared to core manufacturing processes. The QC framework in gene therapy encompasses all stages of the therapeutic lifecycle, from vector development to post-treatment monitoring, highlighting the significance of rigorous testing, validation, and standardization.
End-Use Insights
Biopharmaceutical Companies Held a 50% Market Share in 2025
The biopharmaceutical companies segment dominated the automation, digitization and robotics in cell and gene therapy manufacturing market with approximately 50% share in 2025, driven by substantial investments in advanced therapy manufacturing and the need for scalable production platforms. These organizations benefit from robust infrastructure, dedicated specialist teams to manage potential complications, and highly controlled, well-equipped environments that support commercialization. In addition, biopharmaceutical companies are strongly connected with key stakeholders across the value chain, further reinforcing their leading market position.
Automation, Digitization and Robotics in Cell and Gene Therapy Manufacturing Market Share, By End-Use, 2025-2035 (%)
| End-Use | 2025 | 2035 | CAGR (%) |
| Biopharmaceutical Companies | 50.00% | 48.00% | 17.5% |
| Contract Development and Manufacturing Organizations (CDMOs) | 35.00% | 38.00% | 20.5% |
| Research Institutes & Academic Centers | 15.00% | 14.00% | 16.0% |
The contract development and manufacturing organizations (CDMOs) segment held a 35% share of the market in 2025 and is projected to expand at the fastest CAGR of 20.5% over the forecast period. This is mainly due to the rising global trend of outsourcing complex biologics manufacturing. CDMOs facilitate the expedited and efficient introduction of new pharmaceutical products to the market by offering specialized expertise, adaptable scalability, and significant cost savings.
The research institutes & academic centers segment held nearly 15% market share in 2025 and is projected to grow at a CAGR of 16% in the coming years, driven by their critical role in early-stage development and innovation in automation technologies for therapy manufacturing. These institutions are primarily involved in preclinical research and are supported by dedicated teams and robust resource networks. They operate well-equipped platforms to analyze multiple dimensions of research before therapies advance to clinical and commercial phases.
Regional Insights
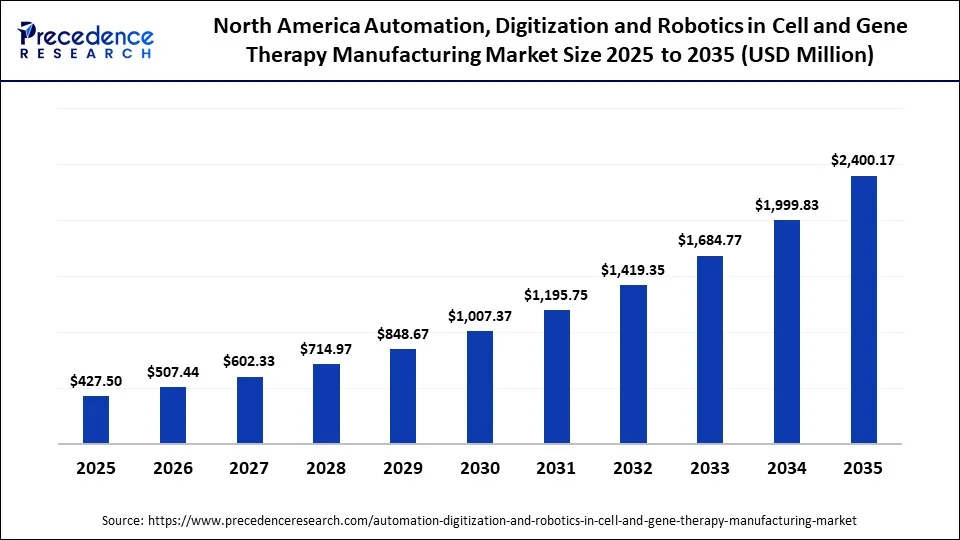
North America Automation, Digitization and Robotics in Cell and Gene Therapy Manufacturing Market Size and Growth 2026 to 2035
The North America automation, digitization and robotics in cell and gene therapy manufacturing market size is estimated at USD 427.50 million in 2025 and is projected to reach approximately USD 2,400.17 million by 2035, with a 18.83% CAGR from 2026 to 2035.
North America Dominated the Market With a 45% Share In 2025
North America dominated the automation, digitization and robotics in cell and gene therapy manufacturing market with approximately 45% share in 2025. The region's dominance is supported by a robust presence of biotechnology companies, advanced manufacturing infrastructure, and strong regulatory support for cell and gene therapies. The region's leadership is further strengthened by the involvement of key companies and stakeholders, along with a significant number of contract development and manufacturing organizations (CDMOs).
U.S. Automation, Digitization and Robotics in Cell and Gene Therapy Manufacturing Market Size and Growth 2026 to 2035
The U.S. automation, digitization and robotics in cell and gene therapy manufacturing market size is calculated at USD 320.63 million in 2025 and is expected to reach nearly USD 1,812.13 million in 2035, accelerating at a strong CAGR of 18.91% between 2026 and 2035.
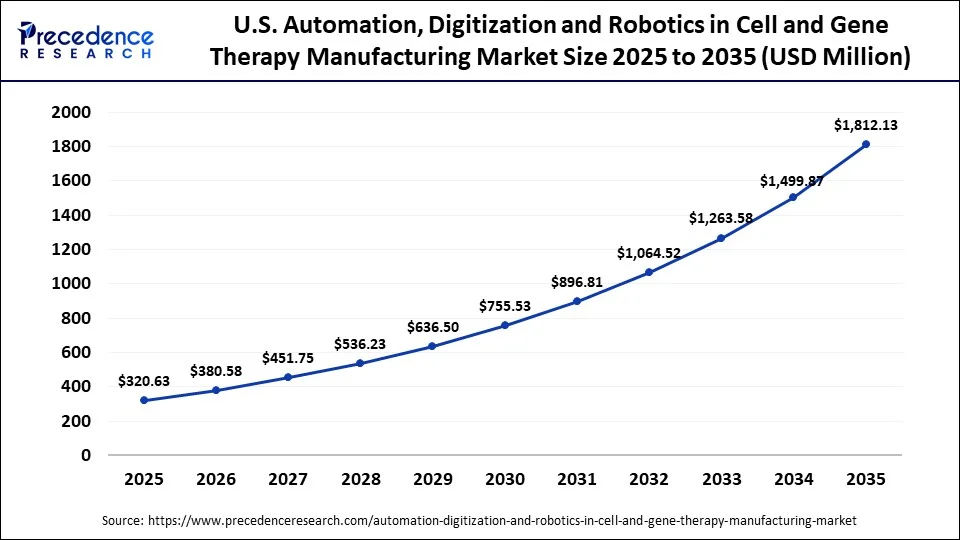
The U.S. leads the North American market, driven by the swift adoption of advanced healthcare technologies and the oversight of the US FDA and other regulatory bodies. Additionally, US CDMOs are enhancing their production capabilities for viral vectors, autologous and allogeneic cell processing, and cryogenic logistics, thereby supporting market growth.
Europe: The Second-Largest Market
Europe held approximately 25% share of the automation, digitization and robotics in cell and gene therapy manufacturing market in 2025 and is projected to grow at a CAGR of 17.0% in the coming years. This growth is supported by rising investments in advanced therapy medicinal products (ATMPs) and automation technologies. The swift implementation of cell and gene therapies in Europe is fortified by strong collaborations with the private sector and substantial government funding, along with the presence of the European Medicines Agency (EMA).
Germany is leading the charge in Europe, thanks to significant investments from both public and private sectors that have accelerated the development of the country's cell and gene therapy market. The country is actively engaged in the development and testing of innovative cell and gene therapy medicines, offering considerable benefits to patients.
How is the Opportunistic Rise of Asia Pacific in the Market?
Asia Pacific held a 22% share of the automation, digitization and robotics in cell and gene therapy manufacturing market in 2025 and is anticipated to grow at a CAGR of 22.5% during the forecast period. This growth is fueled by the expansion of biopharmaceutical manufacturing capabilities and strong government initiatives supporting biotech innovation, including the establishment of accelerated approval pathways, rising healthcare demands, and increasing investments from both private and public sectors. The rapid expansion of the healthcare and pharmaceutical sectors also contributes to market growth.
China is a major player in the Asia Pacific market, driven by a high prevalence of chronic diseases that heighten the demand for advanced and novel cell and gene therapies, along with significant government backing for infrastructure development. Regulatory support and intensive research and development efforts also drive the market in the country.
Automation, Digitization and Robotics in Cell and Gene Therapy Manufacturing Market Companies
- Thermo Fisher Scientific Inc.
- Sartorius AG
- Danaher Corporation (Cytiva)
- Lonza Group AG
- Merck KGaA
- Fujifilm Diosynth Biotechnologies
- GE HealthCare Technologies Inc.
- Siemens AG
- ABB Ltd.
- Universal Robots A/S
- FANUC Corporation
- KUKA AG
- Rockwell Automation, Inc.
- Emerson Electric Co.
- Eppendorf SE
Recent Developments
- In January 2026, the US Food and Drug Administration (FDA) introduced greater regulatory flexibility for cell and gene therapy (CGT) manufacturers, easing certain quality requirements to accelerate patient access. Clinical and commercial quality controls have been relaxed in specific areas as the FDA seeks to expedite therapy approvals for conditions with high unmet medical need.(Source: https://www.clinicaltrialsarena.com)
- In July 2025, a new UK consortium secured a $1.34 million grant to launch an automated cell therapy manufacturing platform. Cellular Origins, CGT Catapult, and Resolution Therapeutics have partnered to design a fully automated, scalable CGT manufacturing solution aimed at accelerating patient access to cell therapies.(Source: https://www.pharmaceuticalcommerce.com)
Segments Covered in the Report
By Technology Type
- Automated Bioprocessing Systems (Closed & Modular Systems)
- Robotic Cell Handling & Processing Systems
- Digital Twin & Simulation Platforms
- AI-driven Manufacturing Execution Systems (MES)
- Automated Quality Control & Analytics Systems
By Automation Level
- Semi-automated Systems
- Fully Automated Manufacturing Platforms
By Application
- Cell Therapy Manufacturing (CAR-T, Stem Cell Therapy)
- Gene Therapy Manufacturing (Viral Vectors, Non-viral Systems)
- Process Development & Scale-up
- Quality Control & Release Testing
- Cold Chain & Logistics Automation
By End-Use
- Biopharmaceutical Companies
- Contract Development and Manufacturing Organizations (CDMOs)
- Research Institutes & Academic Centers
By Deployment Type
- On-premise Automation Systems
- Cloud-based Digital Manufacturing Platforms
- Hybrid Digital-Physical Systems
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
