What is the Chemical API CDMO Market Size in 2026?
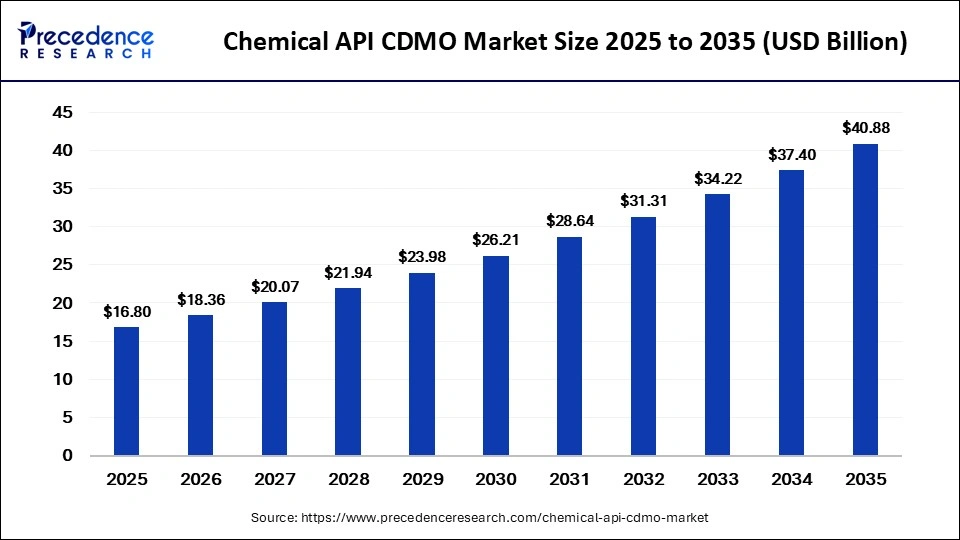
The global chemical API CDMO market size accounted for USD 16.80 billion in 2025 and is predicted to increase from USD 18.36 billion in 2026 to approximately USD 40.88 billion by 2035, expanding at a CAGR of 9.30% from 2026 to 2035. The chemical API CDMO market is driven by increasing outsourcing by pharmaceutical companies, rising complexity of drug molecules, and demand for cost-efficient and scalable manufacturing.
Key Takeaways
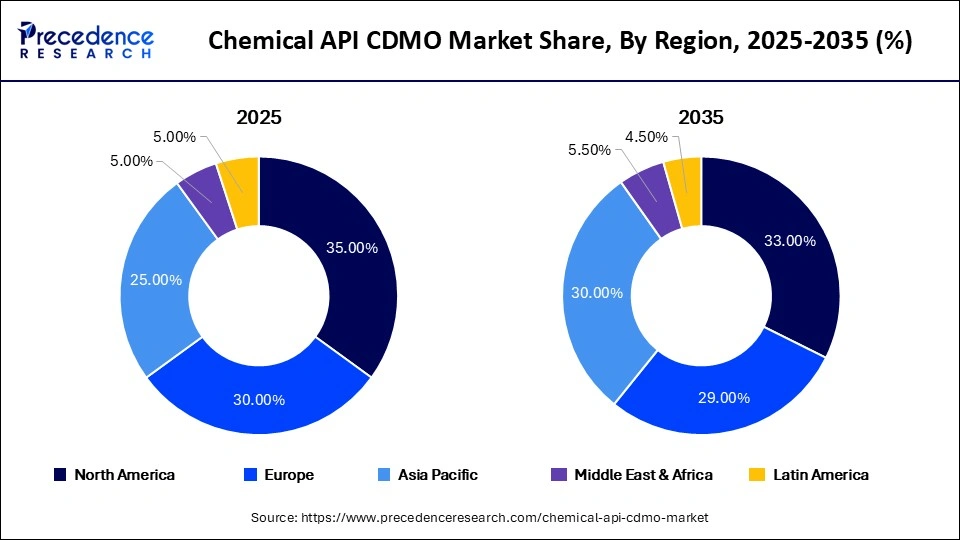
- North America dominated the global chemical API CDMO market with a share of 35% in 2025.
- Asia Pacific is expected to grow at the fastest CAGR of 14.5% during the forecast period.
- By service type, the API manufacturing segment held a dominant position in the market, accounting for 50% of the market share in 2025.
- By service type, the formulation development segment is the second-largest shareholder and is expected to grow at a 10.5% CAGR between 2026 and 2035.
- By drug type, the branded drugs segment held a 40% market share in 2025.
- By drug type, the generic drugs segment is the second-largest shareholder and is expected to grow at a 9.5% CAGR between 2026 and 2035.
- By API type, the small molecule APIs segment held a major revenue share of 55% in the market in 2025.
- By API type, the large molecule APIs segment is the second-largest shareholder and is expected to grow at a 12.5% CAGR between 2026 and 2035.
- By outsourcing model, the full-service CDMOs segment led the global market with a share of 60% in 2025.
- By outsourcing model, the dedicated capacity CDMOs segment is the second-largest shareholder and is expected to grow at a 10% CAGR between 2026 and 2035.
Market Overview
The chemical API CDMO market comprises specialized service providers that help pharmaceutical companies develop, scale up, and commercially produce APIs. This market is experiencing stable growth, driven by the complexity of drug molecules, the need for cost-efficient manufacturing processes, and the rising reliance on outsourcing among pharma and biotech companies. Further advances in technologies such as continuous production, green chemistry, and electronic process monitoring are accelerating market evolution. The growth of biologics and high-potency APIs is also increasing demand for specialized CDMO capabilities, making the market a strategic partner for innovation, speed, and scalability in the pharmaceutical industry.
How is AI Impacting the Chemical API CDMO Market?
The chemical API CDMO market is transforming through artificial intelligence to improve efficiency, accuracy, and decision-making in the entire drug development and manufacturing lifecycle. AI-based predictive analytics is being introduced to optimize the route to chemical synthesis, minimize trial-and-error in process development, and shorten the API discovery timeline. The AI-based process control systems can be implemented in the manufacturing industry to allow real-time tracking of the process to maintain the quality of the goods and adherence to the strict rules and regulations.
Chemical API CDMO Market Trends
- The outsourcing of API development and manufacturing to CDMOs is increasing, driven by the need for cost efficiency and operational flexibility. This trend significantly boosts the demand for external manufacturing services.
- The growing need for highly specific therapies, especially in oncology, is propelling the demand for high-potency APIs, requiring CDMOs to invest in special containment systems and specialized safety equipment to handle complex compounds.
- CDMOs are increasingly adopting continuous manufacturing to enhance efficiency, reduce waste, and ensure consistent product quality, enabling faster and more scalable production compared to traditional batch processes.
- The use of digital tools, data analytics , and automation is on the rise among CDMOs, simplifying processes, real-time monitoring, and predictive maintenance , which subsequently leads to more operational efficiency and regulatory compliance.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 16.80 Billion |
| Market Size in 2026 | USD 18.36 Billion |
| Market Size by 2035 | USD 40.88 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 9.30% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Service Type, Drug Type, Therapeutic Area, API Type, Outsourcing Model, Scale of Manufacturing, Manufacturing Technology, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Drivers
Rising Outsourcing Demand
The growing trend toward outsourcing by pharmaceutical and biotechnology companies in search of cost efficiency, accelerated development time, and access to advanced technical skills is a major driver of the chemical API CDMO market. The need to manufacture complex and high-potency APIs, in particular, to treat cancer and chronic diseases, is further driving the shift toward specialized CDMOs. Also, the development of biologics and specialty drugs is increasing the necessity of scalable and flexible manufacturing.
Restraint
Operational and Regulatory Challenges
High capital investment requirements for advanced manufacturing facilities, particularly for high-potency APIs, along with stringent and evolving regulatory standards, create significant barriers to entry in the chemical API CDMO market. Moreover, concerns over intellectual property protection further limit the outsourcing of sensitive molecule development.
Opportunity
Expanding Innovation and Emerging Market Potential
The chemical API CDMO market has strong growth potential due to the rising demand for complex, niche, and high-value pharmaceutical products, along with the expansion of personalized and targeted therapies that require small-batch, high-potency API production. Additionally, increasing focus on supply chain diversification and the reshoring of API manufacturing in developed countries is boosting investments in new facilities.
Segment Insights
Service Type Insights
The API Manufacturing Segment Held a 50% Market Share in 2025
The API manufacturing segment led the chemical API CDMO market by holding the largest share of 50% in 2025, driven by the growing focus on cost-efficient, high-volume production of generics, biologics, and biosimilars . Outsourced manufacturing enables pharmaceutical and biotech companies to access scalable and efficient production capabilities, reducing operational costs and time to market while encouraging capacity expansion and technological upgrades at API facilities.
The formulation development segment held a 15% market share in 2025 and is expected to grow at a CAGR of 10.5% during the projection period, driven by increasing demand for complex drug formulations , biologics, and personalized medicine . The need for improved bioavailability and targeted therapies is driving demand for specialized formulation expertise. Outsourcing of formulation development helps the company reduce the burden of research and development within its own company.
Chemical API CDMO Market Share, By Service Type, 2025-2035 (%)
| Service Type | 2025 | 2035 | CAGR (%) |
| API Manufacturing | 50.00% | 47.00% | 9.50% |
| Formulation Development | 15.00% | 16.00% | 10.50% |
| Contract R&D | 15.00% | 16.00% | 11.00% |
| Regulatory Services | 10.00% | 10.50% | 8.50% |
| Packaging Services | 10.00% | 10.50% | 7.50% |
The contract R&D segment held a 15% share of the market in 2025 and is expected to grow at the fastest CAGR of 11% during the projection period, rising investment in novel drug discovery , precision medicine, and biologics. R&D services are critical for the development of new chemical APIs, particularly in precision medicine and biologics. These services also play a significant role in the early-stage development of drugs that require custom research solutions to help in designing, screening, and optimizing molecules. The increased demand to develop certain treatments and biologics has increased the requirement to subcontract R&D services with advanced scientific expertise and skills.
The regulatory services segment held a 10% share of the market in 2025 and is expected to grow at a CAGR of 8.5% during the projection period, by increasingly complex global regulatory requirements and stringent drug approval processes. These services assist pharmaceutical and biotech companies in ensuring compliance, managing clinical trial approvals, and navigating evolving regulatory frameworks.
Drug Type Insights
Chemical API CDMO Market Share, By Drug Type, 2025-2035 (%)
| Drug Type | 2025 | 2035 | CAGR (%) |
| Branded Drugs | 40.00% | 35.00% | 7.00% |
| Generic Drugs | 35.00% | 37.00% | 9.50% |
| Biosimilars | 15.00% | 18.00% | 14.00% |
| OTC Drugs | 10.00% | 10.00% | 6.50% |
Why Did the Branded Drugs Segment Dominate the Market in 2025?
The branded drugs segment dominated the chemical API CMO market with a 40% share in 2025, driven by strong demand for novel and high-value pharmaceutical products. Branded drugs continue to lead due to patent protection, high product differentiation, and continuous innovation by major pharmaceutical companies. These drugs often target multiple disease areas and require advanced API manufacturing and strict regulatory compliance, increasing reliance on CDMO partnerships.
The generic drugs segment held a 35% share of the market in 2025 and is expected to grow at a CAGR of 9.5% during the projection period, driven by a growing number of patent expirations and increasing demand for affordable treatment options. Generics are increasing at an accelerated rate due to the fact that healthcare systems and consumers are seeking alternatives to expensive branded medicines, which are cheaper.
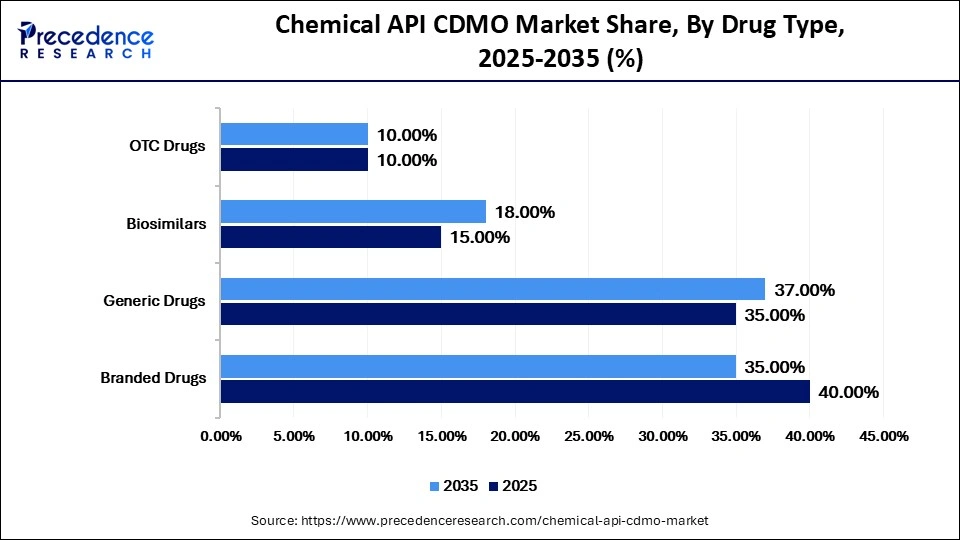
The biosimilars segment held a 15% share of the market in 2025 and is expected to grow at the fastest CAGR of 14% during the projection period, driven by increasing demand for affordable biologic therapies and growing regulatory approvals. Biosimilars are gaining traction as cost-effective alternatives to originator biologics, particularly in oncology , autoimmune disorders, and chronic diseases. Due to their complex development and manufacturing requirements, companies increasingly rely on CDMOs for biosimilar API production.
The OTC drugs segment held a 10% market share in 2025 and is expected to grow at a CAGR of 6.5% during the projection period, due to the increasing self-medication trend and greater consumer access to non-prescription medicines. Common applications include pain relief, allergy management, and gastrointestinal treatments, sustaining steady demand for API manufacturing services.
API Type Insights
The Small Molecule APIs Segment Held a 55% Share of the Market in 2025
The small molecule APIs segment dominated the chemical API CDMO market with the highest share of 55% in 2025. This is mainly due to their well-established manufacturing processes and cost-effective, scalable chemical synthesis routes. Small molecules remain central to drug production due to their widespread use across therapeutic areas such as cardiovascular, metabolic, and infectious diseases. Their standardized production requirements also make them highly suitable for large-scale outsourcing, enabling pharmaceutical companies to focus on efficiency, compliance, and cost optimization.
The large molecule APIs (biologics, biosimilars) segment held a 25% share of the market in 2025 and is expected to grow at a CAGR of 12.5% during the projection period, driven by the rapid increase in the development of biologics and biosimilars. The complexity of production, stringent regulatory requirements, and need for advanced bioprocessing capabilities are increasing reliance on CDMO partnerships. Rising demand for targeted and precision therapies is further accelerating investment in biologic drug development.
Chemical API CDMO Market Share, By API Type, 2025-2035 (%)
| API Type | 2025 | 2035 | CAGR (%) |
| Small Molecule APIs | 55.00% | 52.00% | 8.00% |
| Large Molecule APIs (Biologics, Biosimilars) | 25.00% | 28.00% | 12.50% |
| Peptide-based APIs | 10.00% | 12.00% | 14.00% |
| Monoclonal Antibodies (mAbs) | 10.00% | 8.00% | 13.00% |
The peptide-based APIs segment held a 10% share of the market in 2025 and is expected to grow at a CAGR of 14% during the projection period, driven by their increased use in targeted drug discovery and more advanced therapeutic applications. Due to selectivity and lower toxicity compared with conventional molecules, peptides are becoming more significant as medicine to address cancer, metabolic disorders, and rare diseases.
The monoclonal antibodies segment held a 10% share of the market in 2025 and is expected to grow at a CAGR of 13% during the projection period, driven by rising demand for precision medicine and targeted therapy in immunology and oncology. The advent of mAbs as one of the most rapidly evolving biologic categories is due to their agility and efficacy, as well as their ability to target disease pathways. The growth in the biologics pipeline and the rise in chronic disease rates are boosting the demand for the outsourcing of monoclonal antibody development.
Outsourcing Model Insights
Full-service CDMOs Held a 60% Share of the Chemical API CDMO Market in 2025
The full-service CDMOs segment dominated the global market with the highest share of 60% in 2025, driven by rising demand for end-to-end pharmaceutical development solutions. Full-service CDMOs offer integrated services across drug discovery, development, manufacturing, and commercialization, enabling pharmaceutical companies to streamline operations and reduce time to market. Their ability to manage complex projects across the entire product lifecycle makes them highly attractive to both large pharmaceutical companies and biotech firms.
Chemical API CDMO Market Share, By Outsourcing Model, 2025-2035 (%)
| Outsourcing Model | 2025 | 2035 | CAGR (%) |
| Full-service CDMOs | 60.00% | 58.00% | 8.00% |
| Dedicated Capacity CDMOs | 40.00% | 42.00% | 10.00% |
The dedicated capacity CDMOs segment held a 40% share of the market in 2025 and is expected to grow at the fastest CAGR of 10% during the projection period, driven by increasing concerns among large pharmaceutical companies regarding data security, process control, and intellectual property protection. These CDMOs operate customized manufacturing facilities tailored to specific client requirements, offering greater control over quality, supply chain stability, and confidentiality.
Regional Insights
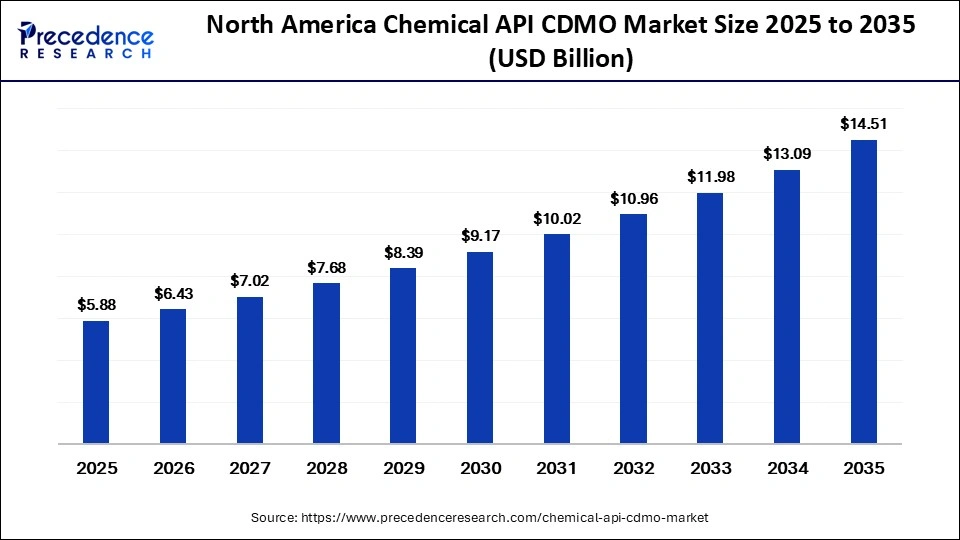
North America Chemical API CDMO Market Size and Growth 2026 to 2035
The North America chemical API CDMO market size is estimated at USD 5.88 billion in 2025 and is projected to reach approximately USD 14.51 billion by 2035, with a 9.45% CAGR from 2026 to 2035.
Why Did North America Lead the Chemical API CDMO Market in 2025?
North America led the chemical API CDMO market with a share of 35% in 2025, driven by high drug production volumes, strong regulatory frameworks, and significant investment in R&D. The region benefits from a well-established ecosystem of pharmaceutical and biotechnology companies , which are increasingly outsourcing API development and manufacturing to CDMOs. Growing demand for complex and high-value drug molecules, particularly in oncology and specialty therapeutics, is further supporting market growth. Outsourcing is also driven by the need to reduce operational costs while maintaining stringent FDA-compliant quality standards.
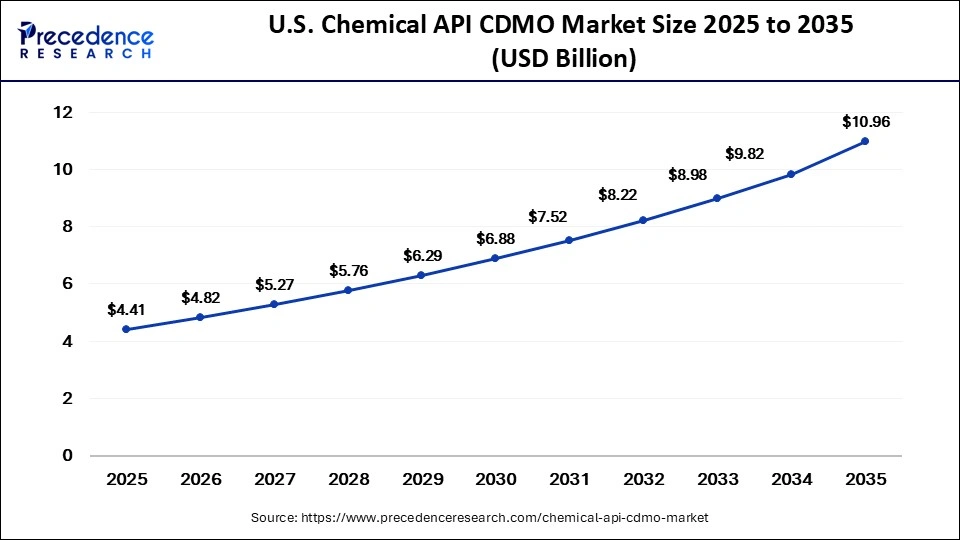
U.S. Chemical API CDMO Market Size and Growth 2026 to 2035
The U.S. chemical API CDMO market size is calculated at USD 4.41 billion in 2025 and is expected to reach nearly USD 10.96 billion in 2035, accelerating at a strong CAGR of 9.53% between 2026 and 2035.
U.S. Market Insights
The U.S. is a major contributor to the North American chemical API CDMO market, supported by a highly advanced pharmaceutical innovation ecosystem and substantial R&D investments. Major U.S.-based pharmaceutical companies increasingly rely on CDMOs for scalable and cost-efficient API production, particularly for complex and high-potency compounds. The country's strong regulatory framework ensures high manufacturing standards, further strengthening its position in the global CDMO landscape.
Europe: The Second-Largest Market
Europe is the second-largest market for chemical API CDMOs, holding a 30% market share in 2025, and is expected to grow at a notable CAGR of 7% during the projection period. The region benefits from a strong pharmaceutical manufacturing base, robust regulatory systems, and rising demand for biosimilars and specialty drugs. European CDMOs are recognized for high-quality standards, innovation, and expertise in complex chemical synthesis. Market growth is further supported by increasing focus on supply chain security and the reshoring of pharmaceutical manufacturing.
UK Market Analysis
The UK plays a significant role in the European market, supported by a strong pharmaceutical research base and a globally recognized life sciences industry. The country has a growing focus on innovative therapeutics, including biologics and precision medicine, which increases demand for specialized manufacturing capabilities. Government initiatives supporting life sciences innovation and investment in R&D infrastructure are further strengthening the market environment.
How is the Opportunistic Rise of Asia Pacific in the Chemical API CDMO Market?
Asia Pacific held a 25% share of the chemical API CDMO market in 2025 and is expected to grow at the fastest CAGR of 14.5% during the projection period. This growth is driven by cost-efficient manufacturing capabilities, large-scale drug production, and increasing outsourcing to countries such as China and India. Rising healthcare demand and supportive government policies promoting pharmaceutical exports are also contributing to market expansion. Global pharmaceutical companies are increasingly partnering with Asian CDMOs to enhance production efficiency and reduce costs.
China Market Analysis
The market in China is driven by a strong manufacturing ecosystem and increasing global outsourcing demand. The country benefits from large-scale production capabilities, an extensive chemical manufacturing infrastructure, and government support for pharmaceutical exports. Additionally, China is investing in regulatory upgrades to align with international standards, strengthening its global competitiveness.
Chemical API CDMO Market Companies
- Lonza Group
- WuXi AppTec
- Samsung Biologics
- Boehringer Ingelheim
- Recipharm AB
- BASF SE
- Cipla Limited
- Teva Pharmaceuticals Industries Ltd.
- Siegfried Holding AG
- Patheon (Thermo Fisher Scientific Inc.)
- Hovione
- STADA Arzneimittel AG
- Mylan (Viatris Inc.)
- Fujifilm Diosynth Biotechnologies
- Evonik Industries AG
Recent Developments
- In January 2026, the Eurofins CDMO Alphora developed a high-throughput screening of salts and co-crystals by effectively utilizing AI to aid in the development of solid-state pharmaceuticals. The system uses the collaboration with a Canadian university in Mississauga that provides prediction of the creation of salt and co-crystals with active pharmaceutical ingredients, thereby raising the efficiency and reducing the volume of research.(Source: https://www.businesswire.com )
- In November 2025, LMS, a subsidiary company of Lupin Limited, commissioned a special Oncology Block in its Vizag plant in India. The new high-containment unit improves its end-to-end CDMO service production in high-potency active-pharmaceutical ingredient production, specifically, the production of oncology drugs.(Source: https://www.lupin.com )
- In February 2025, SK Pharmteco expanded its performance with a state-of-the-art analytical testing laboratory dedicated to the high-potency active pharmaceutical ingredients (HPAPIs) field. This facility is included in its array of CDMO services by enhancing the accuracy of tests, safety, and regulatory compliance in sophisticated drug development.(Source: https://www.skpharmteco.com )
Segments Covered in the Report
By Service Type
- API Manufacturing
- Formulation Development
- Contract Research & Development (R&D)
- Regulatory Services
- Packaging Services
By Drug Type
- Branded Drugs
- Generic Drugs
- Biosimilars
- Over-the-Counter (OTC) Drugs
By Therapeutic Area
- Oncology
- Cardiovascular
- Neurology
- Infectious Diseases
- Endocrinology
- Immunology
- Others
By API Type
- Small Molecule APIs
- Large Molecule APIs (Biologics, Biosimilars)
- Peptide-based APIs
- Monoclonal Antibodies (mAbs)
By Outsourcing Model
- Full-service CDMOs (providing end-to-end services from discovery to commercialization)
- Dedicated capacity CDMOs (providing a dedicated facility for a single client)
By Scale of Manufacturing
- Preclinical & Clinical Manufacturing
- Commercial Manufacturing
By Manufacturing Technology
- Continuous Manufacturing
- Batch Manufacturing
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Tags
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content






 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
