What is the Recombinant Cell Culture Supplements Market Size?
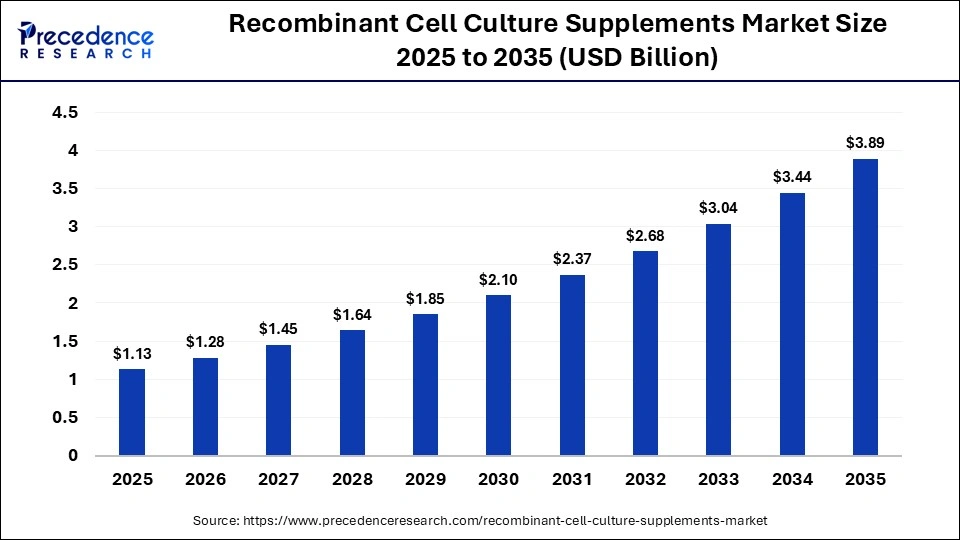
The global recombinant cell culture supplements market size accounted for USD 1.13 billion in 2025 and is predicted to increase from USD 1.28 billion in 2026 to approximately USD 3.89 billion by 2035, expanding at a CAGR of 13.16% from 2026 to 2035. A widespread expansion of the recombinant cell culture supplements is driven by the robust research, manufacturing, and bioprocessing of recombinant growth factors, transport proteins, process enzymes, and other supplements by the leading biotechnology and pharmaceutical firms.
Market Highlights
- North America dominated the market in 2025, with a revenue share of approximately 38%.
- Asia Pacific is expected to grow at the fastest CAGR of 13.21% from 2026 to 2035.
- By product, the recombinant albumin segment dominated the market in 2025, with a market share of approximately 38%.
- By product, the recombinant growth factors/cytokines segment in the market is expected to grow at the fastest CAGR of 9.1% between 2026 and 2035.
- By application, the biopharmaceutical production segment dominated the market in 2025, with a market share of approximately 55%.
- By application, the regenerative medicine & cell therapy segment is expected to grow at a notable rate of 8.9% from 2026 to 2035.
- By formulation/type, the powder segment dominated the market in 2025, with a market share of approximately 53%.
- By formulation/type, the liquid segment is expected to grow at the fastest CAGR of 8.8% between 2026 and 2035.
- By end-use, the pharmaceutical & biotechnology companies segment dominated the market in 2025, with a market share of approximately 38%.
- By end-use, the CDMO/CRO segment is expected to grow at the fastest CAGR of 9% from 2026 to 2035.
Bioprocessing Recombinant Supplements: Fundamental Nutrition Needs of Cell Culture Processes
The recombinant cell culture supplements market comprises biologically engineered supplements (e.g., albumin, insulin, growth factors, cytokines) used to support, sustain, and enhance in vitro cell culture systems in research and biomanufacturing. These defined, animal-free components improve consistency, scalability, and safety in biopharmaceutical production, vaccine development, regenerative medicine, and cell & gene therapy. Increasing biologics output, regulatory pressure for xeno-free processes, and demand for high-quality media drive adoption globally.
The leading biopharmaceutical companies, like Merck, have introduced potential supplements that are used as the animal-free substitute for human/bovine serum albumin, to replace serum transferrin with recombinant insulin and albumin, and in upstream processes for mAbs, viral vaccines, and stem cell expansion. Recombinant manufacturing in cGMP production and state-of-the-art facilities ensures a reliable and consistent supply of products while simplifying regulatory processes.
How does AI Revolutionize the Recombinant Cell Culture Supplements Market?
Artificial intelligence integrated with automation improves laboratory research, renowned as cell culture, which further expands the recombinant cell culture supplements market. AI and automated cell culture procedures improve cell culture protocols and lead to better conclusions in research and drug development. AI algorithms examine data from cameras and sensors to detect variations in cells, including morphology, appearance, and growth rate.
Recombinant Cell Culture Supplements Market Trends
- High-Quality Recombinant Insulin for Cell Culture: The recombinant insulin products are reliable cell culture media supplements that offer precise performance across various bioproduction systems. They deliver improved cell growth, high protein yield, and high purity, further driving the recombinant cell culture supplements market.
- Advancements in Protein Expression Systems: There is a vast use of different protein expression systems like E. coli, S. cerevisiae, Baculovirus/Insect cells, and mammalian cells for stable expression. The widely used E. coli systems are less expensive, enable rapid expression, require simple process scale-up, and have well-characterized genetics.
Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 1.13 Billion |
| Market Size in 2026 | USD 1.28 Billion |
| Market Size by 2035 | USD 3.89 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 13.16% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, Application, Formulation/Type, End-Use, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Segmental Insights
Product Insights
Why Did the Recombinant Albumin Segment Dominate the Recombinant Cell Culture Supplements Market in 2025?
The recombinant albumin segment dominated the market in 2025 with a revenue share of approximately 33%, owing to the expanded and consistent supply of more efficient and more stable products for biopharmaceutical innovation. Research and development focus on the sustainable production of functional human serum albumin, and it is widely used in medicine and biotechnology. The strategic partnerships among major biopharmaceutical firms allow the commercialization of recombinant human albumin across various regions. In November 2025, Evonik partnered with InVitria to expand access to animal-free human serum albumin for next-generation biopharmaceutical processes and advanced therapies.
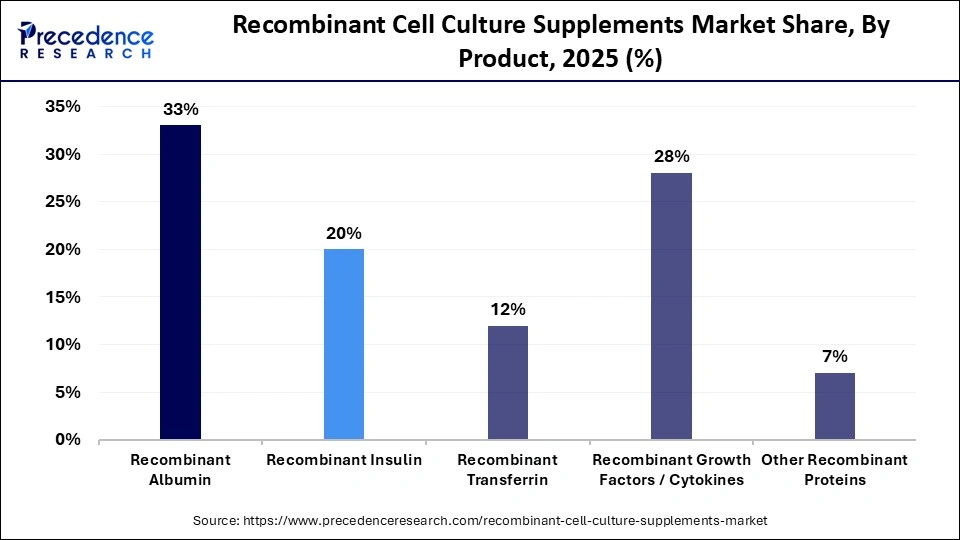
The recombinant growth factors/cytokines segment is expected to grow at the fastest CAGR of 9.1% in the recombinant cell culture supplements market during the forecast period due to the major role of cytokines like interleukins and interferons in mediating immune signaling, and growth factors like EGF and FGF in regulating cell proliferation and differentiation. The cytokines like interleukins (ILs), interferons (IFNs), and tumor necrosis factors (TNFs), and growth factors like epidermal growth factor (EGF), fibroblast growth factor (FGF), and vascular endothelial growth factor (VEGF) are essential tools in immunology, stem cell research, and cell culture. They are advancing biomedical research and drug development through precise and reliable biological activities.
Application Insights
Why Did the Biopharmaceutical Production Segment Hold the Largest Revenue Share in the Recombinant Cell Culture Supplements Market in 2025?
The biopharmaceutical production segment dominated the market in 2025 with a revenue share of approximately 55%, owing to the rising trends in biopharmaceuticals and biotechnology, which include resource allocation, precision medicine, personalized therapies, AI-driven clinical monitoring, patient safety, and many more. Researchers and manufacturers strictly evaluate long-term safety and efficacy studies of novel products. The potential sponsors highly invest in major therapeutic areas with the highest return on investment (ROI).
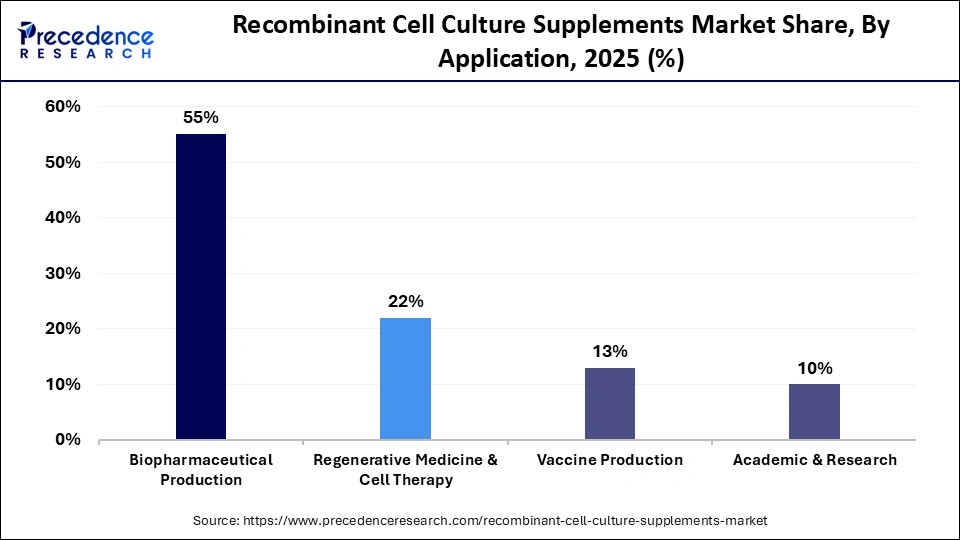
The regenerative medicine & cell therapy segment is estimated to grow at the fastest rate of 8.9% in the recombinant cell culture supplements market during the predicted timeframe due to the huge potential of these two innovative treatments to transition the cure of a wide range of illnesses and accidents. The novel discoveries in regenerative medicine enable researchers to restore body functions to replace damaged or sick cells, tissues, or organs with healthy ones. A treatment of neurological illnesses, including Parkinson's disease, is one of the promising areas for stem cell therapy.
Formulation/Type Insights
How Was the Remarkable Growth of the Powder Segment in the Recombinant Cell Culture Supplements Market in 2025?
The powder segment dominated the market in 2025 with a revenue share of approximately 53%, owing to the advantages of dry powder formulations such as controlled drug release, patient compliance, storage stability, and reduced local and systemic toxicities. The powder formulations have enhanced shelf life and stability, and enable efficient logistics. They have versatile applications in biopharmaceutical production, nutrition, research, and diagnostics.
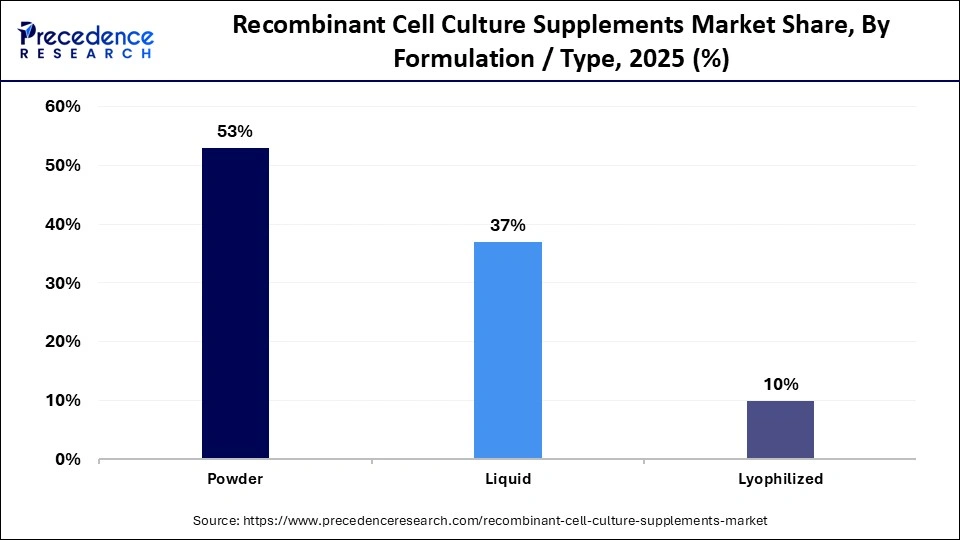
The liquid segment is anticipated to grow at a notable rate of 8.8% in the recombinant cell culture supplements market during the upcoming period due to enhanced operational efficiency, reduced costs, and streamlined preparations compared to powder formulations. These benefits of liquid formulations improve the cost-effectiveness of therapies and allow resource allocation to patient care. They optimize healthcare delivery across different regional healthcare systems.
End-Use Insights
What Makes the Pharmaceutical & Biotechnology Companies the Dominant Segment in the Recombinant Cell Culture Supplements Market in 2025?
The pharmaceutical & biotechnology companies segment dominated the market in 2025 with a revenue share of approximately 38%, owing to robust R&D in vaccines, biologics, recombinant protein products, drug discovery, and public-private partnerships. The major investments are marked by the global leaders like Aurobindo, Glaxosmithkline (GSK), Dr. Reddy's, DuPont, Novartis, Ferring Pharmaceuticals, Biological E. Limited, etc. The global players like Novo Nordisk and Eli Lilly are expanding their manufacturing footprint to meet global consumer demands.
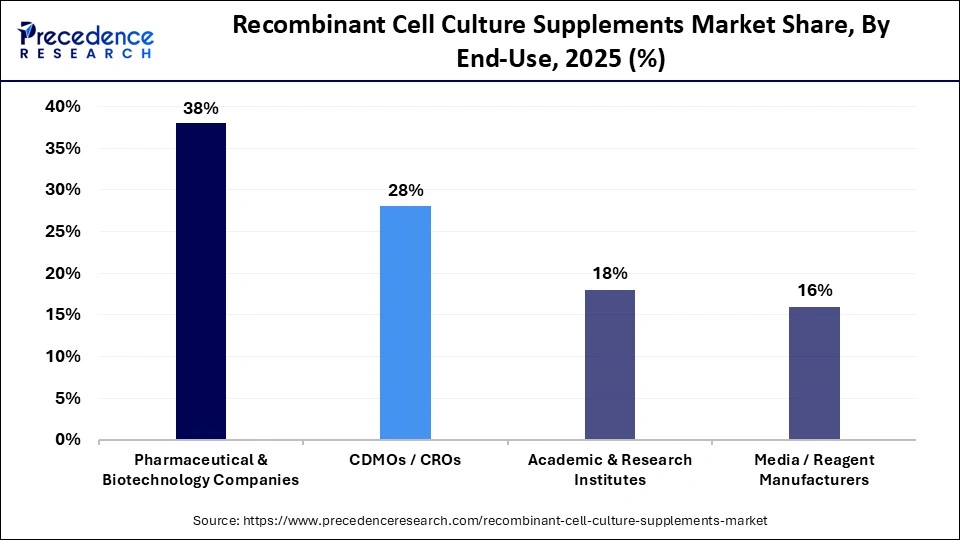
The CDMOs/CROs segment is predicted to grow at a rapid rate of 9% in the recombinant cell culture supplements market during the studied period due to investments and research focused on proteins, peptides, antibody-drug conjugates, monoclonal antibodies, etc. The new policies are introduced for the growth of CROs, CDMOs, and biotechnology. Large and medium-sized biopharmaceutical companies prefer to make partnerships with CDMOs or CROs.
Regional Insights
How Big is the North America Recombinant Cell Culture Supplements Market Size?
The North America recombinant cell culture supplements market size is estimated at USD 429.40 million in 2025 and is projected to reach approximately USD 1,486.60 million by 2035, with a 13.21% CAGR from 2026 to 2035.
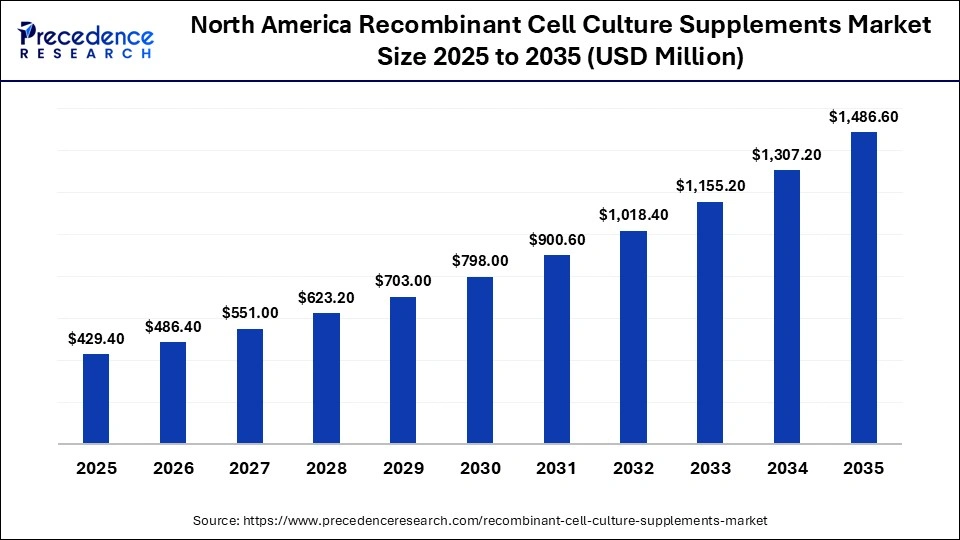
How Does North America Dominate the Recombinant Cell Culture Supplements Market in 2025?
North America dominated the market in 2025 with a revenue share of approximately 38%, owing to the increased demand for specialized cell culture media and recombinant supplements, novel cell-based therapeutics, and significant investments in advanced life sciences research. A Senate bill was launched to establish a National Biopharmaceutical Manufacturing Center of Excellence (COE) for accelerating America's biopharmaceutical manufacturing and advancing the biotechnology industry. In April 2025, Merck launched a Biologics Center of Excellence, a commercial production capacity, with the investment of $1 billion in Wilmington, Delaware. Merck is investing in American innovation and infrastructure with the integration of laboratory, manufacturing, and warehouse capabilities in its new state-of-the-art facility.
What is the Size of the U.S. Recombinant Cell Culture Supplements Market?
The U.S. recombinant cell culture supplements market size is calculated at USD 330.64 million in 2025 and is expected to reach nearly USD 1,152.12 million in 2035, accelerating at a strong CAGR of 13.30% between 2026 and 2035.
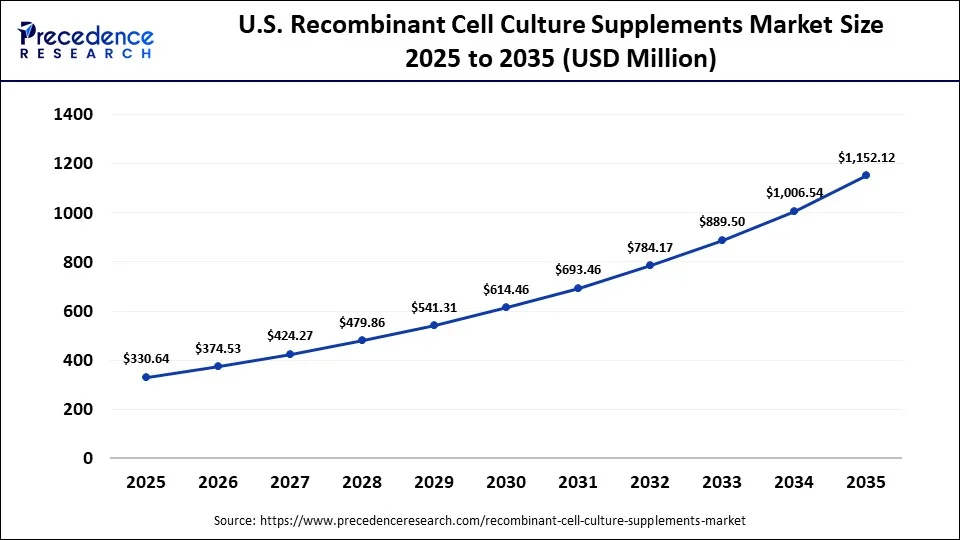
U.S. Recombinant Cell Culture Supplements Market Analysis
The U.S. industry for recombinant cell culture supplements is revolutionizing due to supply chain resilience across chemically defined recombinant alternatives, reduced biopharmaceutical timelines, and expanded commercialization of new products across the recombinant cell culture supplements market. In September 2025, GSK announced the investment of $30 billion in the U.S. R&D, manufacturing, and supply chain infrastructure over the next 5 years. The U.S. Department of Defense (DOD) introduced potential opportunities for American businesses through its distributed bioindustrial manufacturing program to advance biotechnology and domestic supply chains.
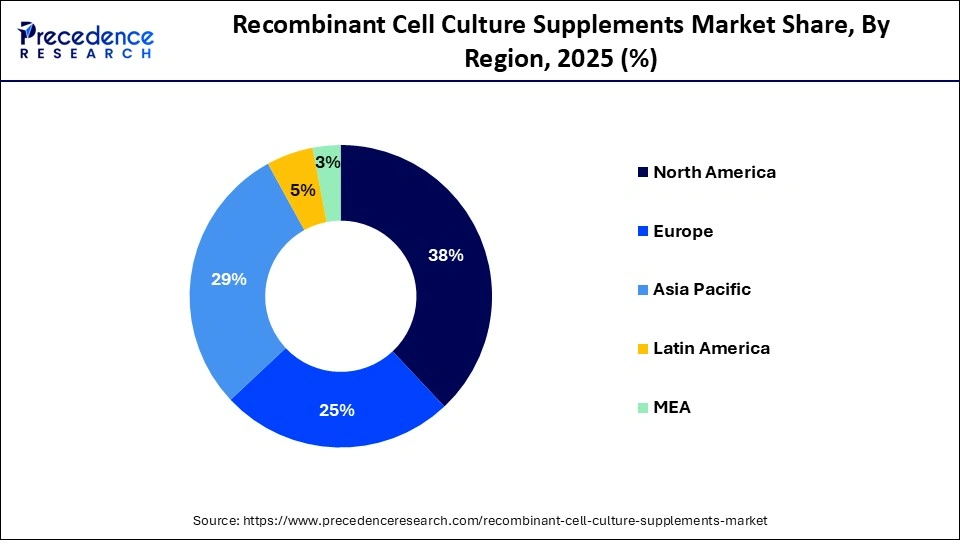
What is the Asia Pacific Recombinant Cell Culture Supplements Market Size?
The Asia Pacific recombinant cell culture supplements market size is expected to be worth USD 976.80 million by 2035, increasing from USD 282.50 million by 2025, growing at a CAGR of 13.21% from 2026 to 2035.
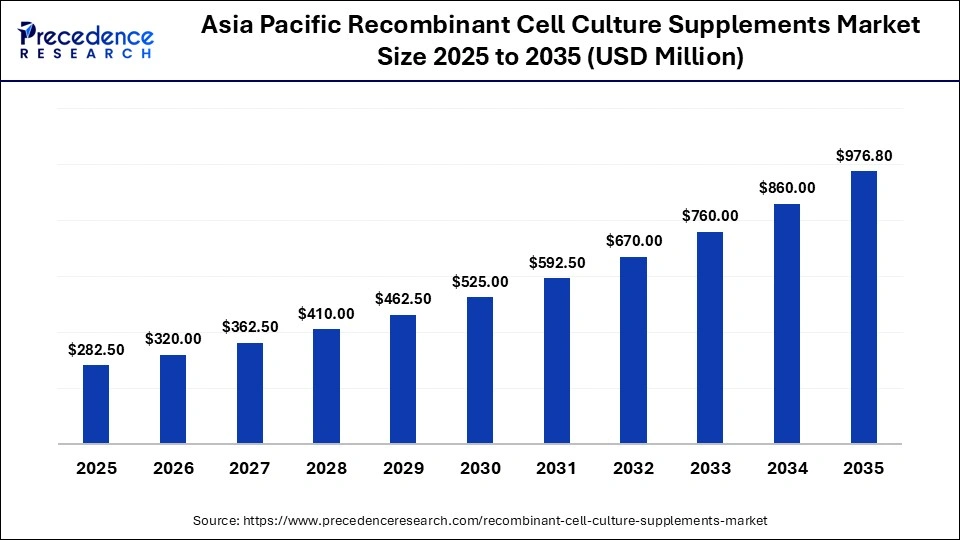
What Is the Potential of the Recombinant Cell Culture Supplements Market in the Asia Pacific?
Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period due to expanded clinical trials, highly efficient cell and gene therapies, and biotechnology innovation across the recombinant cell culture supplements market. The major pillars for this regional expansion are manufacturing infrastructure, regulatory environment, a specialized workforce, innovation hubs across biotechnology and research, and funding from public-private partnerships. The Asian Pacific countries, like India, China, South Korea, and Singapore, have launched biotechnology frameworks, including new manufacturing sites, fast-track approvals, incentives, funding schemes, and talent programs.
India Recombinant Cell Culture Supplements Market Analysis
India is advancing with the rise of Contract Development and Manufacturing Organizations (CDMOs), bulk biomanufacturing of vaccines and biosimilars, and the great adoption of recombinant supplements to enhance production yields for global end-users. The Government of India, the Department of Biotechnology (DBT), and the Biotechnology Industry Research Assistance Council (BIRAC) are committed to promoting entrepreneurship and indigenous manufacturing in the biopharma sector.
Recombinant Cell Culture Supplements Market Value Chain Analysis

Who are the Major Players in the Global Recombinant Cell Culture Supplements Market?
The major players in the recombinant cell culture supplements market include Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, Sartorius AG, Corning Incorporated, BBI Solutions, FUJIFILM Irvine Scientific Inc., Abcam plc., GEMINI Bio-Products LLC, HiMedia Laboratories, InVitria , Sino Biological Inc., STEMCELL Technologies Inc., Novus Biologicals LLC, and R&D Systems.
Recent Developments
- In August 2025, Thermo Fisher Scientific Inc. launched the next-generation expression system, specifically designed to deliver improved protein yields for various low-yield and challenging proteins with speed and high-throughput to expand protein research.(Source: https://www.biospectrumindia.com)
- In August 2025, Sartorius AG introduced customized CHO cell line development solutions for every protein of interest by conducting a case study to overcome challenges in the production of difficult-to-express and complex proteins.(Source: https://www.sartorius.com)
Segments Covered in the Report
By Product
- Recombinant Albumin
- Recombinant Insulin
- Recombinant Transferrin
- Recombinant Growth Factors/Cytokines
- Other Recombinant Proteins
By Application
- Biopharmaceutical Production
- Regenerative Medicine & Cell Therapy
- Vaccine Production
- Academic & Research
By Formulation/Type
- Powder
- Liquid
- Lyophilized
By End-Use
- Pharmaceutical & Biotechnology Companies
- CDMOs/CROs
- Academic & Research Institutes
- Media/Reagent Manufacturer
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
Frequently Asked Questions
Ask For Sample
No cookie-cutter, only authentic analysis – take the 1st step to become a Precedence Research client
 Get a Sample
Get a Sample
 Table Of Content
Table Of Content




.webp)

 sales@precedenceresearch.com
sales@precedenceresearch.com
 +1 804-441-9344
+1 804-441-9344
 Schedule a Meeting
Schedule a Meeting
